Teratogenic Drug Antibiotic for pregnancy Dr Jarir At

Teratogenic Drug & Antibiotic for pregnancy Dr. Jarir At Thobari, Pharm. D, Ph. D 1, 2 1 Dept. Pharmacology and Pharmacotherapy 2 Clinical Epidemiology & Biostatistic Unit Fac. Medicine Universitas Gadjah Mada 1
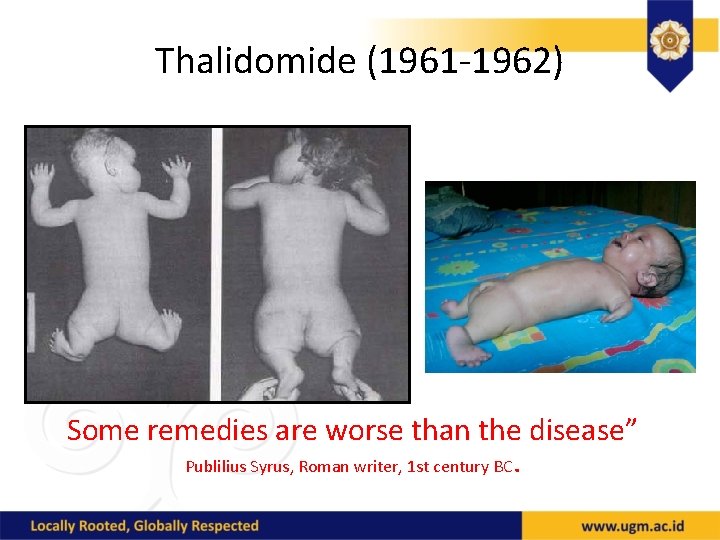
Thalidomide (1961 -1962) Some remedies are worse than the disease” Publilius Syrus, Roman writer, 1 st century BC.

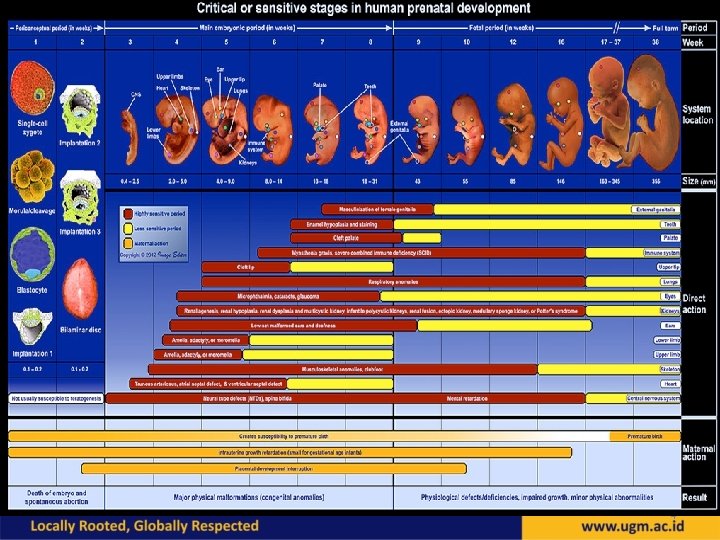
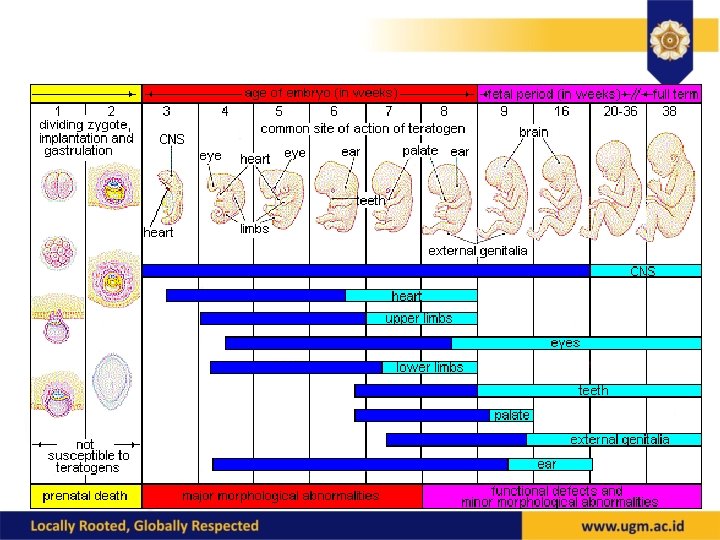
Fetal Development: Period of High Susceptibility • Period of greatest concern begins at 15 -18 days when organogenesis begins • Reaches a peak at 30 days postconception • Susceptibility decreases until days 55 -60 and becomes minimal through day 90
4

