Terms Soluble Insoluble Saturated solution Unsaturated solution Supersaturated

Terms • Soluble • Insoluble • Saturated solution • Unsaturated solution • Supersaturated solution • Concentration • Molarity • Dilution 1

2 Chapter 15 SOLUTIONS

Aqueous Solutions Some solutions contain ELECTROLYTES HCl, Mg. Cl 2, and Na. Cl are strong electrolytes. - dissociate completely into ions. weak electrolytes. - dissociate a little into ions. 3

Aqueous Solutions Some compounds dissolve in water but do not conduct electricity. They are called: nonelectrolytes. Examples include: sugar ethanol ethylene glycol 4

Colligative Properties depend on the concentration of particles in a solution, not of the identity of the particles. q Boiling Point Elevation q Freezing Point Depression q Osmotic Pressure 5

Freezing Point Depression Each mole of solute particles lowers the freezing point of 1 kilogram of water by 1. 86 degrees Celsius. Boiling Point Elevation Each mole of nonvolatile solute particles raises the boiling point of 1 kilogram of water by 0. 51 degrees Celsius. 6

7 Electrolyte or Nonelectrolyte! Test each sample to see if it is a strong electrolyte, weak electrolyte, or nonelectrolyte.

Parts of a Solution • SOLUTE –part that is being dissolved • SOLVENT – part that dissolves the solute • Solute + Solvent = Solution 8
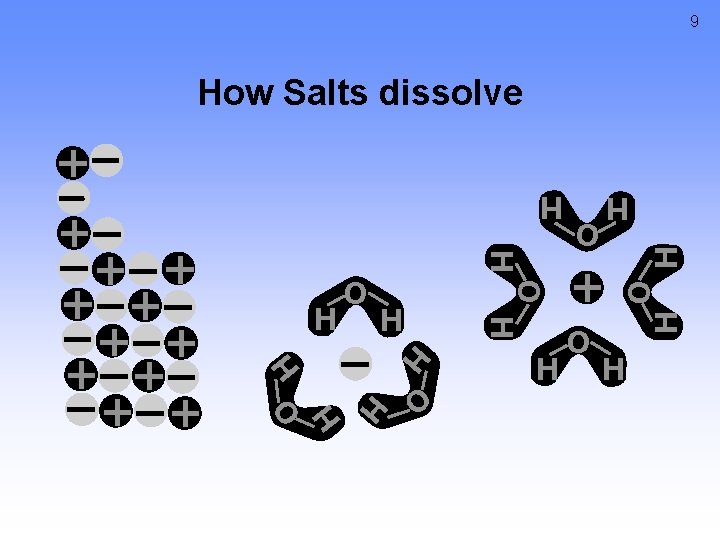
9 How Salts dissolve H O H H H O H
10 Dissolving and Solutions
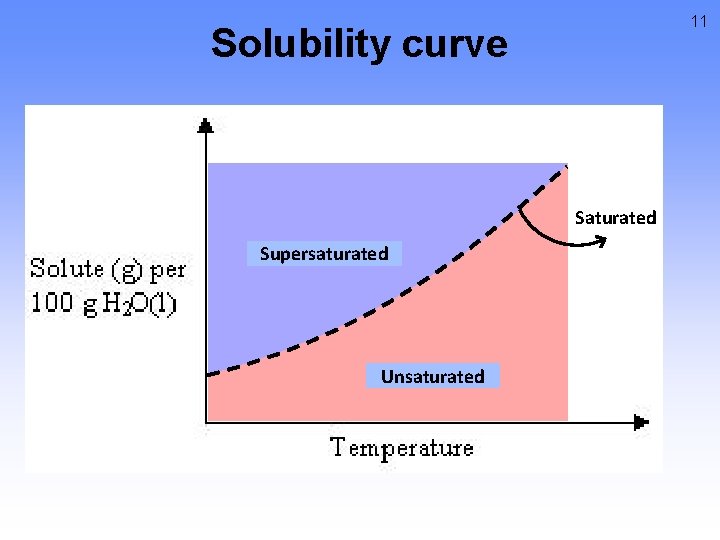
11 Solubility curve Saturated Supersaturated Unsaturated
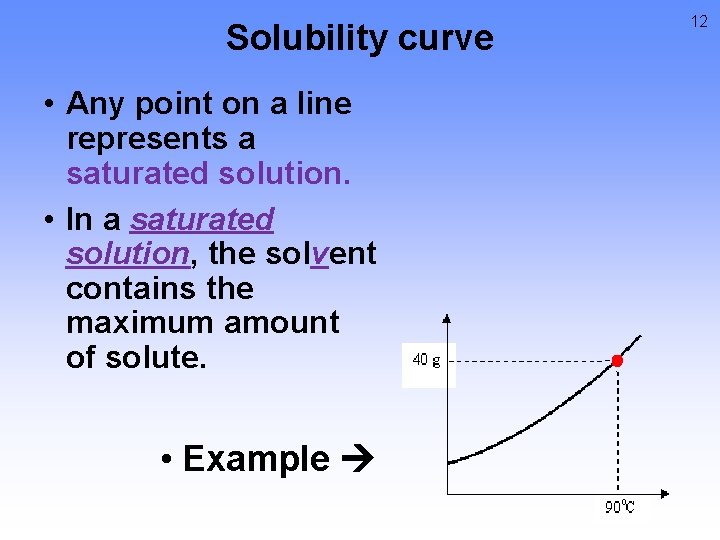
Solubility curve • Any point on a line represents a saturated solution. • In a saturated solution, the solvent contains the maximum amount of solute. • Example 12
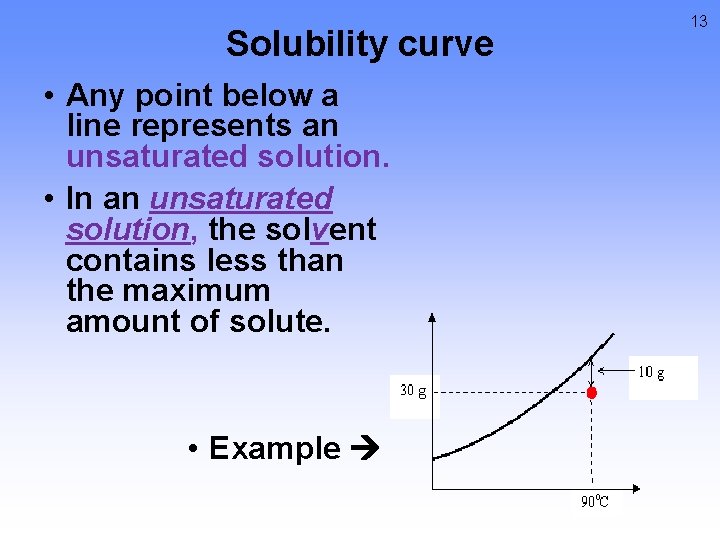
Solubility curve • Any point below a line represents an unsaturated solution. • In an unsaturated solution, the solvent contains less than the maximum amount of solute. • Example 13
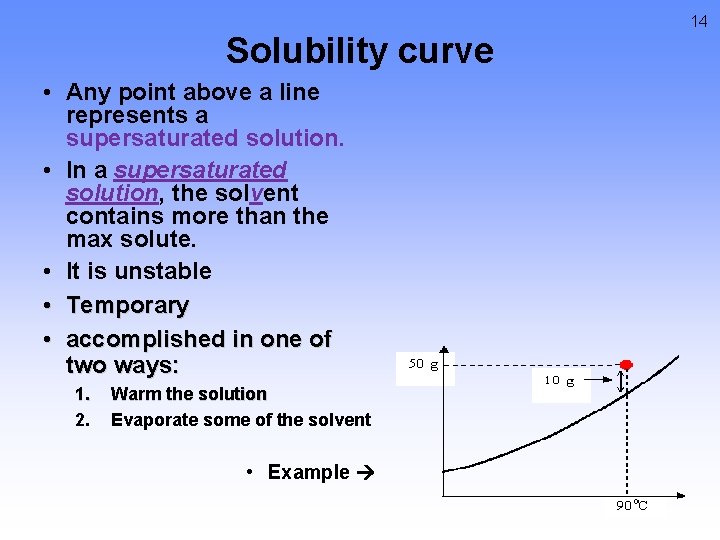
14 Solubility curve • Any point above a line represents a supersaturated solution. • In a supersaturated solution, the solvent contains more than the max solute. • It is unstable • Temporary • accomplished in one of two ways: 1. 2. Warm the solution Evaporate some of the solvent • Example
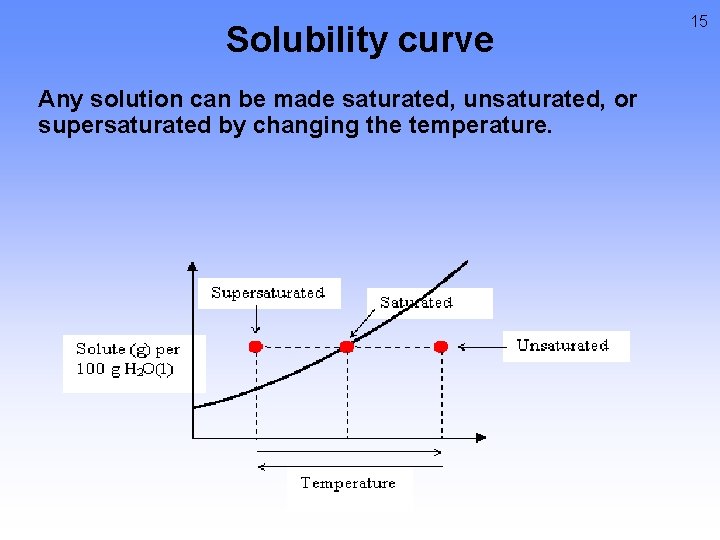
Solubility curve Any solution can be made saturated, unsaturated, or supersaturated by changing the temperature. 15

