Terapia con microinfusore nel paziente con diabete tipo

Terapia con microinfusore nel paziente con diabete tipo 2 Giorgio Grassi Endocrinologia Diabetologia e Metabolismo Citta della Salute e della Scienza Torino 6/102017/

Conflitto di interessi
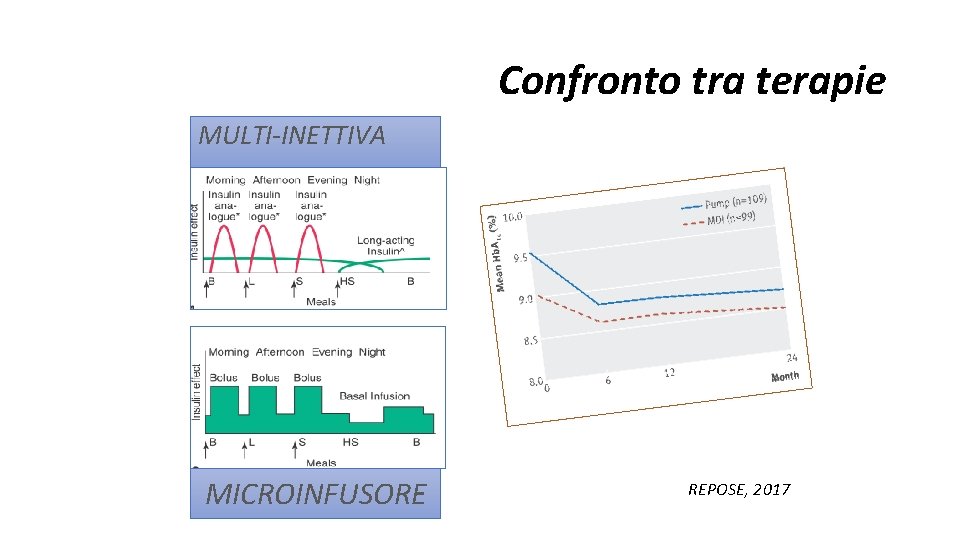
Confronto tra terapie MULTI-INETTIVA MICROINFUSORE REPOSE, 2017
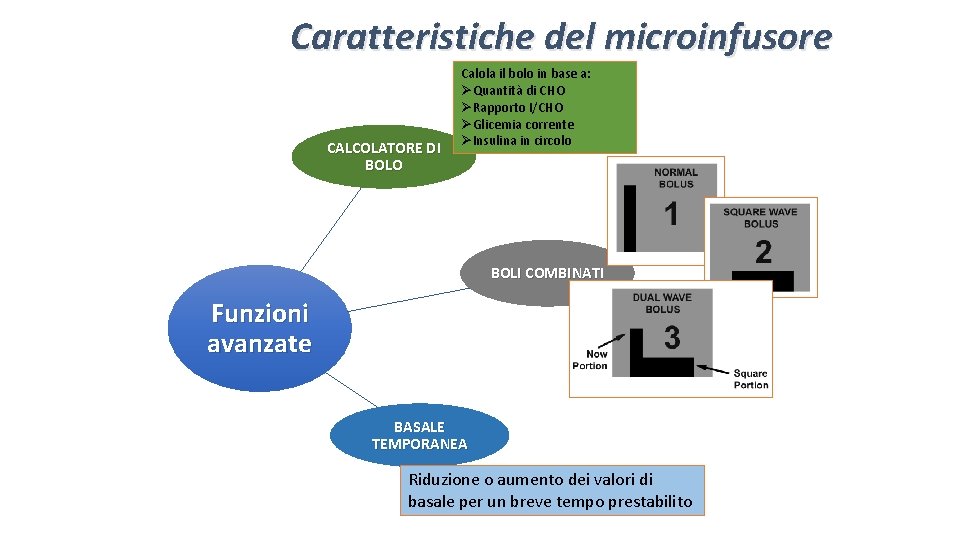
Caratteristiche del microinfusore CALCOLATORE DI BOLO Calola il bolo in base a: ØQuantità di CHO ØRapporto I/CHO ØGlicemia corrente ØInsulina in circolo BOLI COMBINATI Funzioni avanzate BASALE TEMPORANEA Riduzione o aumento dei valori di basale per un breve tempo prestabilito
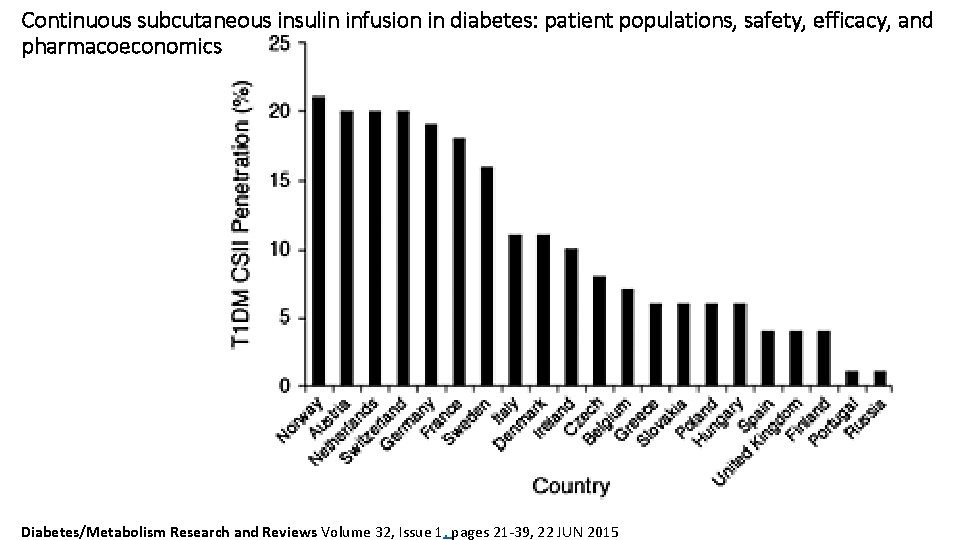
Continuous subcutaneous insulin infusion in diabetes: patient populations, safety, efficacy, and pharmacoeconomics Diabetes/Metabolism Research and Reviews Volume 32, Issue 1, pages 21 -39, 22 JUN 2015
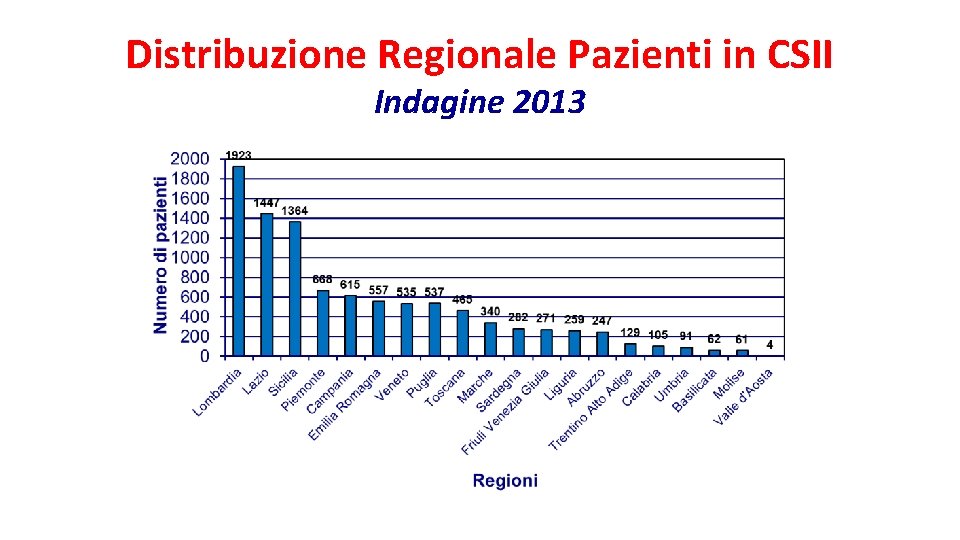
Distribuzione Regionale Pazienti in CSII Indagine 2013
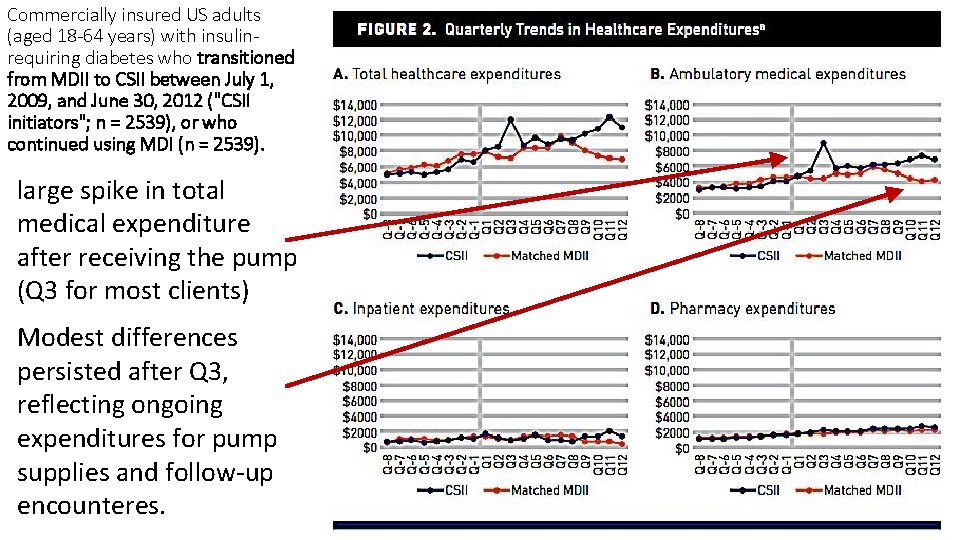
Commercially insured US adults (aged 18 -64 years) with insulinrequiring diabetes who transitioned from MDII to CSII between July 1, 2009, and June 30, 2012 ("CSII initiators"; n = 2539), or who continued using MDI (n = 2539). large spike in total medical expenditure after receiving the pump (Q 3 for most clients) Modest differences persisted after Q 3, reflecting ongoing expenditures for pump supplies and follow-up encounteres.
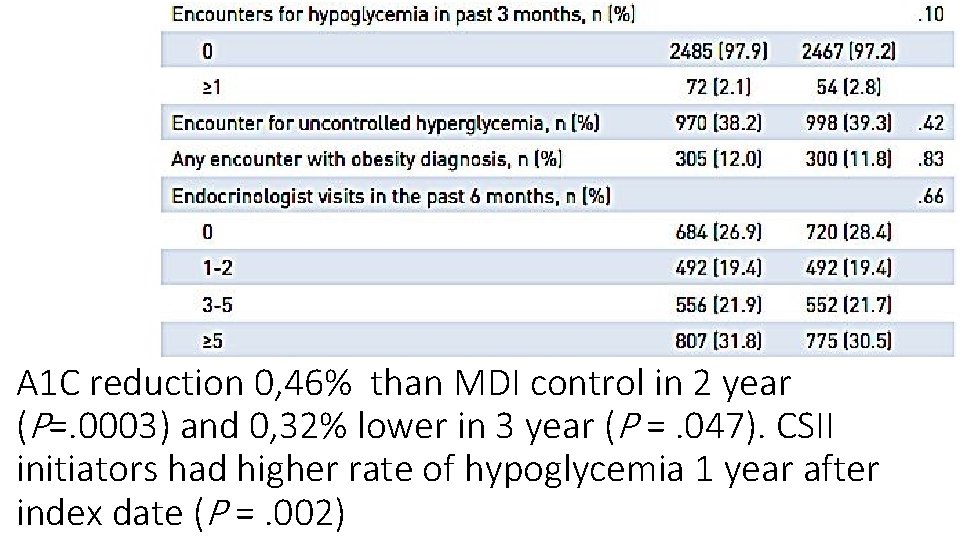
A 1 C reduction 0, 46% than MDI control in 2 year (P=. 0003) and 0, 32% lower in 3 year (P =. 047). CSII initiators had higher rate of hypoglycemia 1 year after index date (P =. 002)

