Techniques for plant transformation In molecularbiology transformation is

Techniques for plant transformation

• In molecularbiology, transformation is genetic alteration of a cell resulting from the direct uptake, incorporation and expression of exogenous genetic material (exogenous DNA) from its surroundings and taken up through the cell membrane(s).

• Transformation occurs naturally in some species of bacteria, but it can also be affected by artificial means in other cells. For transformation to happen, bacteria must be in a state of competence, which might occur as a time-limited response to environmental conditions such as starvation and cell density.

Techniques for plant genetic transformation • Indirect method- Agrobacterium mediated gene transfer • Direct methods- – Particle bombardment (biolistics) – Microprojectile gun method – Electroporation – Silicon carbide fibres – Polyethylene glycol (PEG)/protoplast fusion – Liposome mediated gene transfer

Agrobacterium mediated gene transfer Agrobacterium– Soil borne, gram negative, rod shaped, motile found in rhizosphere – Causative agents of “Crown gall” disease of dicoltyledones – Have ability to transfer bacterial genes to plant genome by nature. – Attracted to wound site via chemotaxis in response to chemicals (sugar and Phenolic molecules: acetosyringone) released from damaged plant cells – Contains Ti plasmid which can transfer its T-DNA region into genome of host plants
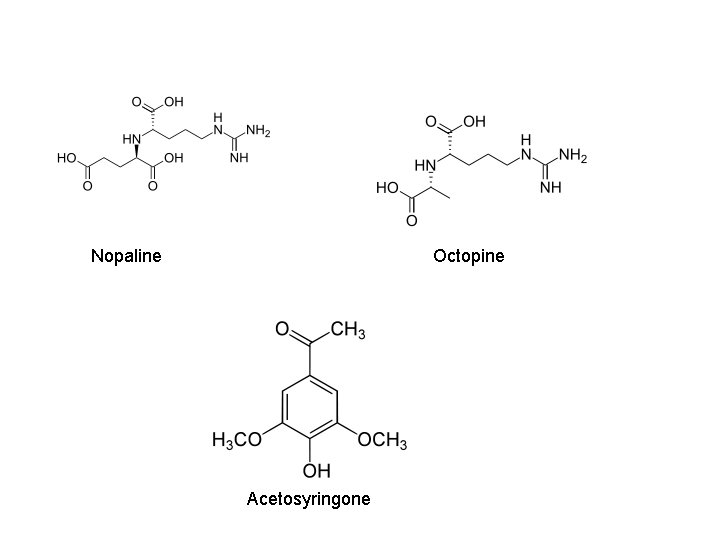
Nopaline Octopine Acetosyringone

