Technology Transfer For Plant Breeders Agricultural Research Service

Technology Transfer For Plant Breeders Agricultural Research Service Office of Technology Transfer

What we will cover: üMaterial Transfer Agreements for plants üARS’ process for releasing plant material üPlant Variety Protection Committee üMethods for protection of the intellectual property in plant varieties

Can unreleased germplasm be shared for breeding and/or evaluation? yes, with appropriate documentation Documentation: ØCooperative research agreement ØMaterial Transfer Agreement (MTA) ØStandard Material Transfer Agreement (SMTA)

What are the purposes of an MTA? Allow others to evaluate ARS germplasm Allow others to breed with ARS germplasm Allow ARS to evaluate non-ARS germplasm Allow ARS to breed with non. ARS germplasm

What are the purposes of an MTA? (cont’d) Confidential transfer of material & related information ØProtects proprietary & patent rights ØRestricts the use of material & related information MTA does not authorize cooperative research and requires the signature of the RL and TTC!

How do you develop an MTA? Different MTA templates exist for evaluation and breeding MTAs can be custom written for specific situations Consult with your TTC for help in selecting or developing an MTA Be sure to have your TTC review any MTA provided by another party

What is an SMTA? Developed by International Treaty on Plant Genetic Resources for Food & Agriculture (PGRFA) Establishes a multilateral system (MLS) for facilitated access to PGRFA and for sharing benefits arising from their use Legally-binding contract with prenegotiated terms under the UN Food & Agriculture Organization (FAO) SMTA must accompany each transfer of covered PGRFA materials

What is covered by an SMTA? 64 food & feed crops listed in Annex I PGRFA in the International Agricultural Research Center Collections PGRFA ex situ collections of national governments or public domain PGRFA donated or available to MLS by public or private entities

What does the SMTA NOT cover? Crops not listed in Annex I Chemical, pharmaceutical or other non-food or non-feed uses of Annex I crops Public collections not under national government control or private collections PGRFA found in situ

What are the SMTA rules? SMTA must be reported to the International Treaty All non-confidential information resulting from using PGRFA must be made available to the MLS Research & breeding materials that incorporate PGRFA must be exchanged under the terms of the SMTA The full text of the SMTA is available at ftp: //ftp. fao. org/ag/agp/planttreaty/agreements/smta/SMTAe. pdf

What sources of germplasm might be subject to SMTA obligations? ØTreaty Party ØInternational Agricultural Research Center ØNPGS, for accessions acquired under SMTA ØResearcher who received PGRFA under an SMTA

NPS authorization is required to use SMTA material in an ARS breeding program Seedlings bred from publically available germplasm (ARS germplasm) with SMTA protected germplasm becomes SMTA protected germplasm If an ARS breeding program incorporates SMTA germplasm, then it is not publically available without restrictions Check with your RL, National Program Leader (NPL) & TTC

What is the procedure for using SMTA material in breeding? SY / RL / NPL decide if SMTA material should be used SY contacts TTC; TTC sends SMTA to OTT for processing OTT gets official NPS approval TTC send approved SMTA to SY If germplasm/variety is released using SMTA material, SY includes this fact in the Official Release Notice

System for releasing plant materials: Policy ARS scientists have a long and successful history of developing both finished varieties and enhanced germplasm and transferring them through public release. The ARS policy is to public release these materials without IP protection. However, exceptions to this policy for certain ARS developed or co-developed varieties may be made on a case-bycase basis.

Plant material release system is designed to: Facilitate the release process for plant materials developed or co-developed by ARS. Allow timely decisions on IP protection on a case-by-case basis.

Process Use of a new electronic systems, which is a subsystem, of ARIS for ALL plant releases whether or not IP protection is requested. A new committee to make decisions on seeking IP for new plant varieties based on Training for ARS breeders established criteria. and management on the new system. Outreach to our customers and stakeholders.

What plant materials can be officially released? ØFinished varieties ØEnhanced germplasm ØGenetic lines / stocks
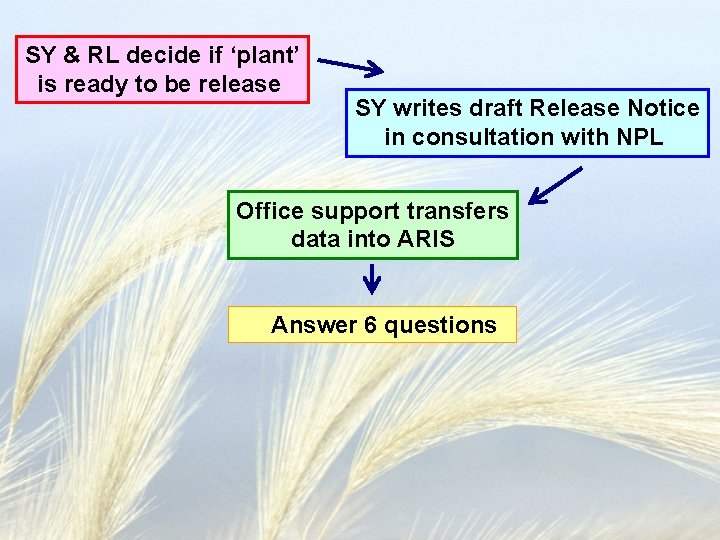
SY & RL decide if ‘plant’ is ready to be release SY writes draft Release Notice in consultation with NPL Office support transfers data into ARIS Answer 6 questions

Question 1 Was this variety developed in cooperation with a university experiment station or other organization? What resources were contributed by the cooperator (e. g. technicians, breeders, land, facilities, services, test data, etc. )? List all cooperator employees who were directly involved in the breeding and selection of this material. Do not forget to state if SMTA material was used

Question 2 Enter the complete draft text of proposed release notice. Do not forget to state if SMTA material was used

Question 3 Who is expected to use the released plant material and how they will use it? What public or private organizations have requested this material for research, breeding or testing purposes? Have you consulted with any commodity groups?