Comparative effectiveness and costs of insulin pump therapy for diabetes. • OBJECTIVES: Continuous subcutaneous insulin infusion (CSII), or "insulin pump" therapy, is an alternative to multiple daily insulin injections (MDII) for management of diabetes. This study evaluates patterns of healthcare utilization, costs, and blood glucose control for patients with diabetes who initiate CSII. • STUDY DESIGN: Pre-post with propensity-matched comparison design involving commercially insured US adults (aged 18 -64 years) with insulin-requiring diabetes who transitioned from MDII to CSII between July 1, 2009, and June 30, 2012 ("CSII initiators"; n = 2539), or who continued using MDI (n = 2539). • RESULTS: Over 3 years, mean per-person total healthcare expenditures were $1714 (95% confidence interval [CI], $1184 -$2244) higher per quarter for CSII initiators compared with matched MDII patients (total mean 3 year difference of $20, 565). Compared with matched controls, mean A 1 C concentrations became lower for CSII initiators by 0. 46% in year 2 (P =. 0003) and by 0. 32% in year 3 (P =. 047). CSII initiators also had a higher rate of hypoglycemia encounters in year 1 (P =. 002). • CONCLUSIONS: For adults with insulin-requiring diabetes, transitioning from MDII to CSII was associated with modest improvements in A 1 C but more hypoglycemia encounters and increased healthcare expenditures, without significant improvement in other potentially offsetting areas of healthcare consumption. Am J Manag Care. 2017 Jun; 23(6): 353 -359.
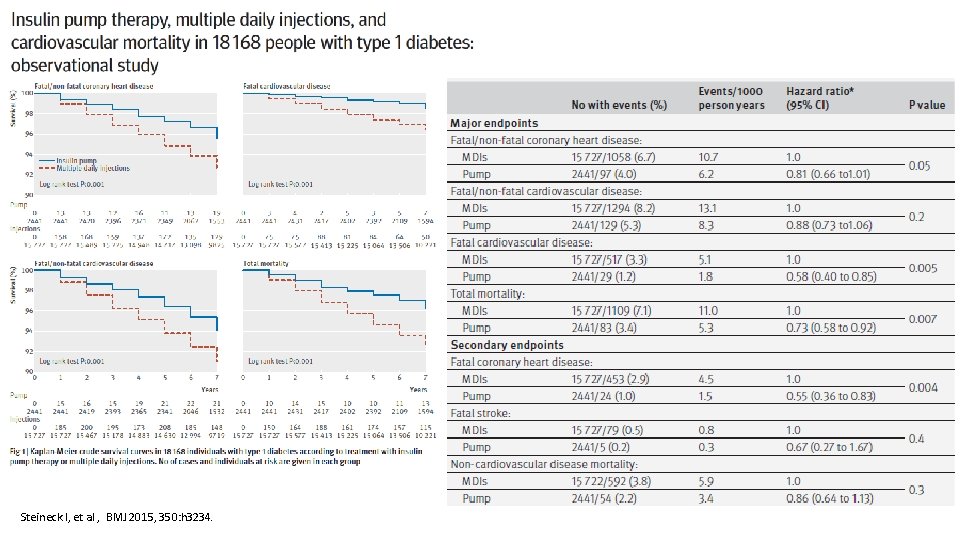
Steineck I, et al , BMJ 2015, 350: h 3234.

ECONOMIC OUTCOMES • Due to the capital cost of an average insulin pump with, say, a 4 -year lifetime, and the need to purchase regular pump supplies such as cannulae, reservoirs and batteries, CSII is on average about 40% more expensive than MDI in many healthcare settings. • However, pump therapy may be considered cost-effective for healthcare systems if cost savings are made in the long term, for example because CSII-induced lower Hb. A 1 c levels reduce the risk of diabetic complications developing. Technology appraisal guidance Published: 23 July 2008

