Temperature Heat Thermal Expansion and Heat Transfer PHYSICS










- Slides: 16

Temperature, Heat, Thermal Expansion, and Heat Transfer PHYSICS I

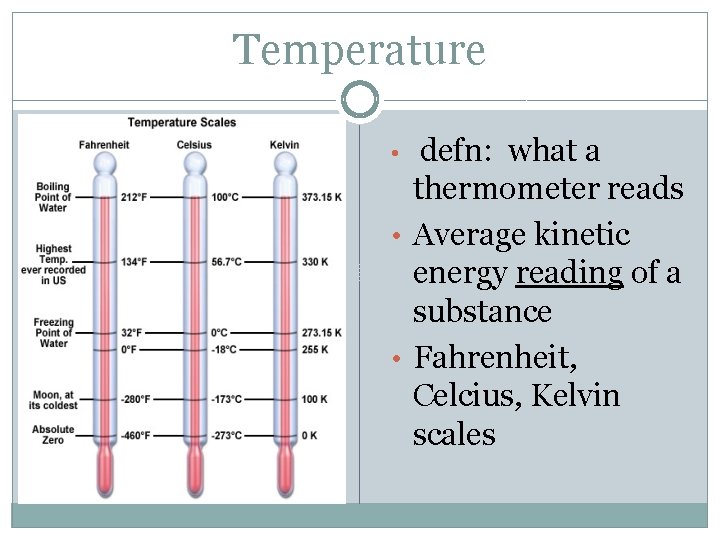
Temperature defn: what a thermometer reads • Average kinetic energy reading of a substance • Fahrenheit, Celcius, Kelvin scales •

Temperature Conversions �°F = (1. 8 x °C) + 32 �°C = (°F – 32) ÷ 32 �K = °C + 273. 15

Heat �Defn: transfer of energy from one object to another due to difference in temp. �Coffee mug examples 80°C coffee sitting on counter of kitchen that is 26°C – what happens? What if cold (5°C) pop can sitting on the counter of same kitchen?

Heat �Internal Energy A substance contains internal energy… NOT heat There is more internal energy in a bathtub full of cold water than a red-hot pin (due to the higher number of molecules; therefore, bathtub has more kinetic energy) But! Heat flows from red-hot pin to tub water

Measuring Heat �Heat is a form energy (Joules) �Calories: calorie Calorie Heat required to raise 1 g of H 2 O by 1°C Kilocalorie = heat required to raise 1 kg of H 2 O by 1°C Unit of energy 1 calorie = 4. 184 joules Used on food labels

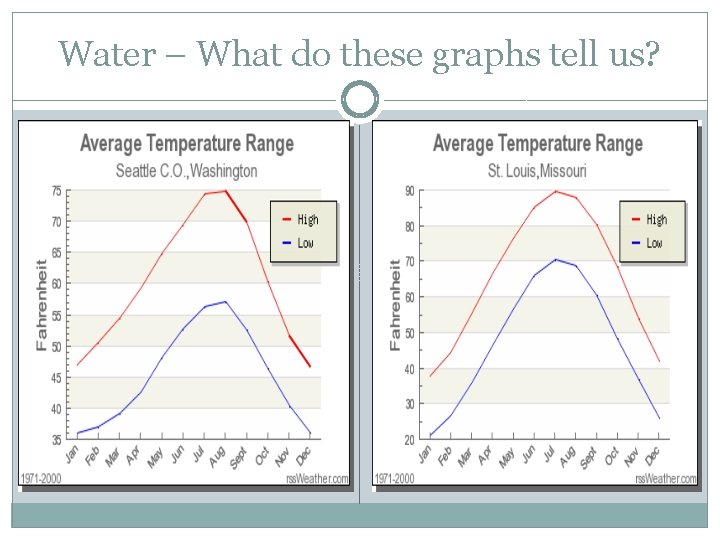
Specific Heat �Defn: quantity of heat required to change temperature of a unit mass of substance by 1° �Water (discussion – see map next slides)

Jet Stream?

Water – What do these graphs tell us?

Thermal Expansion �As Temp. decreases => Substances contract (become more dense) �Except Water! H 2 O MOST dense at 4°C From 4°C to 0°C, water expands (becomes less dense) Which is why ice floats

Conduction �Heat transferred by successive collisions of atoms �Good conductors of heat and electricity: Metals - #1 Ag, #2 Cu, #3 Al… �Poor conductors: Wool, wood, styrofoam = insulators �In February you drive by a house with snow on its roof. Does the house have good insulation or poor? Explain.

