Temperature and Volume Relationship of Gases Charless Law










- Slides: 14

Temperature and Volume Relationship of Gases (Charles’s Law) Temperature and Pressure Relationship of Gases (Gay Lussac’s Law) A. 10/A. 11 In text

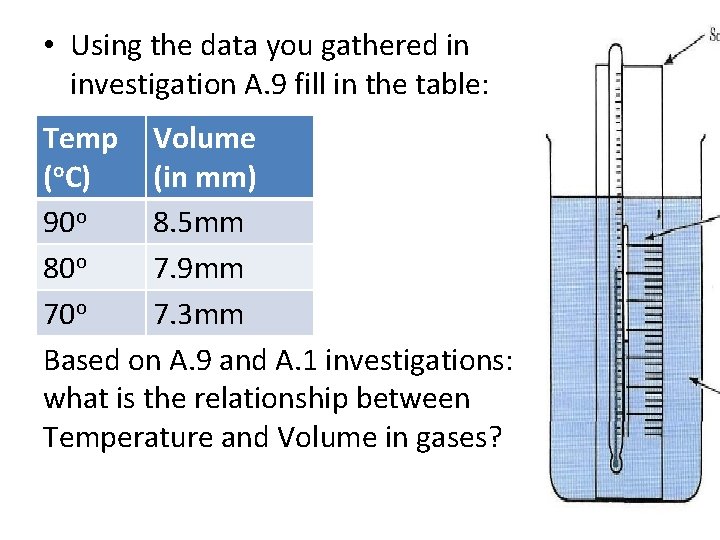
• Using the data you gathered in investigation A. 9 fill in the table: Temp Volume (o. C) (in mm) 90 o 8. 5 mm 80 o 7. 9 mm 70 o 7. 3 mm Based on A. 9 and A. 1 investigations: what is the relationship between Temperature and Volume in gases?

A. 1 Investigation 2: Balloons in Water of Differing Temperatures • What happened to the volume of the balloon in hot water: • What happened to the volume of the balloon in ice water:


• Summarize the relationship between pressure and volume for a gas at a constant temperature. • What is the quantitative way to summarize this relationship? (the mathematical formula? )

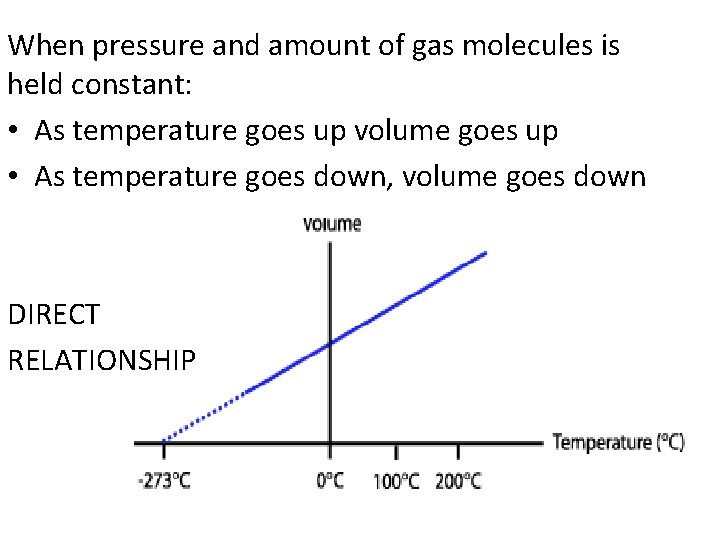
When pressure and amount of gas molecules is held constant: • As temperature goes up volume goes up • As temperature goes down, volume goes down DIRECT RELATIONSHIP