General approach • Virtually all drugs cross to some degree • We really don’t know much about what a given drug can or cannot do • Use any drug only when necessary, when risk/benefit indicates, and then only in minimal effective dose

Maternal/Fetal Transfer • • • Maternal/Fetal blood flow Placental binding Maternal, Placental, & Fetal metabolism Diffusion capacity Maternal & Fetal protein binding Maternal Dose/Drug Uptake

Placental drug transfer • Depends upon: – Physical and chemical properties of drug – Characteristics of maternal, fetal, and placental circulations – Anatomy and physiology of the placenta

Placental Transport • • Molecular weight Lipid solubility Ionization Concentration gradient Protein binding Changes in maternal physiology Route of delivery

Documented Teratogenic ACE Inhibitors Mercury Alcohol Phenytoin Androgens PPIs (some evidence) Antithyroid drugs Radiation (>5 rad) Carbamazepine Streptomycin/kanamycin Chemotherapy agents Isotretinoin (Accutane) Cocaine Tetracycline Coumadin Thalidomide Diethylstibestrol Trimethadione Lead Valproic acid Lithium Vit A derivatives (Retinoic acid)

Teratogen • Is an agent that can disturb the development of the embryo or fetus • Teratogens halt the pregnancy or produce a congenital malformation • Include radiation, maternal infections, chemicals, and drugs 11

Teratogenic drugs • directly or indirectly, causes a structural or functional change in the fetus or child if it is administrated to pregnant mother • If mother is taking this drug, It can be either stopped, switched or reduced to the lowest dose possible • typically during sensitive periods of fetal development • Depending on the particular teratogenic process and target organ 12

Teratogenesis �Birth defects are known to occur in 3 -5% of all newborns. �They are the leading cause of infant mortality in the United States, accounting for more than 20% of all infant deaths. �They can do direct damage to the fetus, causing abnormal development. �They can compromise the function of the placenta, often by constricting blood vessels and reducing the supply of oxygen and nutrients from the mother to the fetus. This can lead to underdevelopment or low birth weight, which are risk factors for birth defects. �They can trigger forceful uterine contractions, potentially injuring the fetus or prompting premature birth.

DRUGS PREGNANCY CATEGORIES CATEGORY A • failed to demonstr ate a risk to the fetus in the first trimester of pregnancy • no evidence of risk in later trimesters CATEGORY B • failed to demonstra te a risk to the fetus • no adequate and wellcontrolled studies in pregnant women. CATEGORY C • shown an adverse effect on the fetus • no adequate and wellcontrolled studies in humans • potential benefits may warrant the use of drug despite potential risks. CATEGORY D CATEGORY X • positive • demonstrates evidence of fetal human fetal abnormalities risk based • positive on adverse evidence of reaction human fetal data risk based on • potential adverse benefits reaction data may warrant • the risks use of the involved in drug in use of the pregnant drug in women pregnant despite women potential risks CATEGORY N • FDA has not classified the drug.
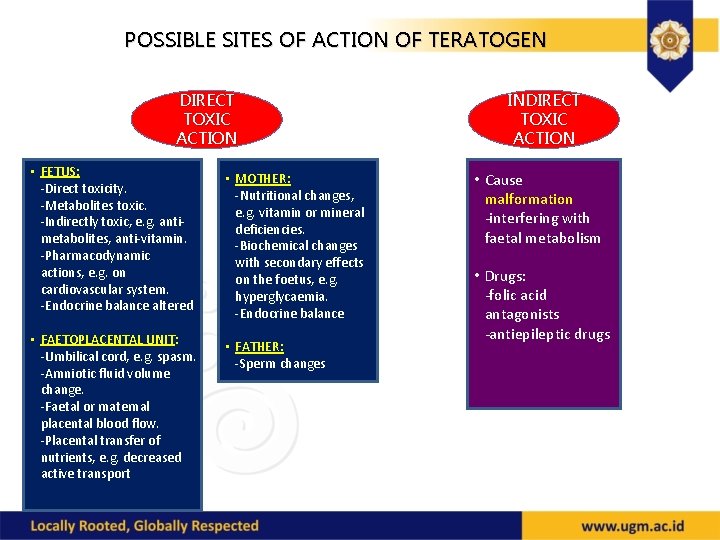
POSSIBLE SITES OF ACTION OF TERATOGEN DIRECT TOXIC ACTION • FETUS: -Direct toxicity. -Metabolites toxic. -Indirectly toxic, e. g. antimetabolites, anti-vitamin. -Pharmacodynamic actions, e. g. on cardiovascular system. -Endocrine balance altered • MOTHER: -Nutritional changes, e. g. vitamin or mineral deficiencies. -Biochemical changes with secondary effects on the foetus, e. g. hyperglycaemia. -Endocrine balance • FAETOPLACENTAL UNIT: -Umbilical cord, e. g. spasm. -Amniotic fluid volume change. -Faetal or maternal placental blood flow. -Placental transfer of nutrients, e. g. decreased active transport • FATHER: -Sperm changes INDIRECT TOXIC ACTION • Cause malformation -interfering with faetal metabolism • Drugs: -folic acid antagonists -antiepileptic drugs