Ti-plasmid features • Two strains of Ti-plasmid: -Octopine strains- contains two T-DNA region: TL (14 kb) and TR ( 7 kb) -Nopaline strains- contain one T-DNA region(20 kb) • Size is about 200 kb • Has a central role in Crown-gall formation • Contains one or more T-DNA region that is integrated into the genome of host plants • Contain a vir region ~ 40 kb at least 8~11 vir genes • Has origin of replication • Contains a region enabling conjugative transfer • Has genes for the catabolism of opines
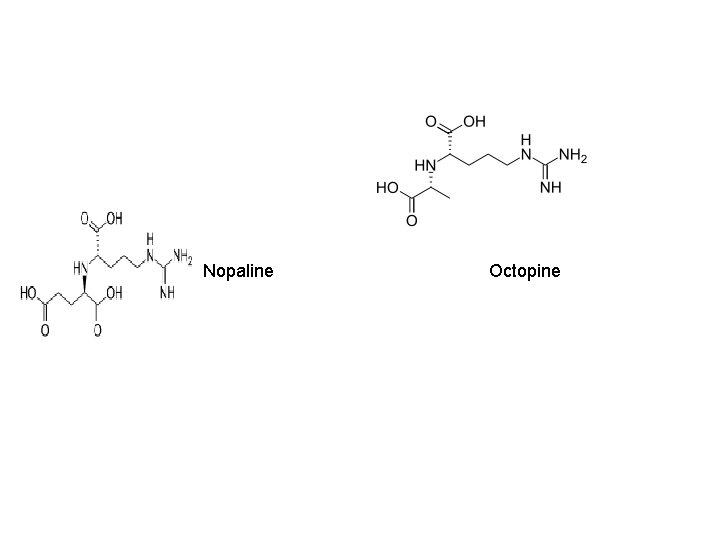
Nopaline Octopine
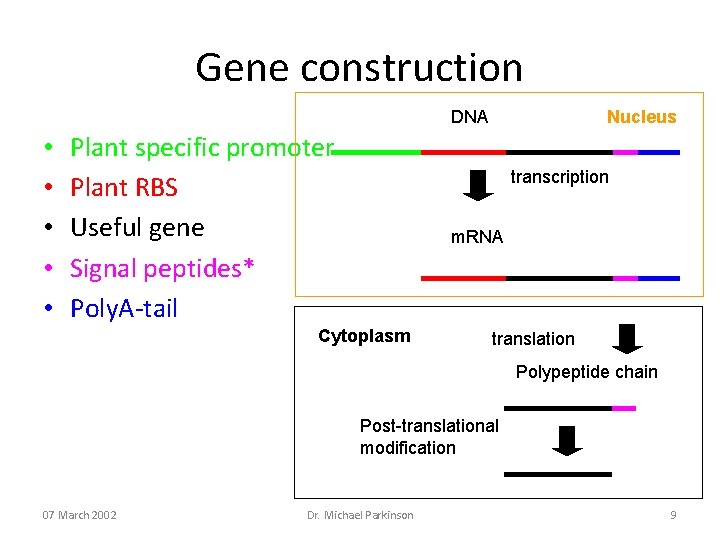
Gene construction DNA • • • Plant specific promoter Plant RBS Useful gene Signal peptides* Poly. A-tail Nucleus transcription m. RNA Cytoplasm translation Polypeptide chain Post-translational modification 07 March 2002 Dr. Michael Parkinson 9
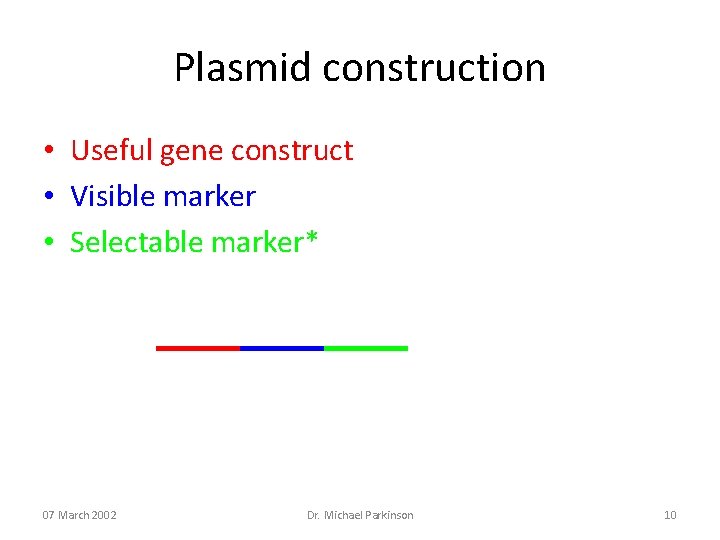
Plasmid construction • Useful gene construct • Visible marker • Selectable marker* 07 March 2002 Dr. Michael Parkinson 10
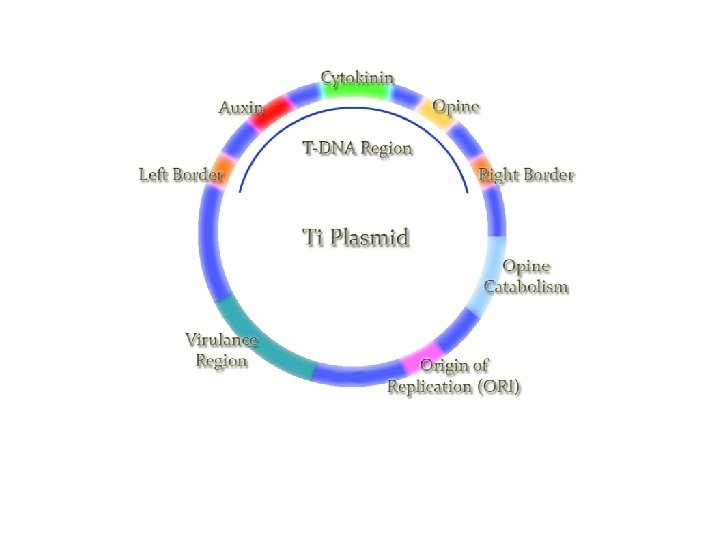