CSII and DMT 2
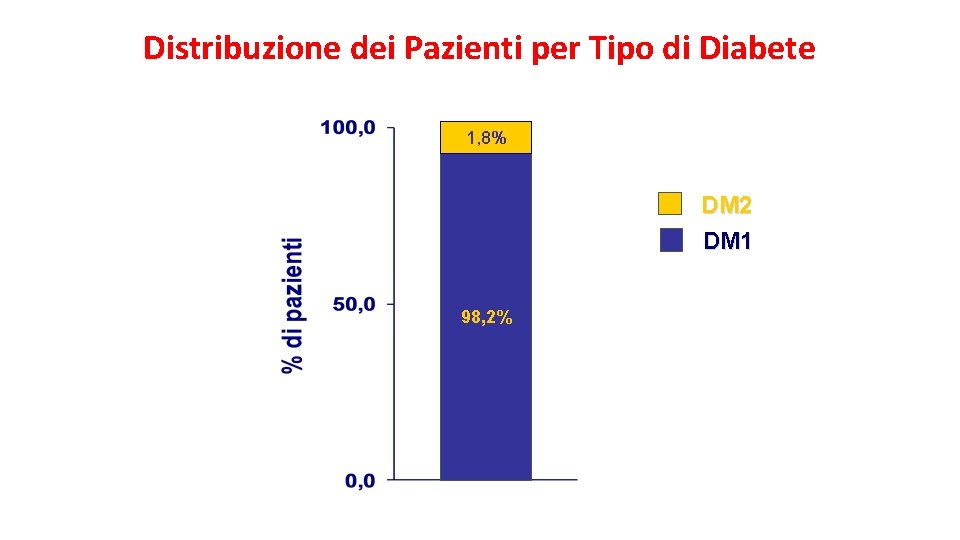
Distribuzione dei Pazienti per Tipo di Diabete 1, 8% DM 2 DM 1 98, 2%
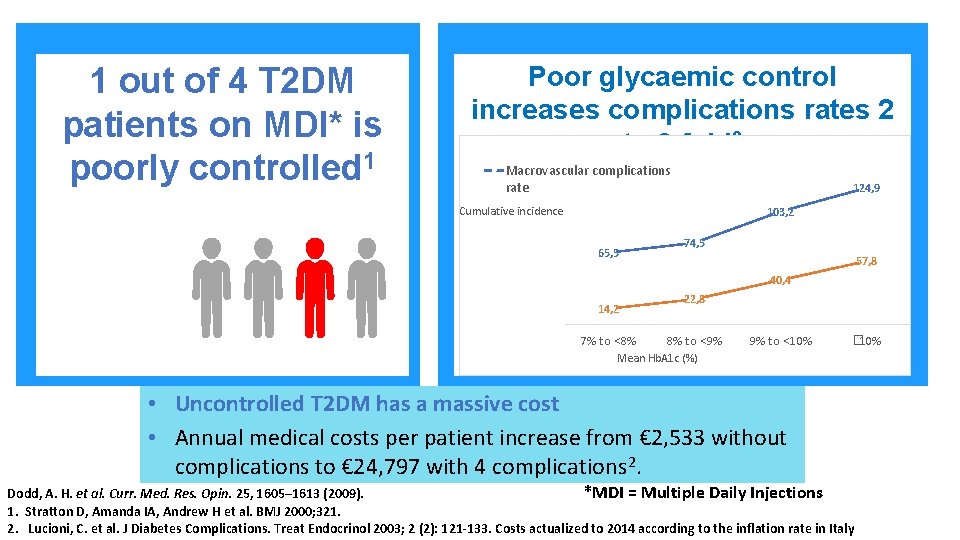
1 out of 4 T 2 DM patients on MDI* is poorly controlled 1 Poor glycaemic control increases complications rates 2 to 3 fold 2 Macrovascular complications rate 124, 9 Cumulative incidence 103, 2 65, 5 74, 5 57, 8 40, 4 14, 2 7% to <8% 22, 8 8% to <9% 9% to <10% � 10% Mean Hb. A 1 c (%) • Uncontrolled T 2 DM has a massive cost • Annual medical costs per patient increase from € 2, 533 without complications to € 24, 797 with 4 complications 2. Dodd, A. H. et al. Curr. Med. Res. Opin. 25, 1605– 1613 (2009). *MDI = Multiple Daily Injections 1. Stratton D, Amanda IA, Andrew H et al. BMJ 2000; 321. 2. Lucioni, C. et al. J Diabetes Complications. Treat Endocrinol 2003; 2 (2): 121 -133. Costs actualized to 2014 according to the inflation rate in Italy

CHALLENGES OF MDI FOR TYPE 2 DIABETES • 51% of type 2 patients on MDI are in poor control (Hb. A 1 c>8%)1 • Increased risk of hypoglycemia and weight gain as insulin regimens intensify 2 • Increased risk of diabetes related complications associated with hyperglycemia 3 • 57% of patients admit to noncompliance by omitting insulin injections 4 1 Annali AMD 2011. http: //www. aemmedi. it/pages/annali_amd/ 2 Reznik Y, Cohen O, Aronson R, et al. Insulin pump treatment compared with multiple daily injections for treatment of type 2 diabetes. Lancet 2014; 384: 1265 -72. 3 Stratton I, et al. Asssociation of glycaemia with macrovascular and microvascular complications of type 2 diabetes (UKPDS 35). BMJ 2000; 321; 405 -412. 4 Peyrot M, Rubin RR, Kruger DF, Travis LB. Correlates of insulin injection omission. Diabetes Care. 2010 Feb; 33(2): 240 -5 The limitations of MDI highlight the need for new treatment strategies in this subgroup of patients ?

Exp Clin Endocrinol Diabetes. 2009 May; 117(5): 220 -2 Continuous subcutaneous insulin infusion versus multiple daily insulin injections in type 2 diabetes: a meta-analysis. Monami M 1, Lamanna C, Marchionni N, Mannucci E. • RESULTS: A total of 4 RCTs was included in the analysis. CSII did not produce any significant improvement of Hb. A 1 c in comparison with MDI (Standardized difference in mean: 0. 09(-0. 08; 0. 26)%; p=0. 31). No significant difference was observed in the rate of hypoglycemic episodes. CSII was associated with a nonsignificant trend toward the reduction of insulin doses used at the end of trial. • CONCLUSIONS: Available data do not justify the use of CSII for basalbolus insulin therapy in type 2 diabetes

CSII THERAPY IN NEWLY DIAGNOSED T 2 D PATIENTS • The clinical study on short term CSII therapy at onset of the disease, published in 2008 by Weng et al. , demonstrated that early use of CSII can sustain prolonged glycemic clinical remission, protecting residual β-cell function

CSII in type 2: Latest articles published Factors associated with improved glycemic control following subcuttaneous insulin infusion therapy in patients with type 2 diabetes uncontrolled with bolus-basal insulin regimen: an analysis from the Op. T 2 ise randomized trial. Muriel Metzger et al, Diabetes Metabolism and Obesity, in press. Glycemic control during continuous subcutaneous insulin infusion versus multiple injections in type 2 diabetes: individual patient data meta analysis and metaregression of randomized controlled trials. John C. Pickup, Diabetes Care, Vol 43, May 2017 Factors affecting the benefit of insulin dose intensification in people with Type 2 diabetes: an analysis from the Op. T 2 mise randomized trial. Schütz-Fuhrmann, J. et al. , Diabet Med. 2017 Feb; 34(2)
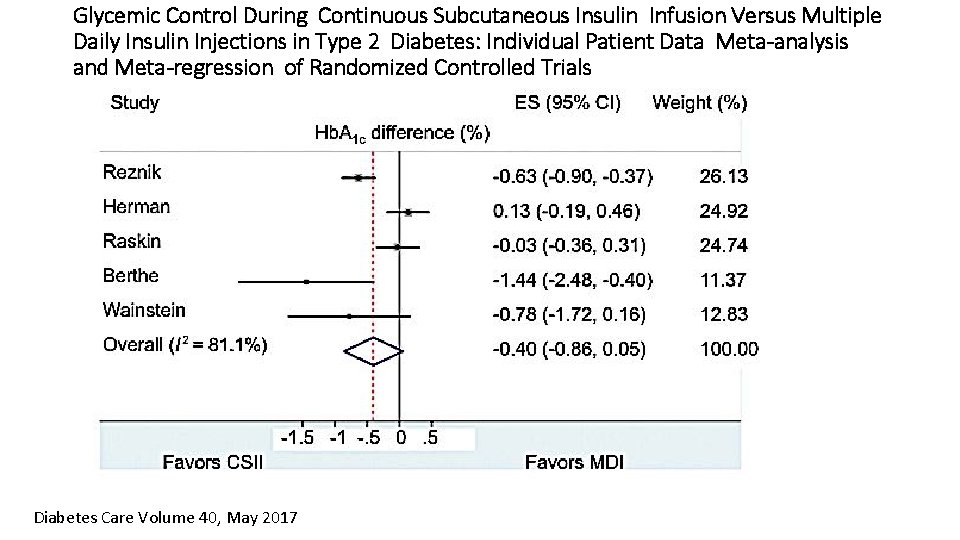
Glycemic Control During Continuous Subcutaneous Insulin Infusion Versus Multiple Daily Insulin Injections in Type 2 Diabetes: Individual Patient Data Meta-analysis and Meta-regression of Randomized Controlled Trials Diabetes Care Volume 40, May 2017
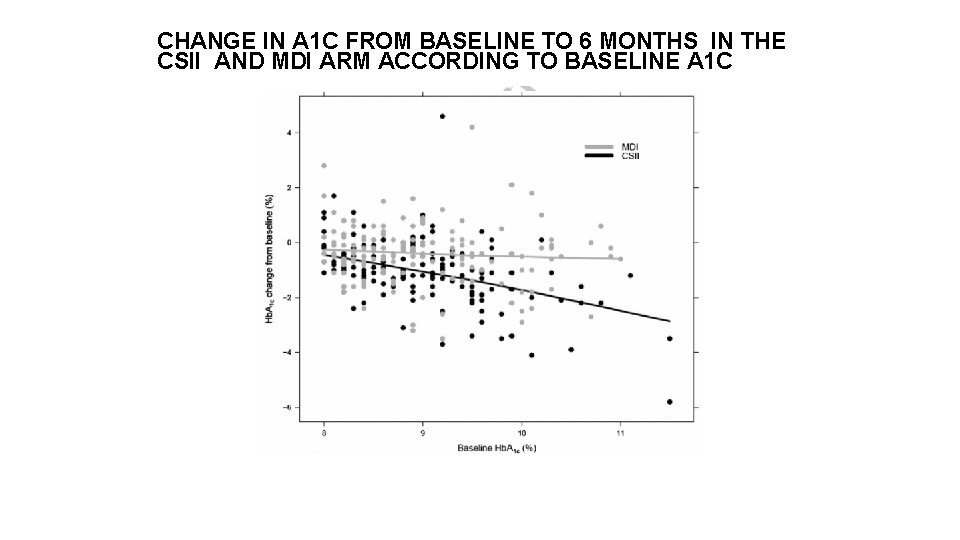
CHANGE IN A 1 C FROM BASELINE TO 6 MONTHS IN THE CSII AND MDI ARM ACCORDING TO BASELINE A 1 C Presentation Title (Edit on Slide Master) | June 1, 2015 | Confidential, for Internal Use Only 20