TERATOGENIC MECHANISMS OF DRUGS • Folate antagonism • Neural crest cell disruption • Endocrine disruption • Oxidative stress • Vascular disruption and specific receptor • Enzyme-mediated teratogenesis

Teratogenic Drug (TERATOWA) • • Thalidomide Epileptic drugs (valproic acid/phenytoin) Retinoid (Vitamin A) ACE-inhbitors/ARC Third element (lithium) Oral contraceptives - hormone Warfarin Alcohol 17

Analgesic (aspirin) • Gastroschisis • Decrease prostaglandin decrease uterine contraction delayed onset of labor & prolonged period of pregnancy • During delivery severe bleeding because aspirin decrease platelet aggregation

Anticonvulsant • Fetal hydantoin syndrome : • cranio facial malformation: -Cleft lip and palate -Broad nasal bridge -Ocular hypertelorism -Abnormal ears • congenital heart disease • limb malformation • mental and growth retardation
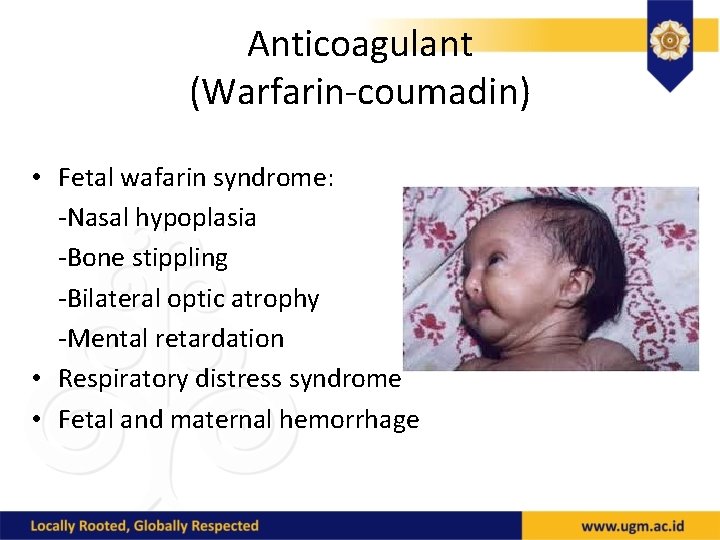
Anticoagulant (Warfarin-coumadin) • Fetal wafarin syndrome: -Nasal hypoplasia -Bone stippling -Bilateral optic atrophy -Mental retardation • Respiratory distress syndrome • Fetal and maternal hemorrhage

