Temperature Solubility curves have temperature on the Xaxis


Temperature • Solubility curves have temperature on the X-axis. • Depends on the system, but we have a couple of general rules: – Solubility of all gases as temperature . – Solubility of most solids as temperature .

Solubility of Gases 1. Depends on temperature. Gases are more soluble in colder solvents. 2. Depends on pressure.

Pressure & Solubility of Gases • Henry’s Law: • At a given temperature, the solubility of a gas in a liquid is directly proportional to the pressure of the gas above the liquid. • Solubility = k·Pgas • Many bottling plants use pressures of 5 to 7 atmospheres to carbonate beverages (i. e. , dissolve CO 2 in them).
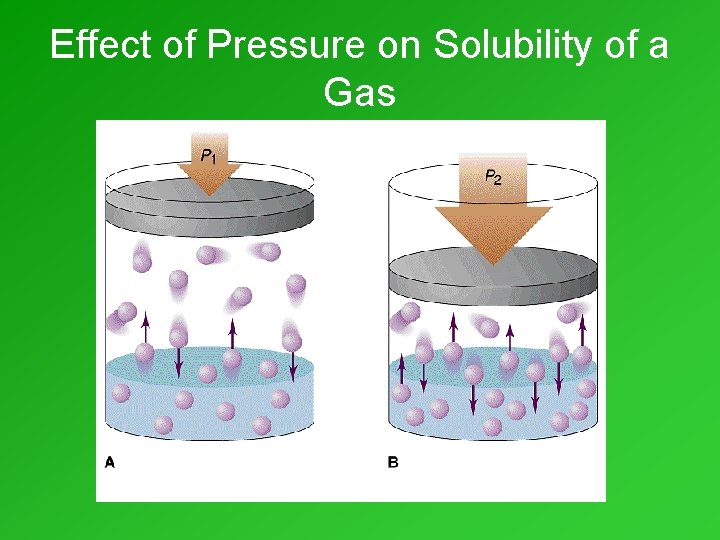
Effect of Pressure on Solubility of a Gas
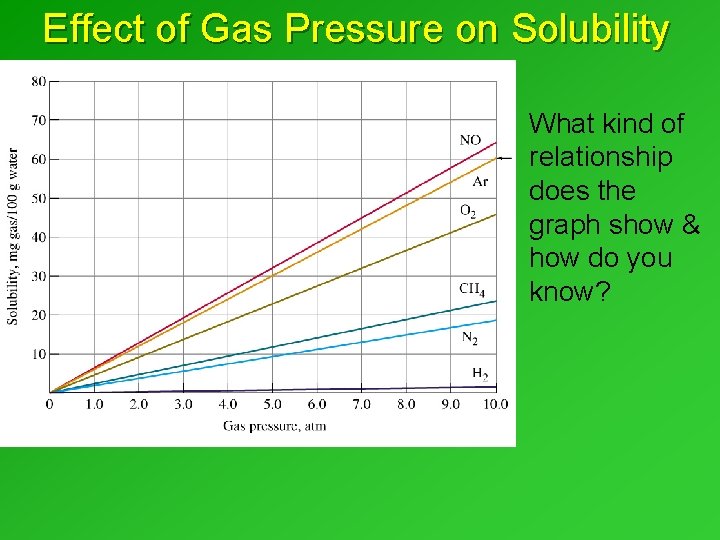
Effect of Gas Pressure on Solubility What kind of relationship does the graph show & how do you know?

Do pressure changes affect other systems? • Solubility of liquids & solids is not influenced by pressure changes.

Which system is most affected by pressure changes? a) Na. Cl(s) Na+(aq) + Cl-(aq) b) C 6 H 12 O 6(s) C 6 H 12 O 6(aq) c) NH 3(g) NH 3(aq) It’s the only one involving a gas!!!

Nature of the solute & the solvent • Another factor that determines solubility. • NATURE: – Ionic – Nonpolar covalent – Polar covalent • Rule is “Like Dissolves Like” CCA, Volume 2

Possibilites are: • Solvent: Nonpolar covalent or polar covalent • Solute: Ionic, nonpolar covalent, polar covalent

Possibilites are: • Nonpolar solute in nonpolar solvent. • Polar solute in polar solvent. • Ionic solute in polar solvent.

Identify the nature of each of the following as PC, NPC, or ionic. • • • CH 4 Nonpolar Cov. • Na. Cl Ionic • H 2 O Polar Covalent • CCl 4 Nonpolar Cov. • CH 3 OH Polar Cov. H 2 S Polar Cov. Ca. Br 2 Ionic C 6 H 14 Nonpolar Cov. NH 3 Polar Cov. NH 4 OH Ionic C 4 H 10 Nonpolar Cov. PH 3 CH 3 CH 2 OH Polar Cov.

IONIC vs. COVALENT • Ionic: Substance is composed of positive & negative ions (usually metal + nonmetal). • Covalent: Substance is composed of neutral molecules (all nonmetal elements).

NONPOLAR COVALENT • • • Have a symmetrical electron cloud. Have no separation of charge. Have weak van der Waals forces. Are easy to pull apart. Have low melting points, low boiling points, low heats of fusion. • Have high vapor pressures.