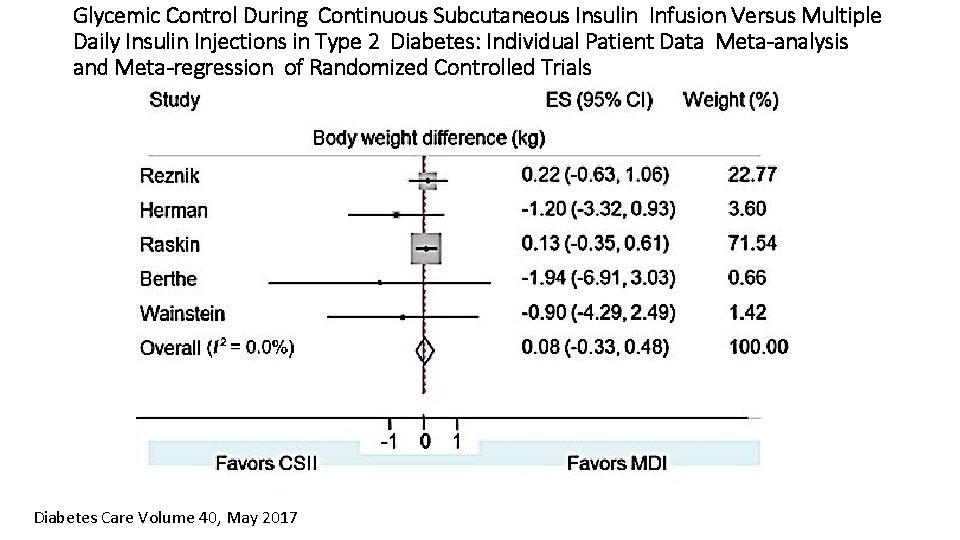
Glycemic Control During Continuous Subcutaneous Insulin Infusion Versus Multiple Daily Insulin Injections in Type 2 Diabetes: Individual Patient Data Meta-analysis and Meta-regression of Randomized Controlled Trials Diabetes Care Volume 40, May 2017
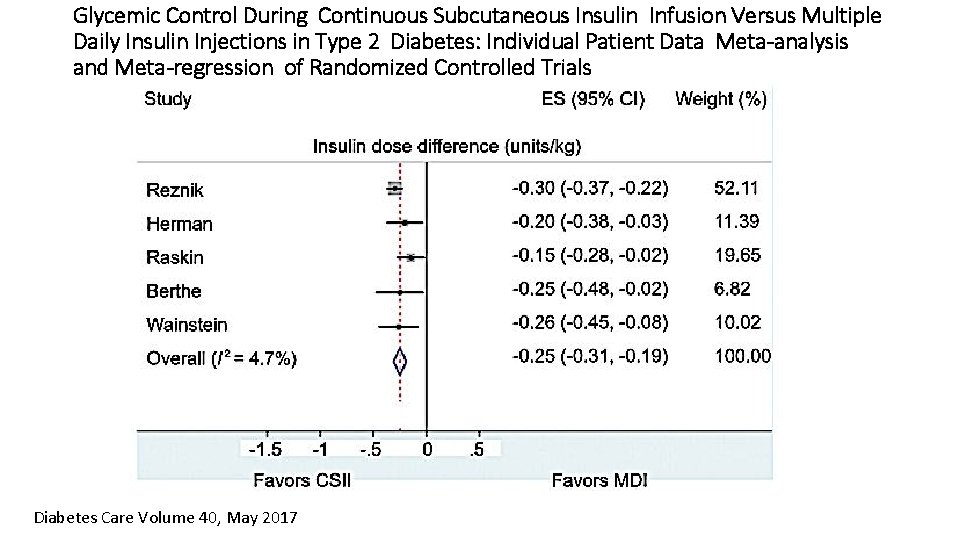
Glycemic Control During Continuous Subcutaneous Insulin Infusion Versus Multiple Daily Insulin Injections in Type 2 Diabetes: Individual Patient Data Meta-analysis and Meta-regression of Randomized Controlled Trials Diabetes Care Volume 40, May 2017
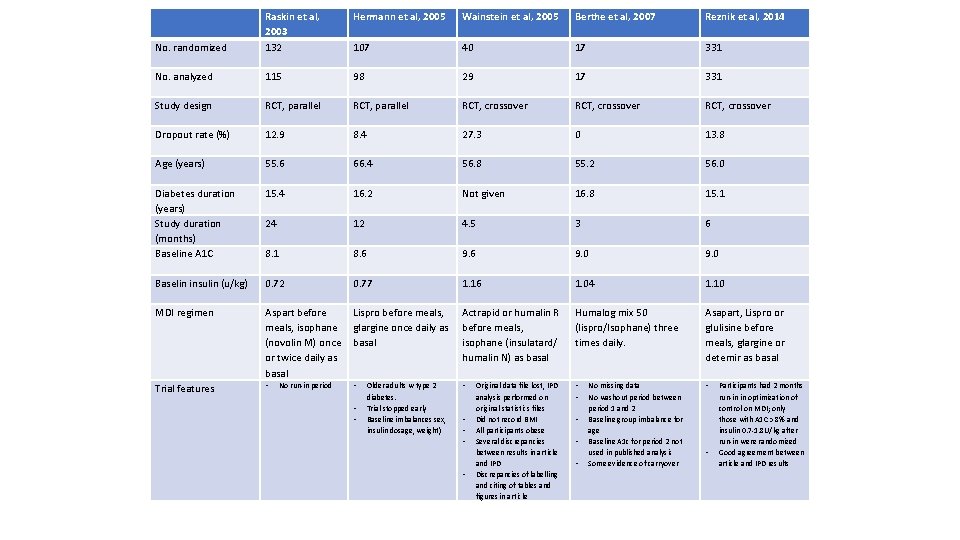
Hermann et al, 2005 Wainstein et al, 2005 Berthe et al, 2007 Reznik et al, 2014 No. randomized Raskin et al, 2003 132 107 40 17 331 No. analyzed 115 98 29 17 331 Study design RCT, parallel RCT, crossover Dropout rate (%) 12. 9 8. 4 27. 3 0 13. 8 Age (years) 55. 6 66. 4 56. 8 55. 2 56. 0 Diabetes duration (years) Study duration (months) Baseline A 1 C 15. 4 16. 2 Not given 16. 8 15. 1 24 12 4. 5 3 6 8. 1 8. 6 9. 0 Baselin insulin (u/kg) 0. 72 0. 77 1. 16 1. 04 1. 10 MDI regimen Aspart before meals, isophane (novolin M) once or twice daily as basal Lispro before meals, glargine once daily as basal Actrapid or humalin R before meals, isophane (insulatard/ humalin N) as basal Humalog mix 50 (lispro/Isophane) three times daily. Asapart, Lispro or glulisine before meals, glargine or detemir as basal Trial features • • • No run-in period • • Older adults w type 2 diabetes. Trial stopped early Baseline imbalances sex, insulin dosage, weight) • • Presentation Title (Edit on Slide Master) | June 1, 2015 | Confidential, for Internal Use Only Original data file lost, IPD analysis performed on original statistics files Did not record BMI All participants obese Several discrepancies between results in article and IPD Discrepancies of labelling and citing of tables and figures in article 24 • • • No missing data No washout period between period 1 and 2 Baseline group imbalance for age Baseline A 1 c for period 2 not used in published analysis Some evidence of carryover • Participants had 2 months run-in in optimization of control on MDI; only those with A 1 C > 8% and insulin 0. 7 -1. 8 U/kg after run-in were randomized Good agreement between article and IPD results