Antidepressant (imipramine) • • Cleft palate Defect in abdomen Adrenal hypoplasia Cardiovascular defect
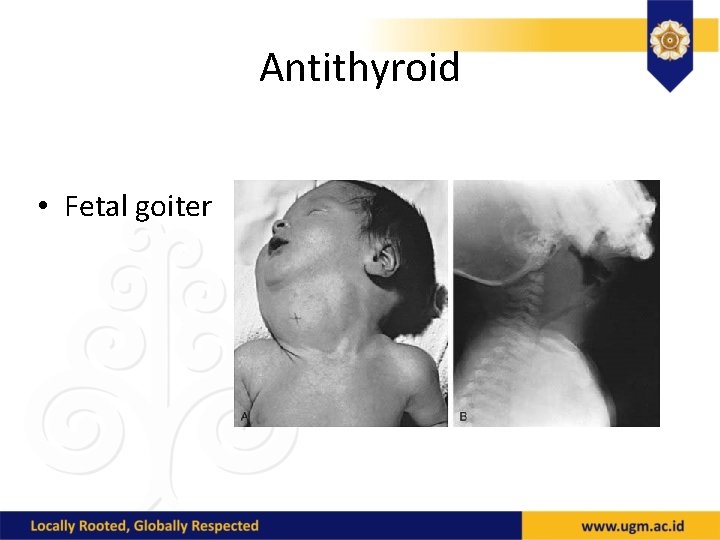
Antithyroid • Fetal goiter
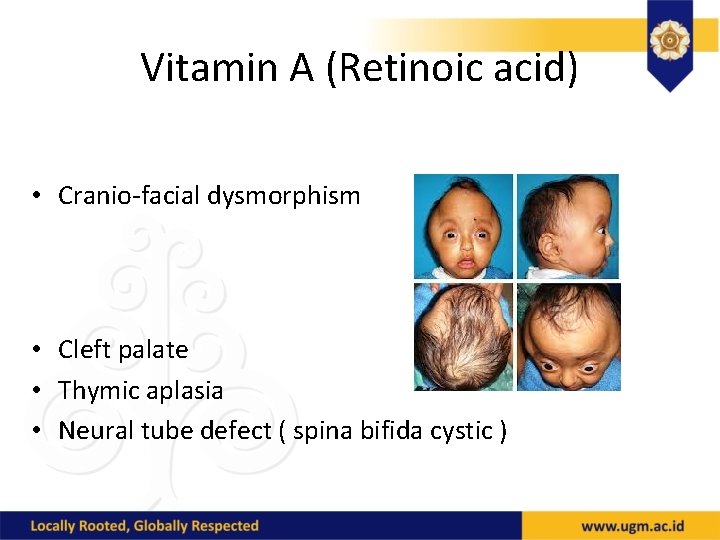
Vitamin A (Retinoic acid) • Cranio-facial dysmorphism • Cleft palate • Thymic aplasia • Neural tube defect ( spina bifida cystic )
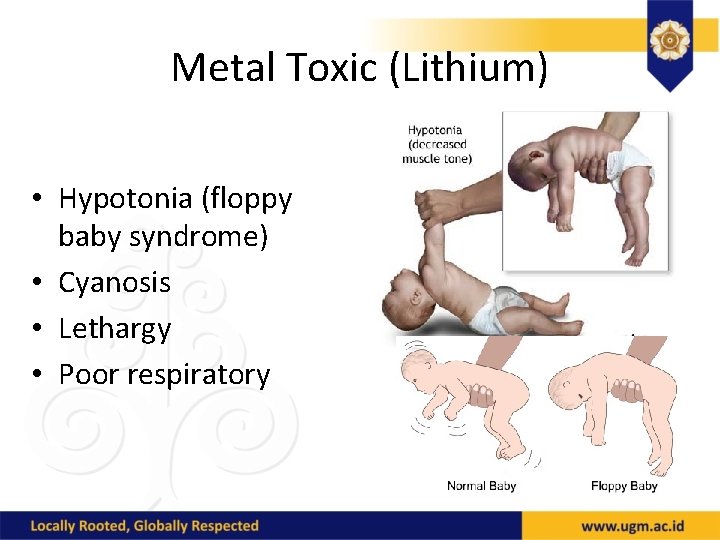
Metal Toxic (Lithium) • Hypotonia (floppy baby syndrome) • Cyanosis • Lethargy • Poor respiratory