Easy categories of nonpolar covalent substances • Noble gases • Homonuclear diatomics • Molecules that are extremely symmetric (CO 2, Si. H 4, CCl 4, CF 4) • Anything that’s just C and H

POLAR COVALENT • • • Have a lopsided electron cloud. Have a separation of charge. Have dipole-dipole interactions. Are harder to pull apart. Have higher melting points, boiling points, and heats of fusion than nonpolar molecules. • Have lower vapor pressures than nonpolar molecules.
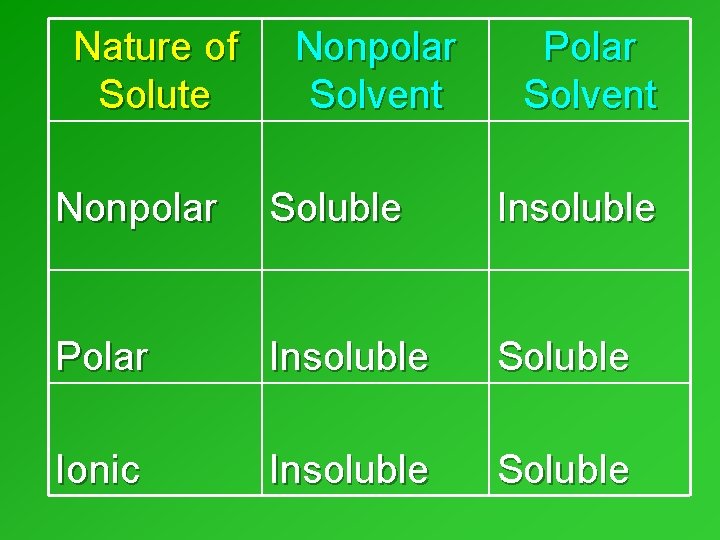
Nature of Solute Nonpolar Solvent Polar Solvent Nonpolar Soluble Insoluble Polar Insoluble Soluble Ionic Insoluble Soluble
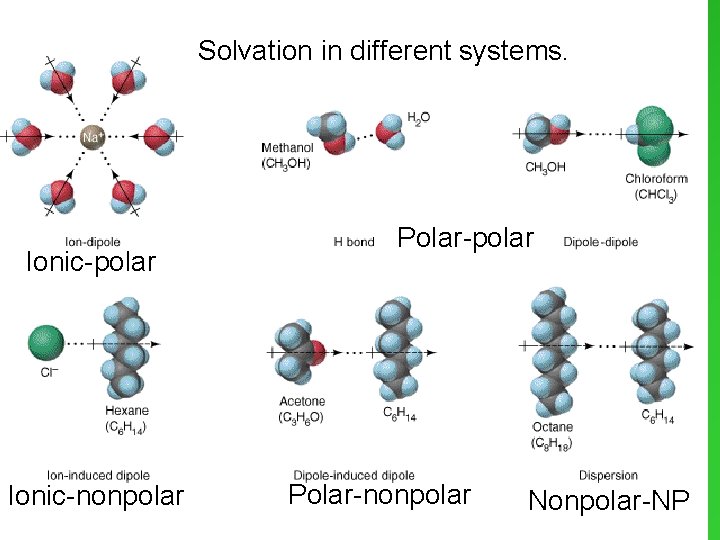
Solvation in different systems. Ionic-polar Ionic-nonpolar Polar-nonpolar Nonpolar-NP

What kind of solvent is H 2 O? • • • Water is polar covalent. The oxygen end: -. The hydrogens: +. Ionic & Polar solutes dissolve well in H 2 O. Nonpolar solutes do not dissolve in H 2 O.

Dissolving Na. Cl in water • Water is a polar molecule. The O has a charge and the H’s have a + charge. Na. Cl is ionic The Na ion has a full +1 charge & the Cl ion has a full -1 charge. • The Na+ is attracted to the - of the O’s. This beats out the attraction to the Cl-. • The Cl- is attracted to the + of the H’s. This beats out the attraction to the Na.

Dissolving Polar substances in water. • Polar substances have a separation of charge. One side of the molecules is and one side +. • The + of the solute interacts with the - or oxygen end of the water molecule and vice versa. Dissolving a sugar cube in water.

Dissolving nonpolar substances in water • Symmetrical molecules are nonpolar. • Many organic molecules (C, H) are nonpolar. • Nonpolar molecules do not dissolve in water because the attractive forces between the polar water molecules and the nonpolar molecules are too weak.

Why soap works • Grease won’t dissolve easily in water. • Grease is nonpolar & water is polar. • Soaps – very long molecules. – One end is polar & dissolves in water. – The other end is nonpolar, & grease will dissolve in it.
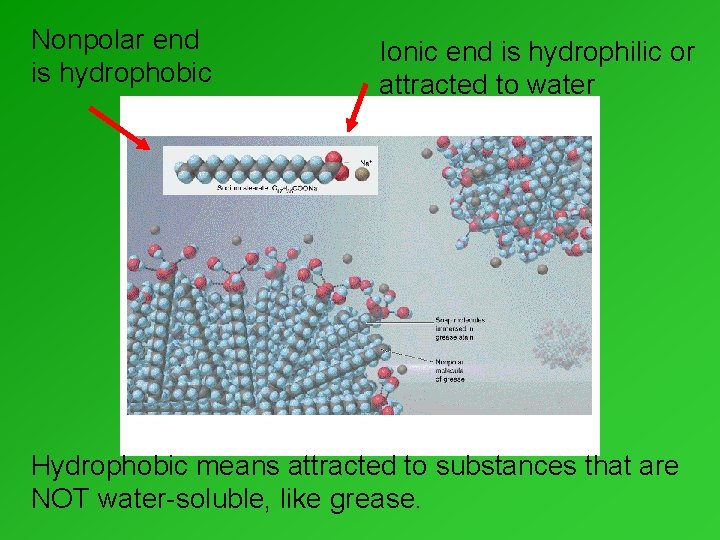
Nonpolar end is hydrophobic Ionic end is hydrophilic or attracted to water Hydrophobic means attracted to substances that are NOT water-soluble, like grease.

Summary: Factors Affecting Solubility • Nature of the solvent and the solute: LIKE DISSOLVES LIKE • Temperature • Pressure (for systems with gases)
- Slides: 25