TBG 722 SNYAL LETM Ras Yola Sezgin GNE

TBG 722 SİNYAL İLETİMİ Ras Yolağı Sezgin GÜNEŞ Ondokuz Mayıs Üniversitesi, Tıp Fakültesi, Tıbbi Biyoloji AD

Ras Ailesi • G proteini α-alt biriminde olduğu gibi kendisine GTP bağlayarak aktive olan bir proteindir. • Hücresel sinyal iletiminde görevli küçük GTPazlar kodlar • Aktivasyon mekanizması – Hücre büyümesi – Farklılaşma – Sağ kalım • Ras hc dışı sinyaller ile uyarılır ve sinyali nükleusa iletir • Ras mutasyonları, RAS’ı kalıcı olarak aktive eder, ekstrasellüler sinyal yokluğunda bile hc içerisine sinyal iletimine neden olur. Bu sinyaller hc büyümesi ve bölünmesine neden olur. Ras sinyal hataları onkogenez ve kanserle ilişkilidir. • Ras’ı aktive eden mutasyonlar tümörlerin %20 -25’inde bulunur, bazı spesifik tümörlerde ise %90 oranında bulunur.

Tarihçe • İlk kez Edward M. Scolnick ve ark. , tarafından NIH’te Harvey (the HRAS oncogene) ve Kirsten (KRAS) sarcoma virusleri tarafından onkogenik transformasyona yol açan ras onkogenlerinde tanımlanmıştır. Bu virüler ilk kez 1960 larda Jennifer Harvey ve Werner Kirsten tarfından tanımlanmıştır. • 1982’de, Harvard’tan Geoffrey M. Cooper, NIH’ten Mariano Barbacid, Stuart A. Aaronson, MIT’den Robert Weinberg hücre hatlarında transformasyona yol açan genler tanımlamışlardır. Sonuç olarak nöroblastoma hücrelerinde NRAS geni tanımlanmıştır. • Üç RAS geni H-Ras, N-Ras ve K-Ras 4 A ve K-Ras 4 B (K-Ras aleternatif splcing ürünü).

Ras üst ailesi • 100’den fazla protein – Yapı, dizi ve işlev özelliklerine göre gruplandırılmış – 9 ana aile • Ras, Rho, Rab, Rap, Arf, Ran, Rheb, Rad, Rit, Miro • Her aile GTPaz ve nükleotid değişim bölgesi içerir. Çevreleyen diziler işlevsel özgüllüğü sağlar • Ras ailesi hc proliferasyonundan sorumlu

Ras ailesi • G proteini α-alt biriminde olduğu gibi kendisine GTP bağlayarak aktive olan bri proteindir. • Ras’ın aktivasyonu, GEF (Guanin nükleotid Exchance Faktör) proteini yardımıyla gerçekleşir. Bunun için GEF, Ras’ın GDP bağını kopararak onun yerine GTP bağlar. Ras-GTP kompleksinin aktivasyonu daha sonra GAP (GTPase aktive edici protein) tarafından hidroliz olur. Yani GTP’den bir fosfat koparılarak, GDP oluşur ve ras proteini inaktif hale geçer. • Kanserlerindeki ras genlerinde oluşan mutasyonlar sonucunda, Ras proteinindeki GTP hidrolize olmaz, yani GTP’den –P- kopararak GDP oluşmaz. Böylece Ras proteini sürekli aktif durumda olan hücreler, büyüme faktör uyarısına gerek kalmadan sınırsız olarak çoğalıp doku oluştururlar.

• Raf proteini bir çeşit protein kinaz olup, ancak ras proteini ile birleşince aktif hale gelir. Oluşan ras-raf kompleksi sinyalin ilerlemesini sağlar. • MAP Kinaz: MAP kinazlar mayalardan insanlara kadar bütün ökaryotik hücrelerde sinyal iletiminde rol oynayan serin/treonin protein kinazlarıdır. • MAP Kinazın en iyi bilinen formu ERK (Ekstrasellüler sinyal regulated kinaz) olup hücrede önemli görevleri vardır. Örneğin, ERK, büyüme faktörü reseptör ilişkisi sonucu alınan hücre çoğalma sinyalinin, hedef bölgeye ulaşmasında temel bir rol oynar. Bu olayda sinyal görevi yapan büyüme faktörleri, ya protein tirozin kinaz reseptörlerine ya da G proteini reseptörlerine bağlanarak hedef proteinin uyarılmasını sağlar.