Sedative-hypnotic (diazepam) • • • Cleft lip and palate Inguinal hernia Congenital heart disease Pyloric stenosis Breathing difficulties
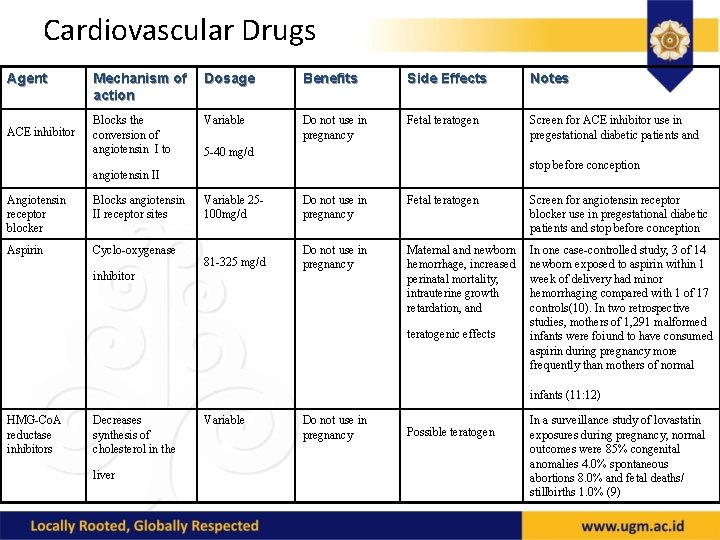
Cardiovascular Drugs Agent ACE inhibitor Mechanism of action Dosage Benefits Side Effects Notes Blocks the conversion of angiotensin I to Variable Do not use in pregnancy Fetal teratogen Screen for ACE inhibitor use in pregestational diabetic patients and 5 -40 mg/d stop before conception angiotensin II Angiotensin receptor blocker Blocks angiotensin II receptor sites Aspirin Cyclo-oxygenase Variable 25100 mg/d Do not use in pregnancy Fetal teratogen Screen for angiotensin receptor blocker use in pregestational diabetic patients and stop before conception 81 -325 mg/d Do not use in pregnancy Maternal and newborn hemorrhage, increased perinatal mortality, intrauterine growth retardation, and In one case-controlled study, 3 of 14 newborn exposed to aspirin within 1 week of delivery had minor hemorrhaging compared with 1 of 17 controls(10). In two retrospective studies, mothers of 1, 291 malformed infants were foiund to have consumed aspirin during pregnancy more frequently than mothers of normal inhibitor teratogenic effects infants (11: 12) HMG-Co. A reductase inhibitors Decreases synthesis of cholesterol in the liver Variable Do not use in pregnancy Possible teratogen In a surveillance study of lovastatin exposures during pregnancy, normal outcomes were 85% congenital anomalies 4. 0% spontaneous abortions 8. 0% and fetal deaths/ stillbirths 1. 0% (9)
Antiemetics • Ondansetron
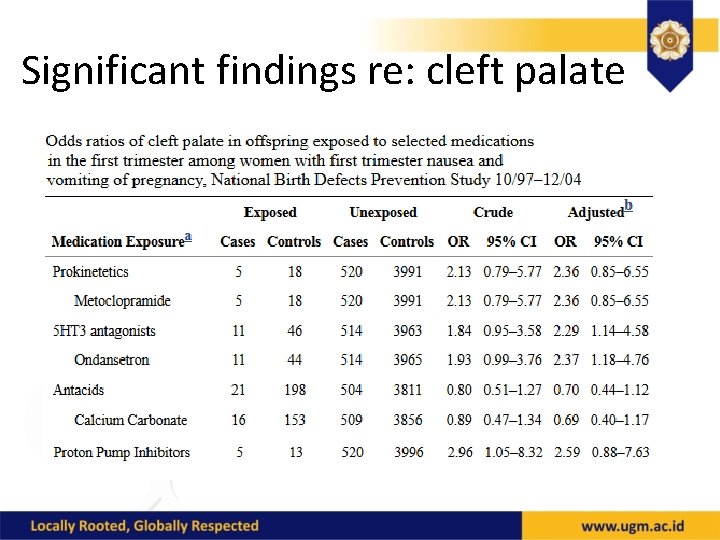
Significant findings re: cleft palate