T-DNA • Size~ 12 to 24 kb • Left and right border sequence(24 -bp) which will be transferred into genome of host plant • Oncogenes e. g Auxin, cytokinin, opines • tm 1 gene for determining the tumour size • the T-DNA contains eight potential genes - these are eukaryotic in nature (eukaryotic promoters, monocistronic, eukaryotic polyadenylation signals, eukaryotic translation mechanisms) • crown gall tumorigenesis is due to the "activation" of unregulated phytohormone synthesis in the transformed cells
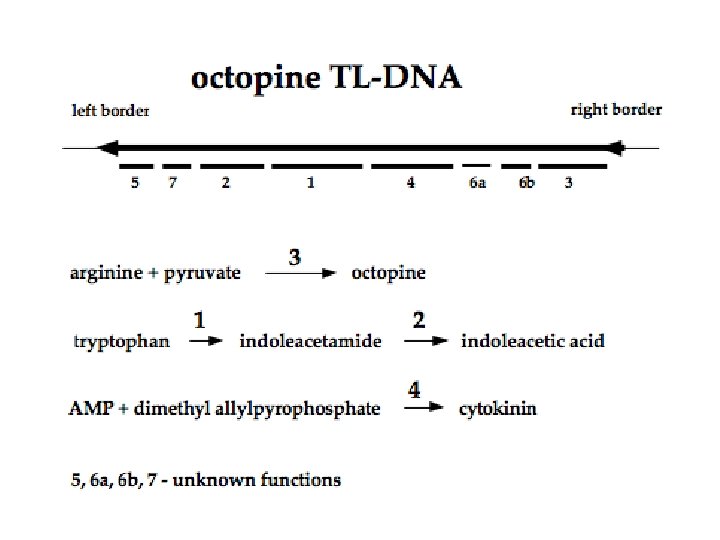

Forms of T-DNA that are found in Agrobacterium • ds circles - found only in induced bacteria, not (apparently) in plant cells • ds linear T-DNA - found only in induced bacteria, not (apparently) in plant cells • ss linear T-DNA - found in bacteria and plant cells • what is not found - Ti plasmids with evidence that TDNA has been precisely deleted

Process of T-DNA transfer and integration 1. Signal recognition by Agrobacterium: -Agrobacterium perceive signals such as sugar and phenolic compounds which are released from plants 2. Attachment to plants cells: Two step processes: i) initial attachment via polysaccharide ii) mesh of cellulose fiber is produced by bacteria. Virulence genes (chv genes) are involved in the attachment of bacterial cells to the plants cells. 3. Vir gene induction: Vir. A senses phenolics ans subsequently phosphorylating and thereby activating Vir. G then induces expression of all the vir genes. 4. T-strand production: Vir. D 1/vir. D 2 complex recognises the LB and RB. vir. D 2 produces single-stranded nicks in DNA. Then

Process of T-DNA transfer and integration (continu) 5. Transfer of T-DNA out of bacterial cells: T-DNA/Vir. D 2 is exported from the bacterial cell by “T-pilus” composed of proteins encoded by vir. B operon and Vir. D 2. Vir. E 2 and Vir. F are also exported from bacterial cells. 6. Transfer of the T-DNA and Vir proteins into the plant nuclear localization: T-DNA/Vir. D 2 complex and other Vir proteins cross the plasma membrane through channels formed from Vir. E 2 protect T-DNA from nucleases, facilitate nuclear localization and confer the correct conformation to the TDNA/vir. D 2 complex for passage through the nuclear pore complex (NPC). The T-DNA/Vir. D 2/Vir. F 2 /plant protein complex the nucleus through nuclear pore complex. And integrated into host chromosome.

Practical application of Agrobacteriummediated plant transformation • 1. Agrobacterium mediated transformation methods are thought to induce less rearrangement of the transgene. • Lower transgene copy number that direct DNA delivery methods. • Successful production of transgenic plants depends on the suitable transformation protocols.