Conclusion from the meta analysis • A 1 C reduction is dependent on pre-randomization A 1 C levels with A 1 C: from -0. 15% with baseline of 8. 0% to -0. 59% with baseline of 10% • Overall mean reduction in A 1 C is -0. 4% for a mean A 1 C of 8. 8% at baseline. • Insulin dose reduction goes from -23. 6 u/day for a baseline treatment of 100 u/day up to -35. 5 u/day for a baseline treatment of 150 u/day. • Insulin dose reduction at the end of study period is -25%, irrespective of baseline insulin treatment. • A 1 C reduction is greater in those patients that undergo pre-randomization insulin dose optimization. CSII in type 2 diabetes may be targeted at those who have failed to achive target A 1 C levels after best attempts with MDI. Presentation Title (Edit on Slide Master) | June 1, 2015 | Confidential, for Internal Use Only 25

Op. T 2 mise INTERNATIONAL STUDY 8 Canada, 23 Europe, 2 South Africa, 3 USA 36 CENTERS AROUND THE GLOBE:
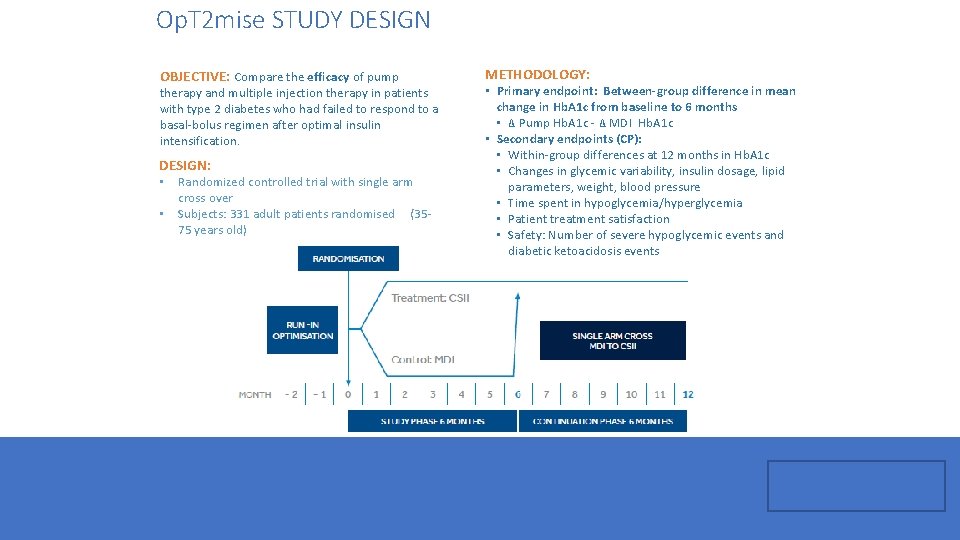
Op. T 2 mise STUDY DESIGN OBJECTIVE: Compare the efficacy of pump therapy and multiple injection therapy in patients with type 2 diabetes who had failed to respond to a basal-bolus regimen after optimal insulin intensification. DESIGN: • • Randomized controlled trial with single arm cross over Subjects: 331 adult patients randomised (3575 years old) METHODOLOGY: • Primary endpoint: Between-group difference in mean change in Hb. A 1 c from baseline to 6 months • Δ Pump Hb. A 1 c - Δ MDI Hb. A 1 c • Secondary endpoints (CP): • Within-group differences at 12 months in Hb. A 1 c • Changes in glycemic variability, insulin dosage, lipid parameters, weight, blood pressure • Time spent in hypoglycemia/hyperglycemia • Patient treatment satisfaction • Safety: Number of severe hypoglycemic events and diabetic ketoacidosis events
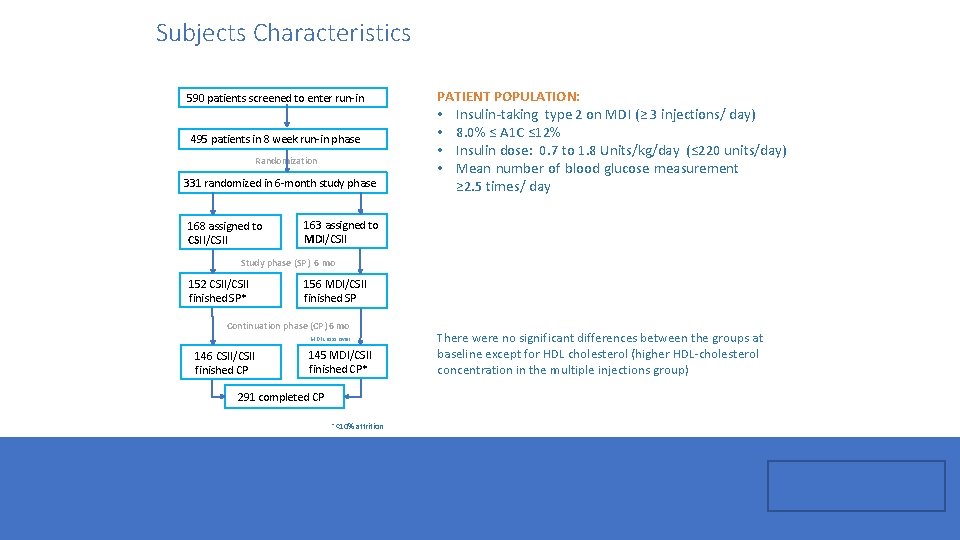
Subjects Characteristics 590 patients screened to enter run-in 495 patients in 8 week run-in phase Randomization 331 randomized in 6 -month study phase 168 assigned to CSII/CSII PATIENT POPULATION: • Insulin-taking type 2 on MDI (≥ 3 injections/ day) • 8. 0% ≤ A 1 C ≤ 12% • Insulin dose: 0. 7 to 1. 8 Units/kg/day (≤ 220 units/day) • Mean number of blood glucose measurement ≥ 2. 5 times/ day 163 assigned to MDI/CSII Study phase (SP) 6 mo 152 CSII/CSII finished SP* 156 MDI/CSII finished SP Continuation phase (CP) 6 mo MDI cross over 146 CSII/CSII finished CP 145 MDI/CSII finished CP* 291 completed CP *<10% attrition There were no significant differences between the groups at baseline except for HDL cholesterol (higher HDL-cholesterol concentration in the multiple injections group)
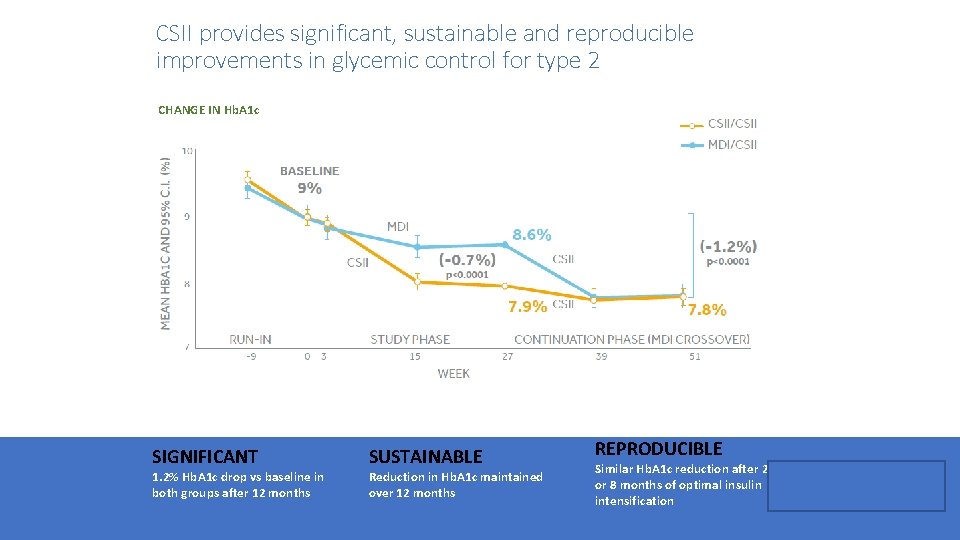