Solubility Curve Determine if each of the following is unsaturated, or supersaturated. • 55 g of NH 3 at 20 o. C. _____ • 80 g of Na. NO 3 at 10 o. C. _____ • 10 g of Ce 2(SO 4)3 at 10 o. C_____ • 145 g of Na. NO 3 at 80 o. C. _____ • 25 g of KNO 3 at 60 o. C. _____ • 35 g of Na. Cl at 100 o. C. _____ • 65 g of NH 4 Cl at 80 o. C. _______ • 12 g of NH 3 at 90 o. C. _______ 16

The concentration of a solution tells you how much solute that is dissolved in a given amount of solvent (water). The molarity is the concentration of a solution. Molarity = Moles Liter 1. 2. 255 moles of Na. Cl is dissolved in 4. 0 L of water, calculate the molarity. Molarity = = 0. 56 moles/liter = 0. 56 M (Molar) where M means moles per 1 liter 17

18 2. 5. 00 g KOH is dissolved in 250. m. L of water, calculate the molarity. Molarity = = 0. 356 M
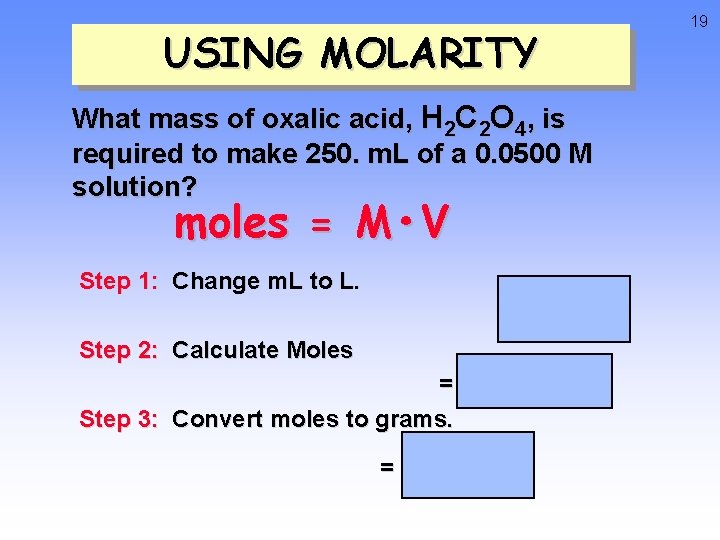
USING MOLARITY What mass of oxalic acid, H 2 C 2 O 4, is required to make 250. m. L of a 0. 0500 M solution? moles = M • V Step 1: Change m. L to L. = 0. 250 L Step 2: Calculate Moles = 0. 0125 moles Step 3: Convert moles to grams. = 1. 13 g 19

20 Molarity Practice • What is the molarity of an aqueous solution containing 40. 0 g of glucose (C 6 H 12 O 6) in 1. 5 m. L of solution? • What is the molarity of bleach solution containing 9. 5 g of Na. OCl per liter of bleach?

