Temperature and Thermometers The temperature of a body
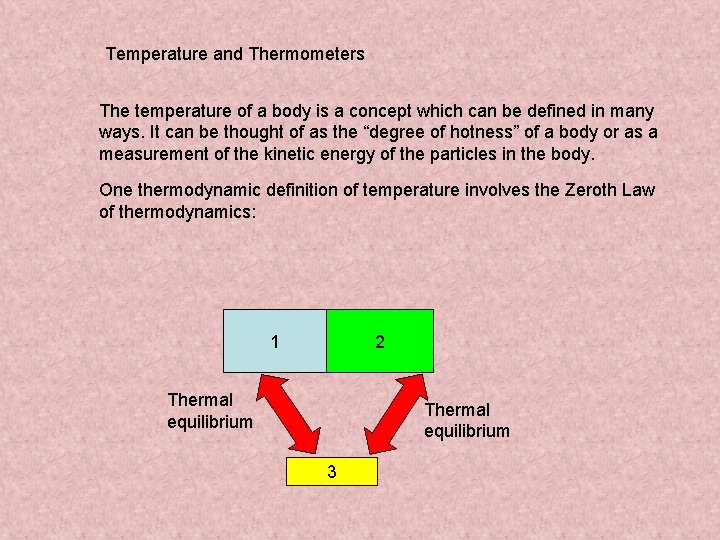
Temperature and Thermometers The temperature of a body is a concept which can be defined in many ways. It can be thought of as the “degree of hotness” of a body or as a measurement of the kinetic energy of the particles in the body. One thermodynamic definition of temperature involves the Zeroth Law of thermodynamics: 1 2 Thermal equilibrium 3
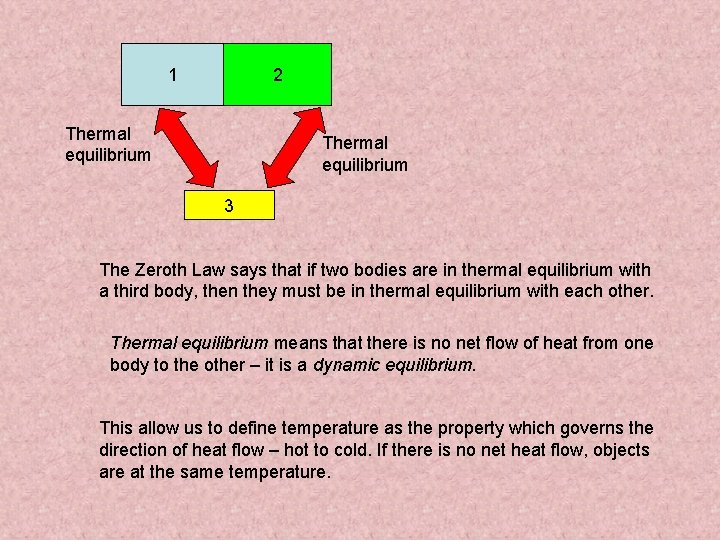
1 2 Thermal equilibrium 3 The Zeroth Law says that if two bodies are in thermal equilibrium with a third body, then they must be in thermal equilibrium with each other. Thermal equilibrium means that there is no net flow of heat from one body to the other – it is a dynamic equilibrium. This allow us to define temperature as the property which governs the direction of heat flow – hot to cold. If there is no net heat flow, objects are at the same temperature.
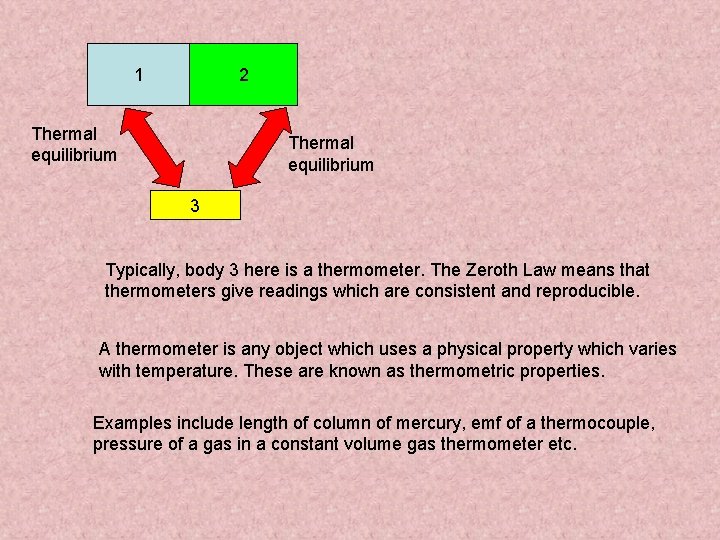
1 2 Thermal equilibrium 3 Typically, body 3 here is a thermometer. The Zeroth Law means that thermometers give readings which are consistent and reproducible. A thermometer is any object which uses a physical property which varies with temperature. These are known as thermometric properties. Examples include length of column of mercury, emf of a thermocouple, pressure of a gas in a constant volume gas thermometer etc.

Temperature scales Any temperature scale requires the two fixed points to be established. These are two places where the temperature is defined to be a certain value. For example, the fixed points of the Celsius scale are the triple point of water and the boiling point of water. The triple point of water is the temperature at which water exists in all three phases in equilibrium with each other. The scale is then divided into 100 divisions between these points, thus making Celsius a centigrade scale.
- Slides: 4