CSII provides significant, sustainable and reproducible improvements in glycemic control for type 2 CHANGE IN Hb. A 1 c SIGNIFICANT 1. 2% Hb. A 1 c drop vs baseline in both groups after 12 months SUSTAINABLE Reduction in Hb. A 1 c maintained over 12 months REPRODUCIBLE Similar Hb. A 1 c reduction after 2 or 8 months of optimal insulin intensification
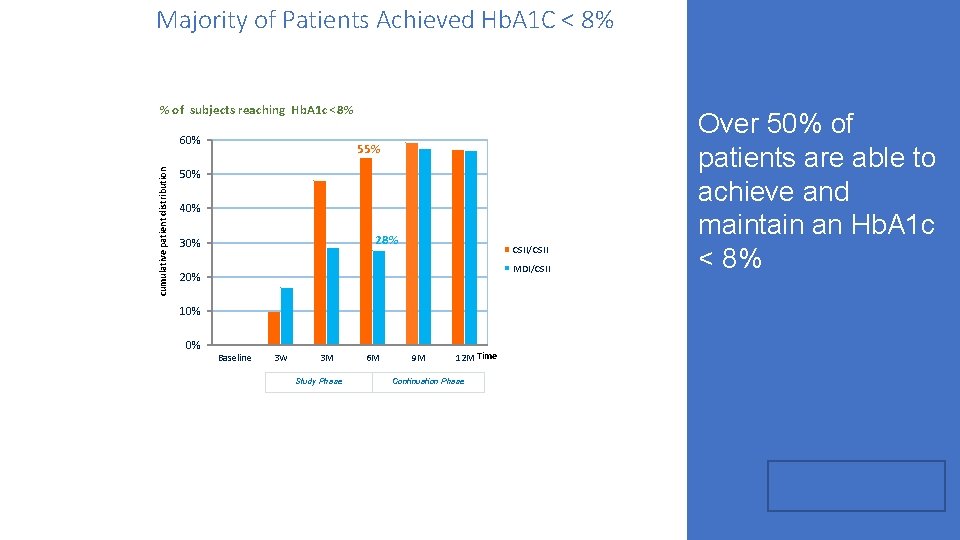
Majority of Patients Achieved Hb. A 1 C < 8% % of subjects reaching Hb. A 1 c <8% cumulative patient distribution 60% 55% 50% 40% 28% 30% CSII/CSII MDI/CSII 20% 10% 0% Baseline 3 W 3 M Study Phase 6 M 9 M 12 M Time Continuation Phase Over 50% of patients are able to achieve and maintain an Hb. A 1 c < 8%
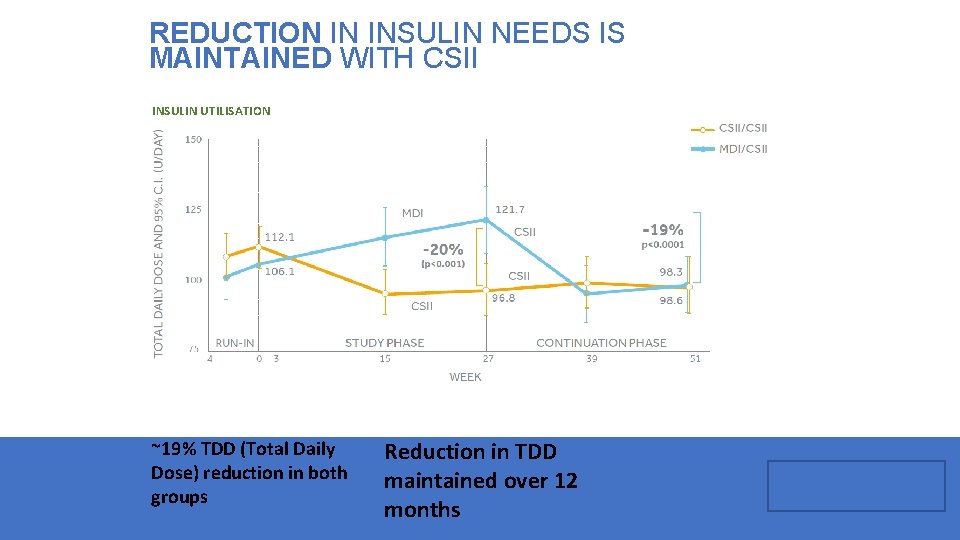
REDUCTION IN INSULIN NEEDS IS MAINTAINED WITH CSII INSULIN UTILISATION ~19% TDD (Total Daily Dose) reduction in both groups Reduction in TDD maintained over 12 months
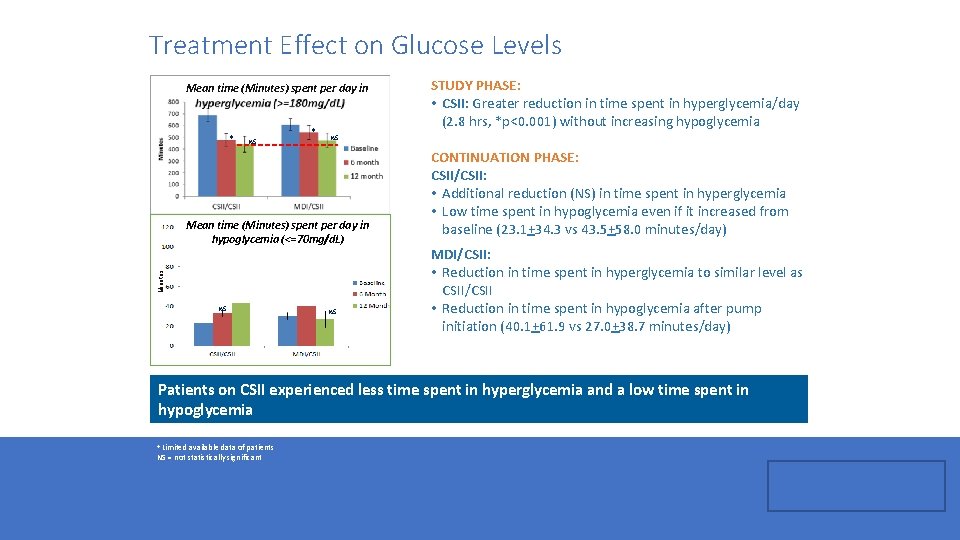
Treatment Effect on Glucose Levels Mean time (Minutes) spent per day in * NS Minutes Mean time (Minutes) spent per day in hypoglycemia (<=70 mg/d. L) NS STUDY PHASE: • CSII: Greater reduction in time spent in hyperglycemia/day (2. 8 hrs, *p<0. 001) without increasing hypoglycemia NS CONTINUATION PHASE: CSII/CSII: • Additional reduction (NS) in time spent in hyperglycemia • Low time spent in hypoglycemia even if it increased from baseline (23. 1+34. 3 vs 43. 5+58. 0 minutes/day) MDI/CSII: • Reduction in time spent in hyperglycemia to similar level as CSII/CSII • Reduction in time spent in hypoglycemia after pump initiation (40. 1+61. 9 vs 27. 0+38. 7 minutes/day) Patients on CSII experienced less time spent in hyperglycemia and a low time spent in hypoglycemia Limited available data of patients NS = not statistically significant +
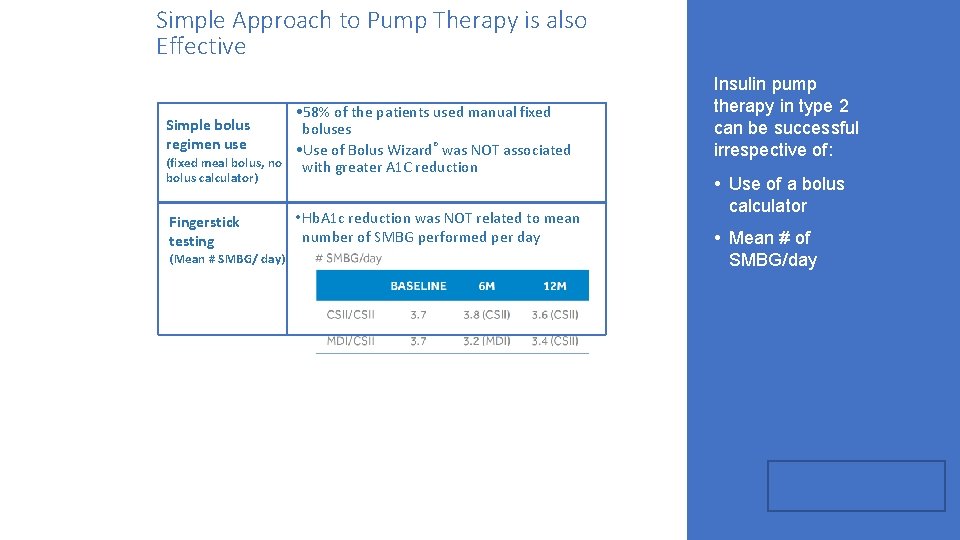
Simple Approach to Pump Therapy is also Effective • 58% of the patients used manual fixed boluses • Use of Bolus Wizard® was NOT associated (fixed meal bolus, no with greater A 1 C reduction Simple bolus regimen use bolus calculator) Fingerstick testing (Mean # SMBG/ day) • Hb. A 1 c reduction was NOT related to mean number of SMBG performed per day Insulin pump therapy in type 2 can be successful irrespective of: • Use of a bolus calculator • Mean # of SMBG/day

Additional Benefits of CSII Observed LIPID PARAMETERS AND WEIGHT • No significant differences were seen in any lipid parameters • No significant differences were seen in body weight change SAFETY AND OTHER RESULTS • No ketoacidosis events • Severe hypoglycemia One episode occurred in CSII/CSII group Two episodes occurred in MDI/CSII group (with same patient) • 7 diabetes, device or study procedure related SAEs occurred in the CSII/CSII group, and 11 in the MDI/CSII group