Question 4 List any publications and/or public use of this plant material. Has the material been provided to anyone outside of USDA for field-testing purposes? Have seeds, cuttings, fruit or other materials been sold by cooperators? If so, give dates.

Question 5 Explain how protection will facilitate technology transfer that would otherwise not occur if the variety is publicly released. Does the variety have any special characteristics that would make protection desirable (e. g. , niche market use, transgenic, incorporates proprietary technology, incorporates third party IP).

Question 6 Is there an international market for this variety? If so, in what countries? Have you sent the variety to anyone outside the U. S. for testing or breeding? If so, provide countries and dates.
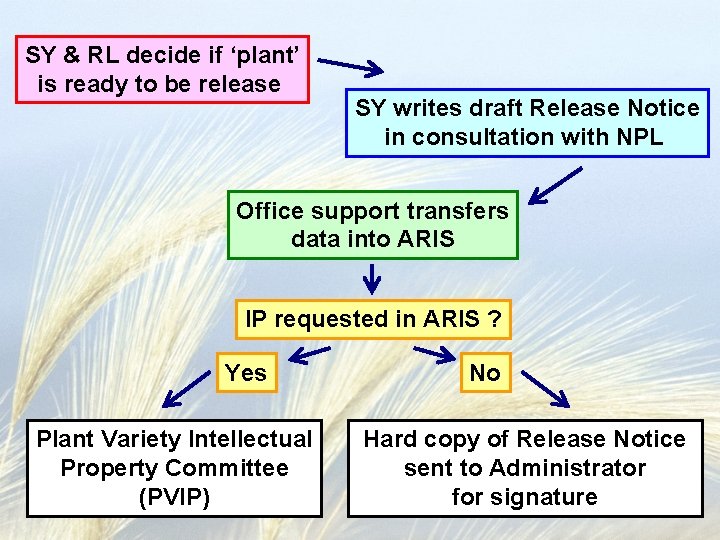
SY & RL decide if ‘plant’ is ready to be release SY writes draft Release Notice in consultation with NPL Office support transfers data into ARIS IP requested in ARIS ? Yes Plant Variety Intellectual Property Committee (PVIP) No Hard copy of Release Notice sent to Administrator for signature

Plant Variety Intellectual Property Committee (PVIP) Meets quarterly Co-chaired by Gail Wisler NPS / Martha Steinbock OTT June Blalock (permanent) *TTC *Breeder *RL *Area Director or designee *NPL *Patent Advisor (if a patentable crop) *Marketing & Information * Member varies based on variety in question

What are criteria for deciding if protection is sought? If co-owned, has the co-owner already expressed a favorable opinion about seeking protection? Do we (or our co-owner) have experience with protecting and/or licensing this type of plant? If so, has this helped facilitate technology transfer? Are there specific characteristics of this variety that make protection desirable? Have commercial seed companies or nurseries expressed an interest in marketing and producing the variety for sale?

What are criteria for deciding if protection is sought? (cont’d) Who are the U. S. end users (stakeholders) of this variety? Have they been consulted yet? If so, do they favor protection? Would seeking protection enhance the competitiveness of U. S. growers in world markets and/or help level the playing field internationally? Could seeking protection and licensing this variety impact negatively the ability of our breeders to access foreign germplasm? In general, would seeking protection enhance technology transfer for this variety?

Committee Recommendations üDefer for more information üRecommend public release üRecommend filing a Plant Patent or PVPC Application & Licensing üRecommend for PVPC & public release by university partner üRecommend for filing Plant Breeder’s Rights & list countries of interest

What are the ways in which plant material may be protected? Asexually propagated plants: Plant Patent (PP) http: //www. uspto. gov/web/offices/pac/plant/ Sexually propagated plants: Plant Variety Protection (PVP) http: //www. ams. usda. gov/AMSv 1. 0/ams. fetch. Template. Data. do? templat e=Template. C&nav. ID=Plant. Variety. Protection. Office&right. Nav 1=Plant. Var iety. Protection. Office&top. Nav=&left. Nav=Scienceand. Laboratories&page =Plant. Variety. Protection. Office&result. Type=&acct=plntvarprtctn

What information is needed for PP or PVP application? Complete botanical description leaves simple, entire, symmetrical, lanceolate, apiculate tip; average 4. 5 cm length, 2. 4 cm width; adaxial surface black (RHS 202 A); etc. Distinctness/novelty of the claimed plant Comparison with data to similar varieties Breeding history related to prior art §Not obvious (PP) §Not essentially derived (PVP) OTT submits application

Handbook for Plant Breeders Available on Web: http: //www. ars. usda. gov/SP 2 User. Files/Place/0000/OTTGeneral/ARSPlant. Breeders. Handbook. pdf

Office of Technology Transfer is here to help Your TTC & Patent Advisor are a source of information & help. Call early & call often.
- Slides: 33