21 3. How many moles are there in 205. m. L of a 0. 172 M solution? 0. 0353 moles

22 4. How many grams Na. Cl are there in 250. 0 m. L of 0. 500 M solution? 7. 31 g

23 Dilutions

Diluting Solutions Concentrated solutions have a relatively high molarity. Dilute solutions have a relatively low molarity. M 1 V 1 = M 2 V 2 M 1= the initial molarity M 2 = the final molarity V 1 = the initial volume V 2 = the final volume 24
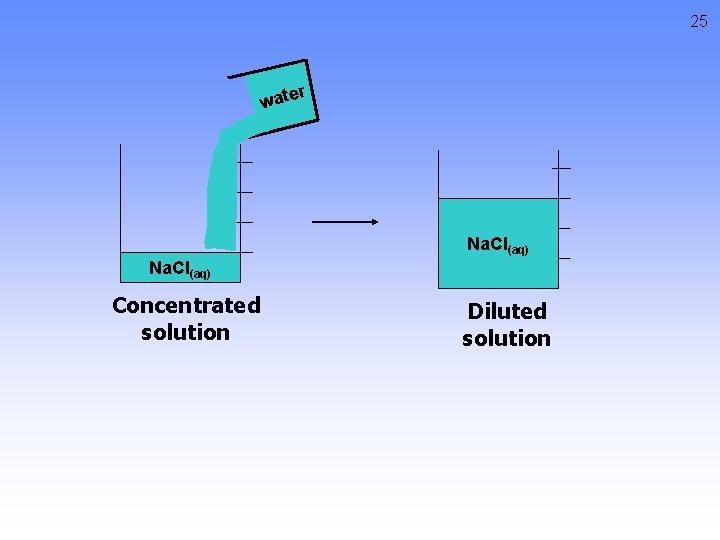
25 r wate Na. Cl(aq) Concentrated solution Diluted solution

Initial Solution 26 1. 25. 0 m. L of 0. 10 M solution is diluted by adding 75. 0 m. L of water. Calculate the molarity of the new solution. M 1 V 1 = M 2 V 2 M 1= 0. 10 M M 2 = ? V 1 =25. 0 m. L + 75. 0 m. L V 2 = 100. 0 m. L (0. 10 M)(25. 0 m. L) = M 2(100. 0 m. L) M 2 = 0. 025 M

Initial Solution 2. What volume of 0. 250 M Co. Cl 2 solution can be diluted to 100. 0 m. L in order to make a 0. 0150 M solution? M 1 V 1 = M 2 V 2 M 1= 0. 250 M V 1 = ? M 2 = 0. 0150 M V 2 = 100. 0 m. L (0. 250 M) V 1 = (0. 0150 M)(100. 0 m. L) V 1 = 6. 00 m. L 27

Initial Solution 28 3. 50. 0 m. L of 0. 500 M Cu. SO 4 solution is diluted to 250. 0 m. L. Calculate the new concentration and the number of grams Cu. SO 4 in the new solution. M 1= 0. 500 M V 1 = 50. 0 m. L M 2 = ? M 1 V 1 = M 2 V 2 = 250. 0 m. L (0. 500 M)(50. 0 m. L) = M 2(250. 0 m. L) M 2 = 0. 100 M 0. 0500 L x 0. 500 mol x 159. 6 g = 3. 99 g 1 L 1 mol

29 Practice 1) If I add 25 m. L of water to 125 m. L of a 0. 15 M Na. OH solution, what will the molarity of the diluted solution be? 2) If I add water to 100 m. L of a 0. 15 M Na. OH solution until the final volume is 150 m. L, what will the molarity of the diluted solution be? 3) How much 0. 05 M HCl solution can be made by diluting 250 m. L of 10 M HCl?

30 Practice 4) I have 345 m. L of a 1. 5 M Na. Cl solution. If I boil the water until the volume of the solution is 250 m. L, what will the molarity of the solution be? 5) How much water would I need to add to 500 m. L of a 2. 4 M KCl solution to make a 1. 0 M solution?

Practice 6. 734 grams of lithium sulfate are dissolved to make 2500. m. L of solution. 7. 6. 70 x 10 -2 grams of Pb(C 2 H 3 O 2)4 are dissolved to make 3. 50 m. L of solution. 8. 83 grams of sodium hydroxide to 750 m. L of water. 31
- Slides: 31