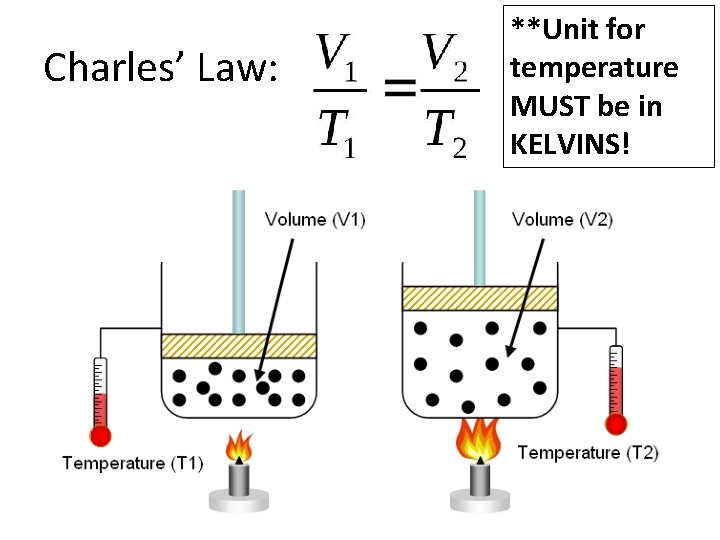
Charles’ Law: **Unit for temperature MUST be in KELVINS!

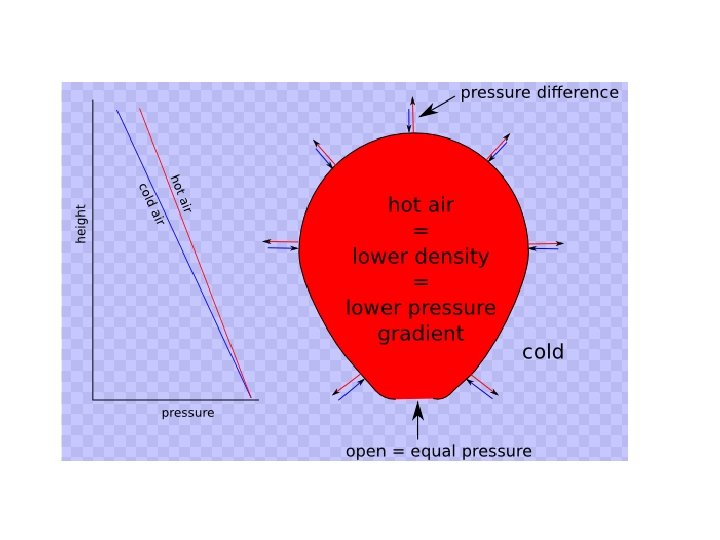
What about the relationship between temperature and pressure at a constant volume? (Gay-Lussac’s Law) • Temperature must still be in units of Kelvin! • Direct relationship

Problem #1 Temperature will affect airships in Tyria – a fantasy world. The small airships have an initial volume of 4, 200 L. To replicate atmospheric conditions in Tyria scientists place it in a chamber and reduce the temperature from 20 to -60 o. C. What will the final volume of the airship be?

Steps to Solve : • First predict how the change will affect the volume (will it go up or down)? • Second: Convert any temperatures to Kelvin (o. C + 273) • Third: define each variable (should be 1 unknown) V 1 : T 1: V 2: T 2: • Fourth: “Plug” the values into the equation

Problem #1:

• First predict how the change will affect the volume (will it go up or down)? If the temperature goes down the volume should be smaller than 4, 200 L. • Second: Convert any temperatures to Kelvin (o. C + 273) T 1: 20 + 273 = 293 K, T 2: -60 + 273 = 213 K • V 1: 4200 L T 1: 293 K V 2: ? T 2: 213 K • Fourth: “Plug” the values into the equation. (Cross multiply) 4200 L = V 2 -> 293 K 213 K V 2 = 3100 L (4200 L) * (213 K) = V 2 (293 K)

When problem has been checked off: 1. Complete A. 11 1 -4 AND A. 11 Extra Practice 1 -6 2. Concept Check 1 -3 All are due next class meeting