Op. T 2 mise 12 Month Results Summary Mini. Med pump therapy treatment in sub-optimally controlled T 2 MDI provides significant improvements in overall glucose control: • Significant decreases in A 1 C levels (-1. 2% vs baseline in both groups) over 1 year • More than half of all patients achieved A 1 C < 8% • Significant and sustained reduction of insulin dose when switched to CSII • Increased and sustained patient satisfaction when switched to CSII • No significant difference in body weight gain • Simple approach to pump therapy is also effective
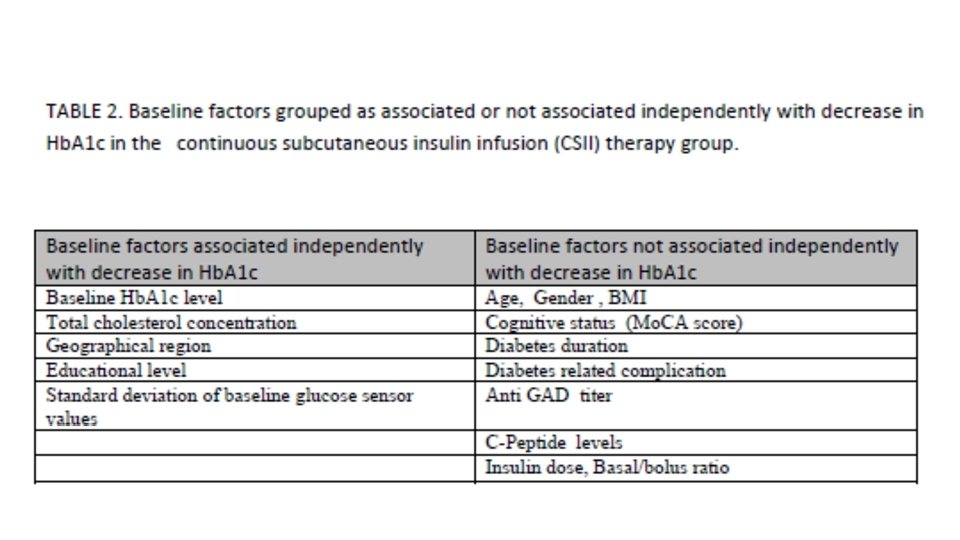
Presentation Title (Edit on Slide Master) | June 1, 2015 | Confidential, for Internal Use Only 36
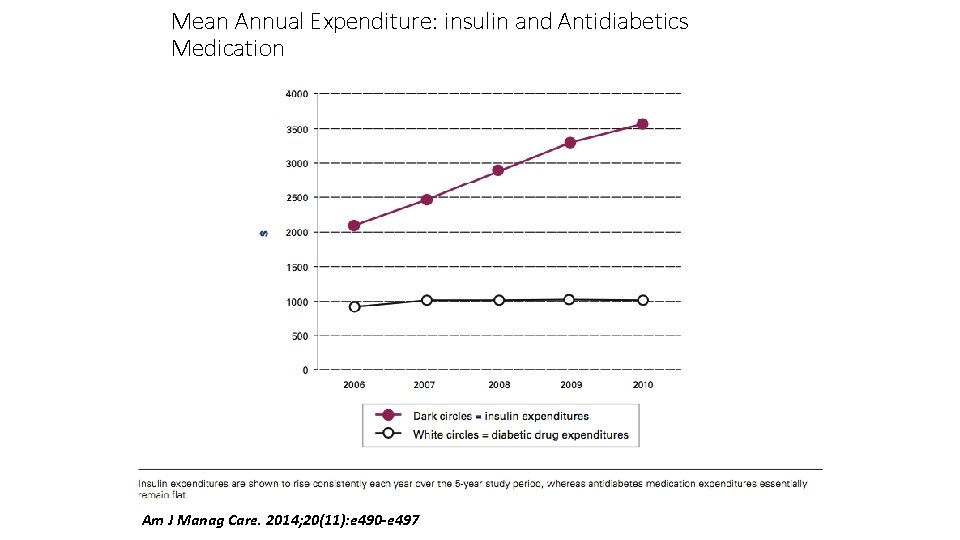
Mean Annual Expenditure: insulin and Antidiabetics Medication Am J Manag Care. 2014; 20(11): e 490 -e 497
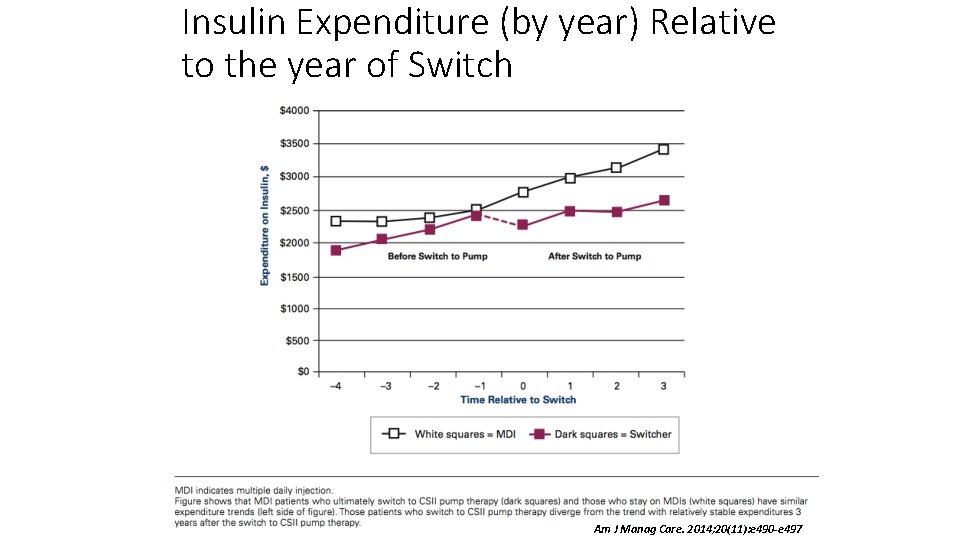
Insulin Expenditure (by year) Relative to the year of Switch Am J Manag Care. 2014; 20(11): e 490 -e 497

Switching from Multiple Daily Injections to CSII Pump Therapy: Insulin Expenditures in Type 2 Diabetes • Take-Away Points • The primary objective of this study was to identify the impact of switching from multiple daily injections (MDIs) to continuous subcutaneous insulin infusion (CSII) pump therapy on insulin and other antidiabetic medication expenditures in individuals with type 2 diabetes mellitus. • This study demonstrated a significant reduction in insulin expenditures among MDI patients who switched to CSII pump therapy throughout the study period. While insulin expenditures rose during the study period, switching to CSII pump therapy led to sizable reductions in insulin expenditures. • This reduction in insulin expenditures due to switching varied between $657 (SE $126; P <. 01) and $1011 (SE $250. 60; P <. 01) per year. Am J Manag Care. 2014; 20(11): e 490 -e 497

Cost-effectiveness CSII in DMT 2: Cost-effectiveness of continuous subcutaneous insulin infusion in people with type 2 diabetes in the Netherlands • In the Netherlands, CSII represents a cost-effective option in patients with type 2 diabetes who continue to have poorly-controlled Hb. A 1 c despite optimization of MDI. Since the ICER falls below the willingness-topay threshold of EUR 80, 000 per QALY gained, CSII is likely to represent good-value for money in the treatment of poorly-controlled T 2 D patients compared with MDI. JOURNAL OF MEDICAL ECONOMICS, 2016 VOL. 19, NO. 8, 742– 749

reduction of complications and associatec costs for type 2 Diabetic Patients Using Continuous subcutaneous insulin infusion in t. He Uk • Methods: The incidence of diabetes-related complications was calculated based on the Core Diabetes Model. The population characteristics, the reduction of Hb. A 1 c, and insulin dose were based on the Opt 2 mise • Results: The diabetes-related complications were reduced with CSII. At 5 years, the incidence reduction in complications associated with eye diseases, renal diseases, ulcer/amputation and cardiovascular diseases were -24%, -26%, -19% and -10%, respectively, in favour of CSII. • conclusions: Improvements in Hb. A 1 c with a decrease in overall insulin requirements observed with CSII versus MDI, may offer important reductions in diabetes-related complications and associated costs in a UK setting for uncontrolled T 2 DM patients. VALUE IN HEALTH 18 (2015) A 335–A 766