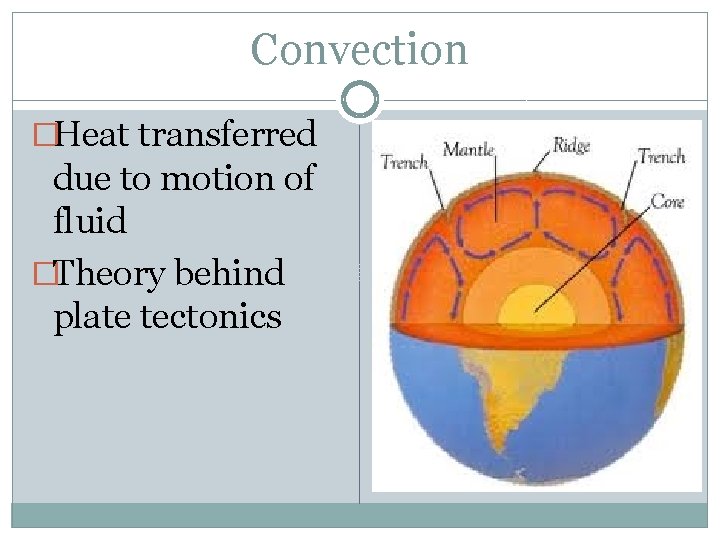
Convection �Heat transferred due to motion of fluid �Theory behind plate tectonics

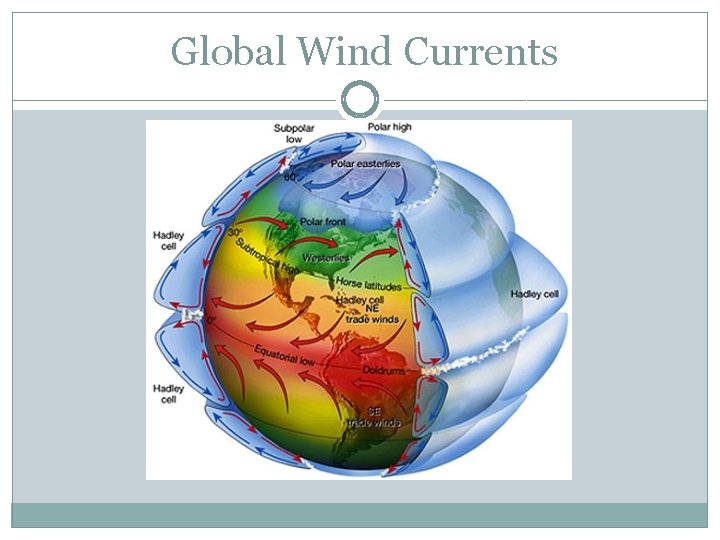
Global Wind Currents

At night or during day? Explain. What happens during other half of earth rotation? Explain.

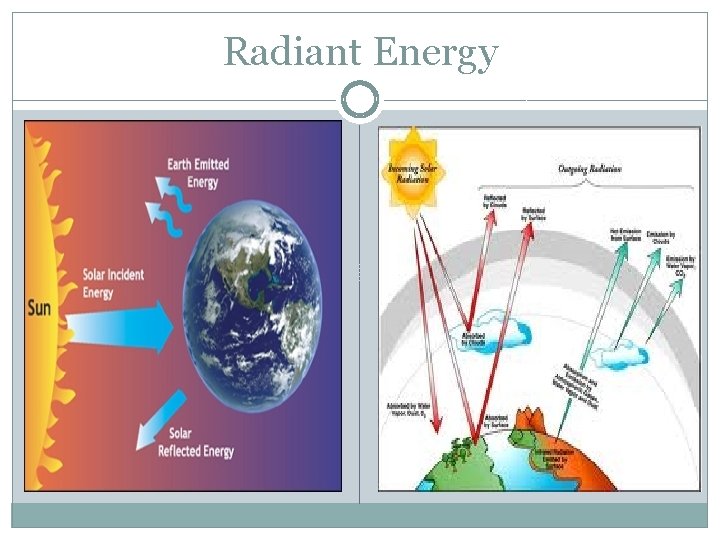
Radiant Energy

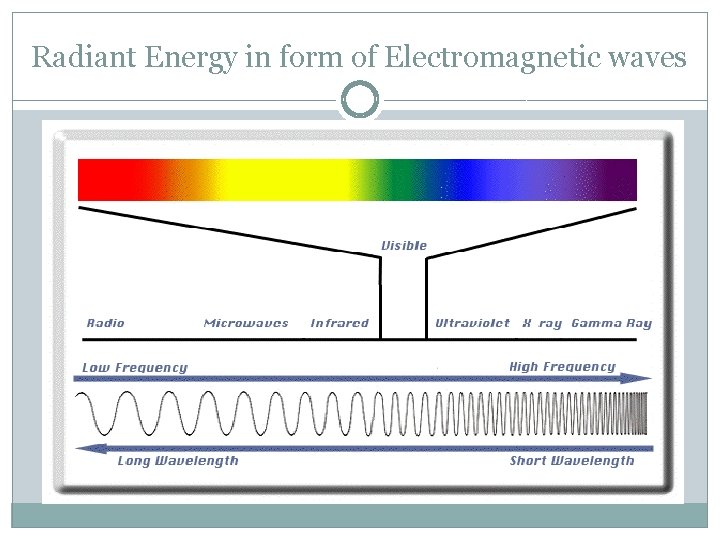
Radiant Energy in form of Electromagnetic waves