Agrobacterium-mediated transformation of Tobacco • Several factors have to be considered in the design and implementation of any plant transformation study: • -1. plant tissue to be transformed. The explant should be capable of producing whole plant and should contain high number of cells that are competent for transformation. • 2. The vector used to deliver the transgene into the genome of plant. Vector should Ti-plasmid binary vector which have LB and RB of Tiplasmid, bacterial selectable marker gene, selectable marker gene for selection of transformed plant. and Multiple cloning sites. • 3. Strain of Agrobacterium used: The choice of strain for crop plants is not critical to the success of transformation but for recalcitrant plants, choice of strains is a major factor to successful transformation. For dicotyledons plants: LBA 4404, GV 3001 etc. , For cereals ( which are not infected by naturally infected) Supervirulent strain such as EHA 101, EHA 105 are used

The basic protocol used for any Agrobacteruim mediated transformation experiments 1. Identify a suitable explants: Suitable plant tissue is removed and sterilized. Leaf is used for Tobacco. 2. Co-cultivate with the Agrobacterium: Leaf tissue is cut into small pieces and placed into a culture of Agrobacterium for about 30 mins. The explants are subsequently removed from the bacterial culture and placed on to the MS medium that contain no selective agent. The incubation of explants with Agrobacterium is allowed to continue for 2 days to allow transfer of the T-DNA transfer to the plant cells. 3. Kill the Agrobacterium with a suitable antibiotic: The explants are removed from the medium and washed in antibiotic (cefotaxime) solution that kill Agrobacterium cells.

The basic protocol used for any Agrobacteruim mediated transformation experiments (Conti-) 4. Select for transformed plant cells: The explant are transferred to fresh solid medium supplemented with a selective agent (kanamycin). It also contains cefotaxime. Auxin, Cytokinin are used to encourage the regeneration of by organogenesis. High cytokinin to auxin ratio promotes shoot formation from the explants. 5. Regeneration of whole plant : The shoot can be rooted by placing them on solid medium containing a high auxin to cytokinin ratio.
Direct gene transfer methods • The trem “Direct gene transfer” is used to discriminate between the methods of plant transformation that rely on Agrobacterium (indirect method) and those that do not (direct methods). Direct gene transfer methods all rely on the delivery of large amount of naked DNA whilst plant is transiently permeabilised. Direct methods. Particle bombardment (biolistics) Microprojectile gun method Electroporation Silicon carbide fibres Polyethylene glycol (PEG)/protoplast fusion Liposome mediated gene transfer

Advantages and disadvantages of direct gene transfer • Adv- Widespread use of transformation of cereal crops that initially proved difficult to transformation with Agrobacterium. • Disadv- They tend to lead higher frequency of transgene rearrangement and higher copy number. This can lead to high frequency of gene silencing.

Particle bombardment • Why Biolistics or Biolistic bombardment? • Is the most powerful method for introducing nucleic acids into plants, because the helium pressure can drive microcarriers through cell walls • Is much easier and less time consuming than microinjecting nucleic acids into plant cells or embryos • Allows transformation of animal cells that have unique growth requirements and that are not amenable to gene transfer using any other method • Requires less DNA and fewer cells than other methods, and can be used for either transient or stable transformation

Principle • The gold or tungsten particles are coated with the DNA that is used to be transform the plant tissue. • The particles are propelled at high speed into the target plant material where the DNA is released within then cell and can integrate into the genome. • Two types of plant tissues are used for particle bombardment: • a) Primary explants that are bombarded and then induced to become embryogenic • b) Proliferating embryonic cultures that are bombarded and then allowed to proliferate further and subsequently regenerate. •

PDS-1000/He bombardment System Fig: The PDS-1000/He system, shown here with magnified view of the Hepta adaptor. Fig: Schematic representation of the PDS-1000/He system upon activation. The arrows indicate the direction of helium flow

How the PDS-1000/He System Works • The sample to be transformed is placed in the bombardment chamber, which is evacuated to subatmospheric pressure • The instrument is fired; helium flows into the gas acceleration tube and is held until the specific pressure of the rupture disk is reached • The disk bursts, and the ensuing helium shock wave drives the macrocarrier disk (which carries the coated microparticles) a short distance toward the stopping screen • The stopping screen retains the macrocarrier, while the microparticles pass through the screen into the bombardment chamber and penetrate the target cells • The launch velocity of microcarriers depends on a number of adjustable parameters: the helium pressure (rupture disk selection, 450– 2, 200 psi), the amount of vacuum, the distance from the rupture disk to the macrocarrier, the distance from the microcarrier launch assembly to the stopping screen, and the distance between the stopping screen and target cells. Adjusting these parameters allows you to produce a range of velocities to optimally transform many different cell types.