Conflicting data • Danish registry covering from 1997 -2010 (897, 018 pregnant women). In contrast to earlier studies from same registry, found a 2 -fold increased risk of cardiac malformations with ondansetron (odds ratio 2. 0; 95% confidence interval 1. 3– 3. 1), leading to an overall 30% increased risk of major congenital malformations. To rule out confounding by indication, also examined metoclopramide taken for morning sickness, detecting no increase in teratogenic risk. • Andersen, J. T. , Jimmenez-Solem, E. , and Andersen, N. L. Ondansetron use in early pregnancy and the risk of congenital malformations. Int Soc Pharmacoepidemiol. 2013;
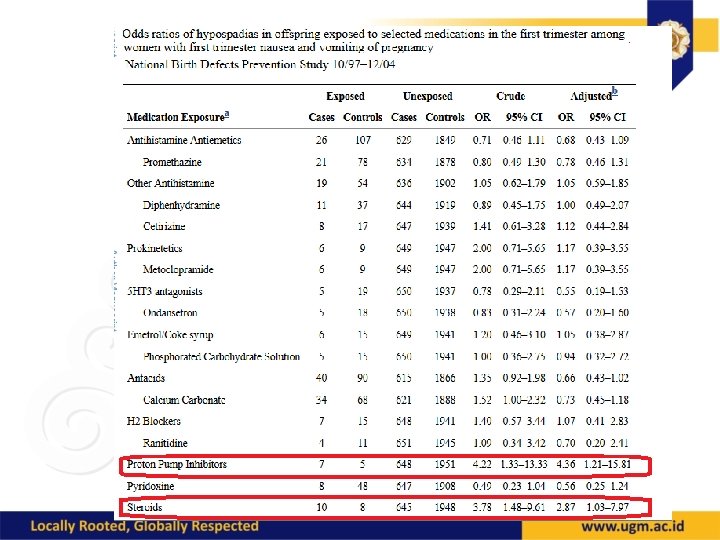

Antibiotics • Penicillins, cephalosporins, EES: OK • Sulfonamides: compete with bili for albumin binding • Tetracycline: OK, except for teeth • Aminoglycosides

Tetracycline • IN PREGNANCY…. . – dental discoloration in children – maternal hepatotoxicity with large Parenteral doses –
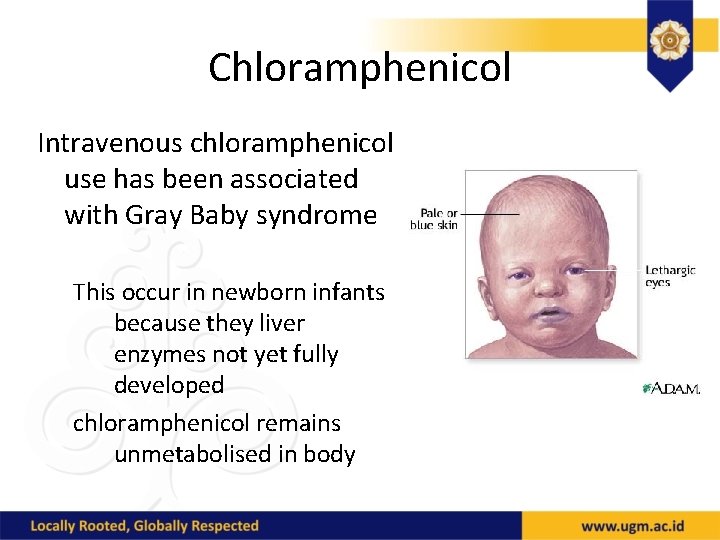
Chloramphenicol Intravenous chloramphenicol use has been associated with Gray Baby syndrome This occur in newborn infants because they liver enzymes not yet fully developed chloramphenicol remains unmetabolised in body
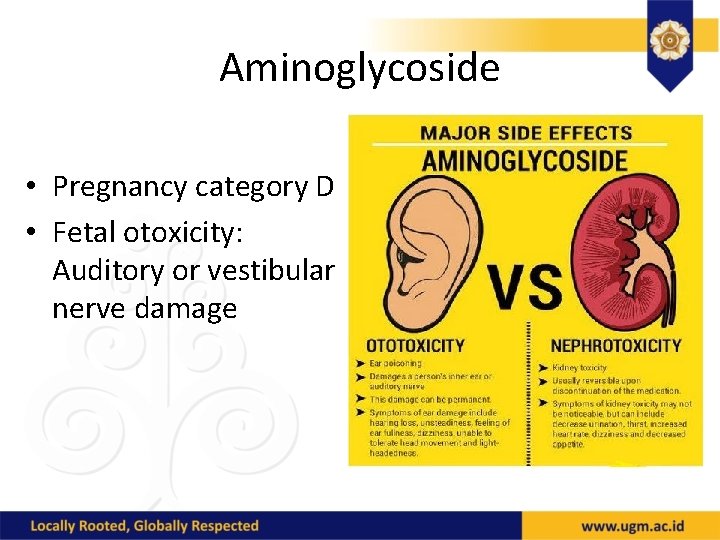
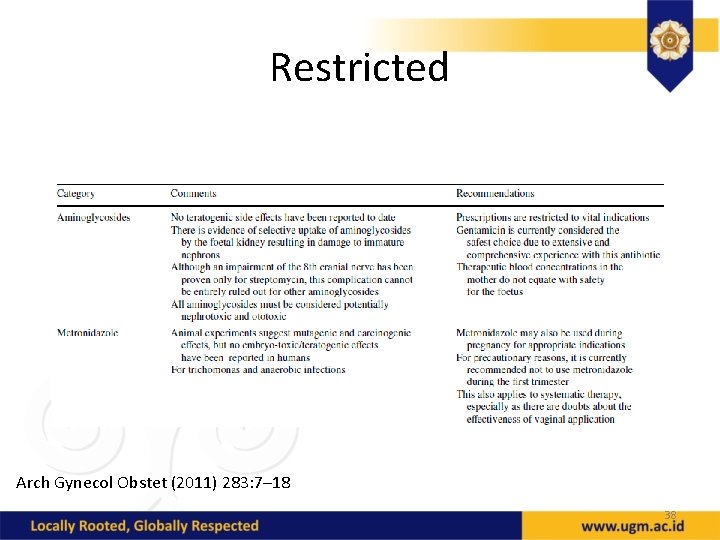
Aminoglycoside • Pregnancy category D • Fetal otoxicity: Auditory or vestibular nerve damage