Only 6 of the 22 guidelines identified
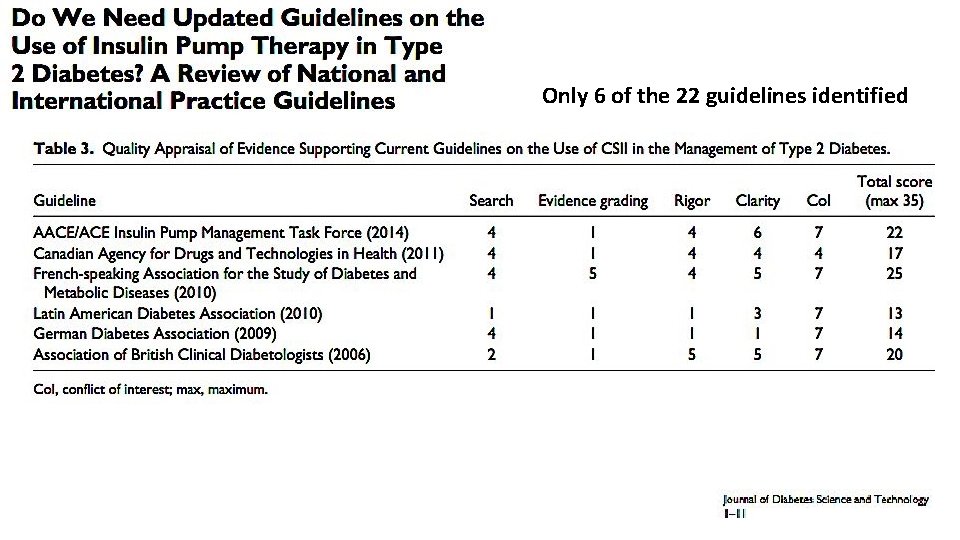
Only 6 of the 22 guidelines identified

lombardia CSII

Maurizi AR, Suraci C, Pitocco D, Schia ni R, Tubili C, Morviducci L, et al. ; C. S. I. I. Study Group of Lazio Region Italy. Position Statement on the management of continuous subcutaneous insulin infusion (CSII): e Italian Lazio experience. J Diabetes 2016; 8: 41 -4
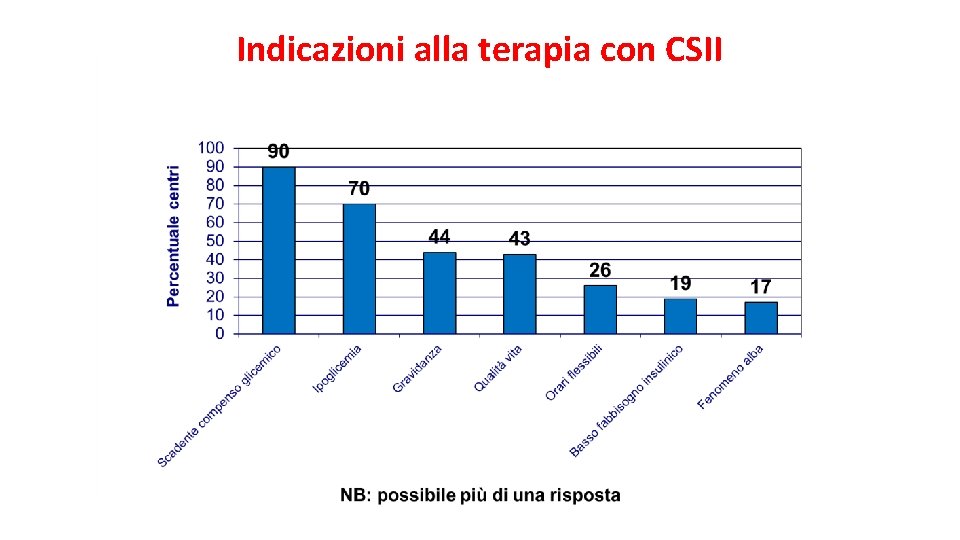
Indicazioni alla terapia con CSII
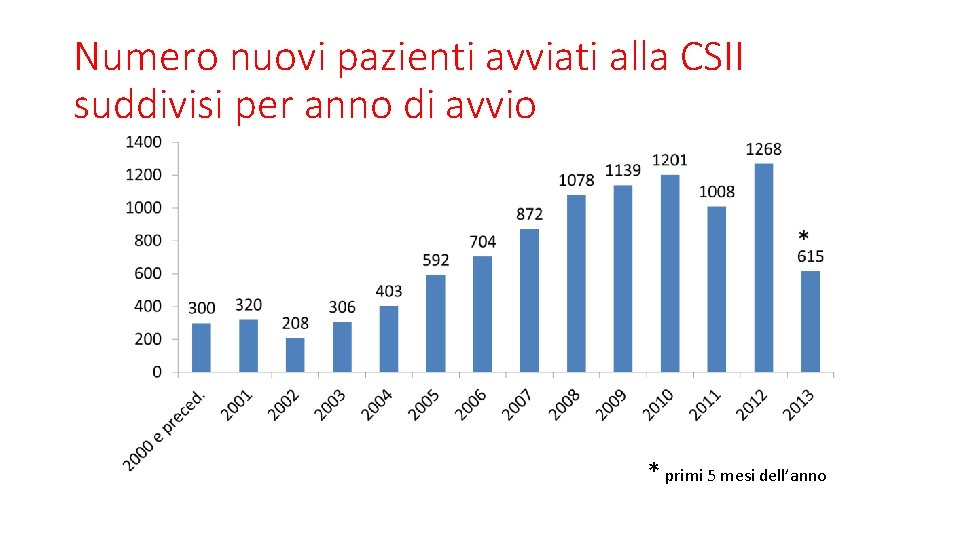
Numero nuovi pazienti avviati alla CSII suddivisi per anno di avvio * primi 5 mesi dell’anno

Pa. Q • Possibile ruolo di patch pump easy ? • Up to 3 days of continuous subcutaneous insulin delivery • Worn on the abdomen • 7 preset basal doses • 2 bolus units delivered with each button push • Delivers up to 330 units over 3 days

COMBINATION THERAPY BASED ON CSII THERAPY IN ADDICTION TO NOVEL ANTI-HYPERGLYCEMIC AGENTS • Recently CSII treatment has been tested in combination with new antihyperglycemic agents. • Ke et al. investigated the effects of liraglutide combined with short term CSII therapy on glycemic control and β-cell function in newly diagnosed T 2 D patients. e combined treatment was effective in further improving β-cell function, but the beneficial effects disappeared after stopping liraglutide

When Intensive Insulin Therapy (MDI) Fails in Patients With Type 2 Diabetes: Switching to GLP-1 Receptor Agonist Versus Insulin Pump Diabetes Care 2016; 39(Suppl. 2): S 180–S 186 • Patients who have not previously failed GLP-1 receptor agonist therapy may show reduc- tion in weight and insulin dose, in addition to moderate improvement in Hb. A 1 c, when GLP-1 receptor agonist therapy is added to MDI regimens. • In subjects with long-standing type 2 diabetes who do not respond to intensive insulin therapies, switching from MDI to CSII and/or the addition of GLP-1 receptor agonists to MDI have the potential to improve glycemic control without increasing the risk of adverse events. Diabetes Care Volume 39, Supplement 2, August 2016

Insulin pump therapy in type 2 diabetes is effective at lowering Hb. A 1 c levels, but, as with type 1 diabetes, it should be targeted at those persons with worst glycemic control and highest insulin dose after best attempts with MDI
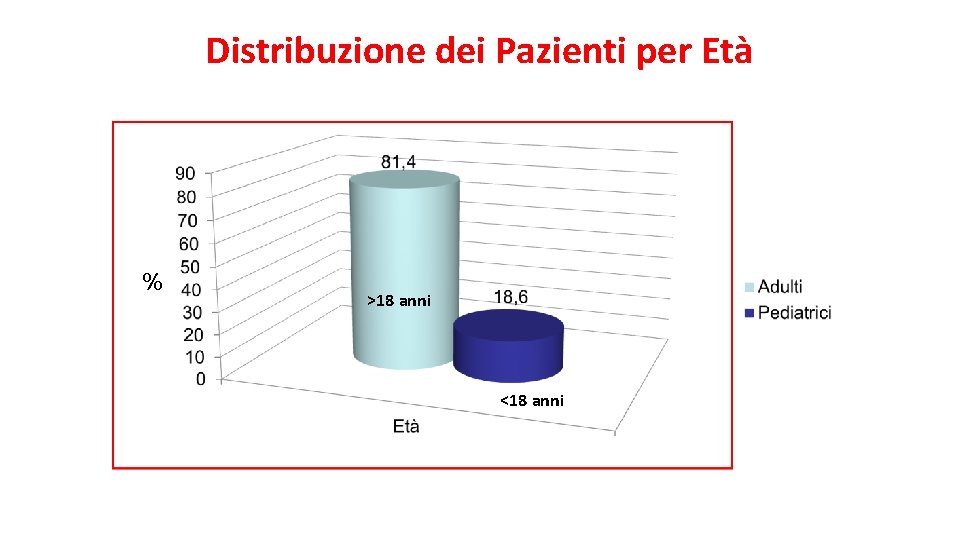
Distribuzione dei Pazienti per Età % >18 anni <18 anni

Grazie !
- Slides: 53