Polyethylene glycol (PEG) mediated transformation method ü Plant protoplast can be transformed with naked DNA by treatment with PEG in the presence of divalent cations e. g. , Calcium. ü PEG and divalent cations destabilize the plasma membrane of the plant protoplast and rendered it permeable to naked DNA. ü DNA enters the nucleus and integrates into the host genome. Disadvantage and advantages: ØRegeneration of fertile plants from protoplasts is a problematic for some species. ØThe DNA used for transformation is also susceptible to degradation and rearrangement. ØDespite the limitations, the technique have the advantages and protoplast can isolated and transformed in number of plants species.


Electroporation • It can be used to deliver DNA into plant cells and protoplasts. • The genes of interest require plant regulatory sequence. • Plant materials is incubated in a buffer solution containing DNA and subjected to high-voltage electric pulse. • The DNA then migrates through high-voltage-induced pores in the plasma membrane and integrates into the genome. • It can be used to transform all the major cereals particularly rice, wheat, maize. • Advantages and disadvantages: • Both intact cells and tissue can be transformed. • The efficiency of transformation depends upon the plant materials, electroporation and tissue treatment conditions used for transformation. • ~40 to 50% incubated cells receive DNA • ~50% of the transformed cells can survive

Silicon carbide fibres-Whiskers • Plant materials (Cells in suspension culture, embryos and embryoderived callus) is introduced into a buffer containing DNA and the silicon fibers which is then vortexed. • The fibers (0. 3 -0. 6 μm in diameter and 10 -100μm long) penetrate the cell wall and plasma membrane, allowing the DNA to gain access to the inside of the cells. • Disadvantages and advantages • The drawbacks of this technique relate to the availability of suitable plant material and the inherent dangers of the fibers, which require careful handing. • Many cereals, produce embryonic callus that is hard and compact and not easily transformed with this technique. • Despite the some disadvantages, this method is recently used for successful transformation of wheat, baerly, and maize without the need to cell suspension.

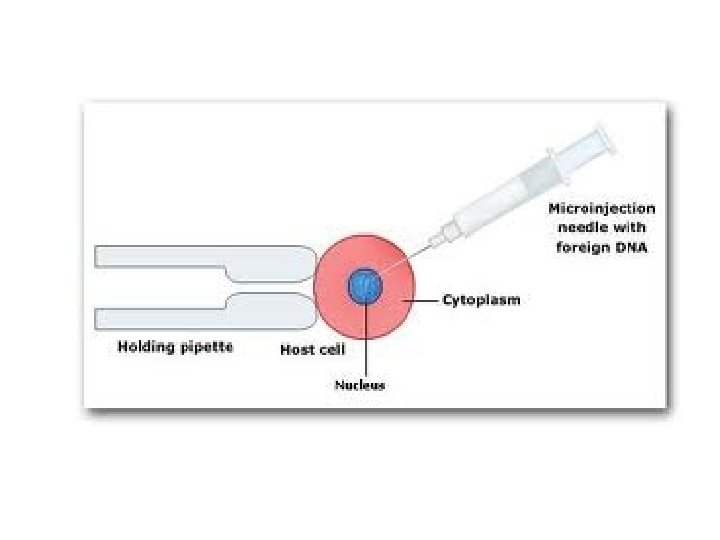
Microinjection ØMicroinjection techniques for plant protoplasts utilize a holding pipette for immobilizing the protoplast while an injection pipette is utilized to inject the macromolecule. Ø In order to manipulate the protoplasts without damage, the protoplasts are cultured for from about 1 to 5 days before the injection is performed to allow for partial regeneration of the cell wall. ØIt was found that injection through the partially regenerated cell wall could still be accomplished and particular compartments of the cell could be targeted. Ø The methods are particularly useful for transformation of plant protoplasts with exogenous genes.
- Slides: 32