Trimethoprim- Sulfamethoxazole • NEONATAL HAEMOLYSIS • METHAEMOGLOBINAEMIA
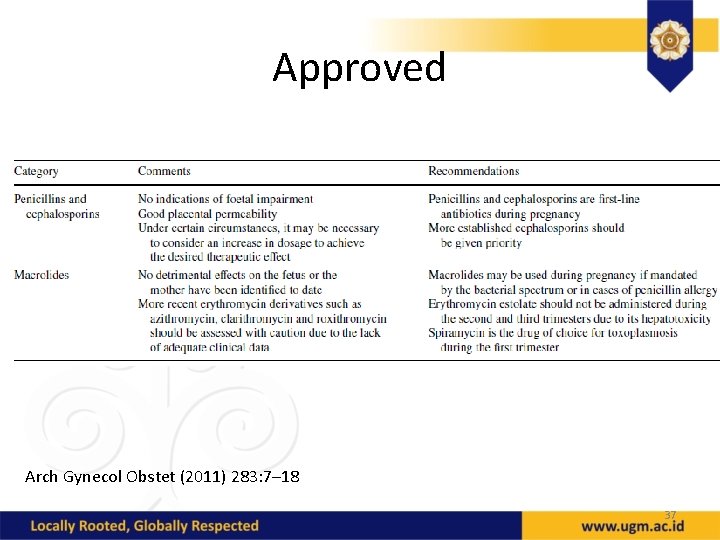
Approved Arch Gynecol Obstet (2011) 283: 7– 18 37
Restricted Arch Gynecol Obstet (2011) 283: 7– 18 38

Restricted Arch Gynecol Obstet (2011) 283: 7– 18

Contraindications Arch Gynecol Obstet (2011) 283: 7– 18 40

Take home messages • Be careful in taking drugs during pregnancy • All physicians including pharmacists are reponsible to counsel patients with complete accurate and current information on the risks and benefits of using medication during pregnancy. 41

All things are poison and nothing is without poison, only the dose permits something not to be poisonous. " ~Paracelcus

THANK YOU
- Slides: 43