Temperature Dependence of Hydrogenbonded Structures of Phenol Cation

Temperature Dependence of Hydrogen-bonded Structures of Phenol Cation Investigated by UV Photodissociation Spectroscopy Itaru KURUSU, Reona YAGI, Ryota KATO, Hikaru SATO, Masataka ORITO, Yasutoshi KASAHARA, Haruki ISHIKAWA School of Science, Kitasato University, Japan harukisc@kitasato-u. ac. jp

Hydrogen-bond networks in molecular clusters • Hydrogen-bond network • • • Ubiquitous and important phenomena Flexible structure, fluctuation Hydrogen-bonded cluster Simple model for the H-bond network at molecular level • Static structures of various systems can be determined by IR spectroscopy and calculation. • • Temperature effect on the hydrogen-bond structure
Temperature effect • Temperature effect on the hydrogen-bond structure Low temperature High temperature Change in relative populations among the isomers • Spectroscopic studies on the temperaturecontrolled cluster ions • • Rare gas tagging H+(Me. OH)n Ref. J. Phys. Chem. A 120, 9203 (2016). Cryogenically-cooled ion trap IR spectroscopy H+(H 2 O)n Ref. J. Phys. Chem. A 107, 4217 (2003).

Spectroscopy of Temperature-controlled H-bonded clusters • UV photodissociation spectroscopy of hydrogenbonded phenol cluster cation trapped in our temperature-variable ion trap Quantitative temperature dependence of relative populations among isomers • [Ph. OH(H 2 O)5]+ • [Ph. OH(Me. OH)3, 4]+ • • Stronger H+ acceptor One H+ donor, two H+ acceptor Different hydrogen-bond networks

Temperature-variable ion trap apparatus 1 st Q-mass filter Ref. Fujihara, et al. J. Phys. Chem. A 113, 8169 (2009). Ishikawa, et al. Chem. Phys. Lett. 514 , 234 (2011). 2 nd Q-mass analyzer Photodissociation laser Octopole ion guide Buffer-gas cooling technique 22 -pole ion trap 8 -pole ion trap Ionization laser Pulsed nozzle Trap Temperature: 20 – 150 K Ion holding time: 5 ms
![Temperature dependence of the PD + spectra of [Ph. OH(H 2 O)5] B 150 Temperature dependence of the PD + spectra of [Ph. OH(H 2 O)5] B 150](http://slidetodoc.com/presentation_image_h2/4c2b10256c6d553fef9adc2dd8e2b6e7/image-6.jpg)
Temperature dependence of the PD + spectra of [Ph. OH(H 2 O)5] B 150 K • At least two isomers exist in our observation. 120 K 100 K • Relative. Apopulations between these isomers strongly depend on the temperature. 80 K 50 K T = 30 K Ref. H. Ishikawa, et al. J. Phys. Chem. Lett. 8, 2541 (2017).
![Stable structures of [Ph. OH(H 2 O)5]+ DFT calculation : Gaussian 09 w. B Stable structures of [Ph. OH(H 2 O)5]+ DFT calculation : Gaussian 09 w. B](http://slidetodoc.com/presentation_image_h2/4c2b10256c6d553fef9adc2dd8e2b6e7/image-7.jpg)
Stable structures of [Ph. OH(H 2 O)5]+ DFT calculation : Gaussian 09 w. B 97 X-D/6 -311++G(3 df, 3 pd) Ring with tail DE 0 = 0. 0 cm-1 Bi-cyclic 99 cm-1 113 cm-1 136 cm-1 46 cm-1 Chain 463 cm-1 465 cm-1 418 cm-1
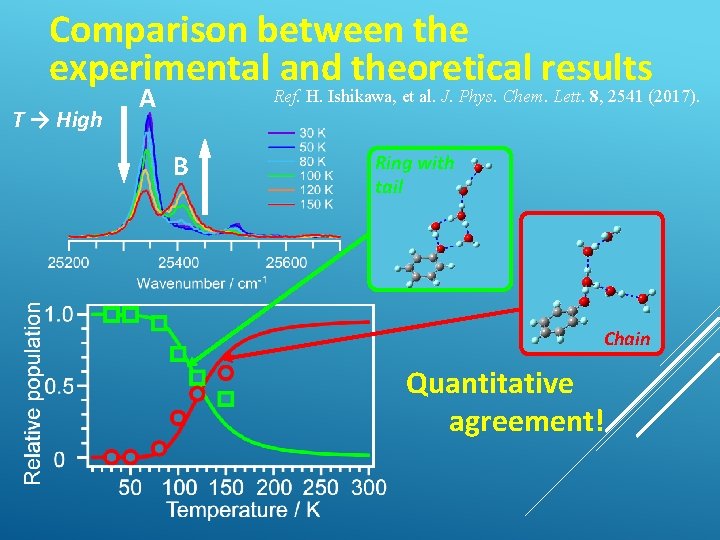
Comparison between the experimental and theoretical results T → High A Ref. H. Ishikawa, et al. J. Phys. Chem. Lett. 8, 2541 (2017). B Ring with tail Chain Quantitative agreement!
![Temperature dependence of the PD + spectra of [Ph. OH(Me. OH)3] 150 K 80 Temperature dependence of the PD + spectra of [Ph. OH(Me. OH)3] 150 K 80](http://slidetodoc.com/presentation_image_h2/4c2b10256c6d553fef9adc2dd8e2b6e7/image-9.jpg)
Temperature dependence of the PD + spectra of [Ph. OH(Me. OH)3] 150 K 80 K • Similar trend to those of [Ph. OH(H 2 O)5]+ Ring type isomer • Stable • Rigid • Dominant at low-T T = 30 K Chain type isomer • Less stable • Flexible • Dominant at high-T
![Temperature dependence of the PD + spectra of [Ph. OH(Me. OH)4] 150 K Ring Temperature dependence of the PD + spectra of [Ph. OH(Me. OH)4] 150 K Ring](http://slidetodoc.com/presentation_image_h2/4c2b10256c6d553fef9adc2dd8e2b6e7/image-10.jpg)
Temperature dependence of the PD + spectra of [Ph. OH(Me. OH)4] 150 K Ring type isomer w/o cooling • Stable • Rigid • Dominant at low-T • Several isomers? Chain type isomer T = 30 K • Much less stable • Flexible • Not so abundant even at high-T

Summary PD spectroscopy of temperature-controlled hydrogen-bonded phenol cations was carried out. • For [Ph. OH(H 2 O)5]+, temperature dependence of the relative abundances between the two distinct isomers were observed and quantitatively discussed. • For [Ph. OH(Me. OH)n]+, similar trend to the hydrated was observed. Theoretical interpretation is still be needed. • Future • • • More detailed temperature dependence Search for other isomers Dynamical properties Thanks to JSPS KAKENHI, Grant-in-Aid for Scientific Research (C)
- Slides: 11