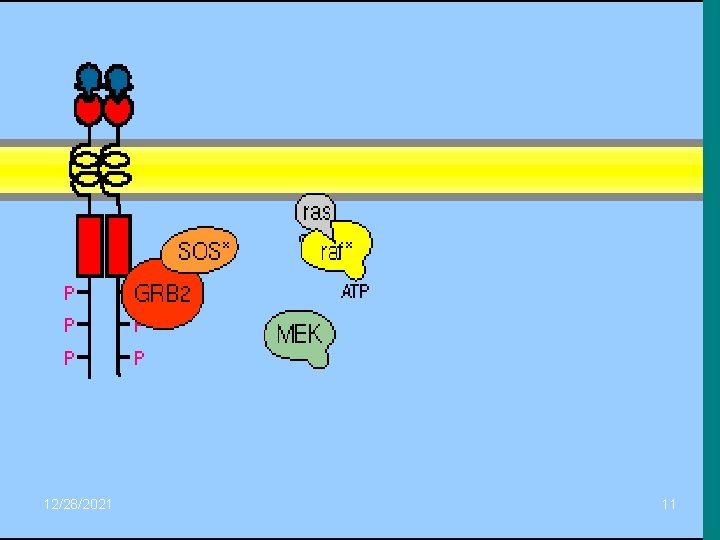
• GF, plazma membranındaki tirozin kinaz reseptörüne bağlanır ve bunu takiben otofosforilasyonla protein-tirozin kinaz domainlerindeki tirozinler fosforlanır. Reseptördeki tirozinlerin fosforlanması, SH 2 bölgesi içeren GRB 2 proteini için bağlanma yeri oluşturur. Proteini için bağlanma yeri oluşturur. • Aktif protein kinazı C’nin önemli bir diğer hedefi, nükleusta DNA’nın çeşitli transkripsiyon faktörlerini fosforlayıp gen ekspresyonunu uyarmaktır. Buna örnek olarak, immun cevabın oluşumunda görev yapan NF-k. B transkripsiyon faktörünün aktifleşmesini verebiliriz. Bu olayda sitozoldeki inaktif NF-k. B ile Ik. B kompleks halinde bulunur. Protein kinaz C aktifleşince, Ik. B fosforlanır ve proteolitik olarak parçalanır. Ik. B’nin parçalanması sonucu serbest kalan NF-k. B aktif hale geçer. Böylece sitozoldeki aktif NF-k. B, nükleus porundan içine geçerek DNA’da uygun bölgeye bağlanıp hedef gen ekspresyonunu düzenler. •

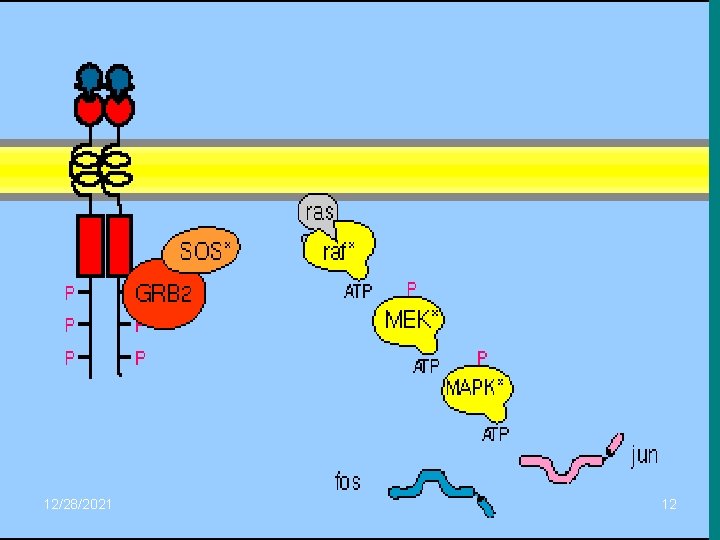
• Daha sonra Grb 2, SOS (GEF=Guanin nucleotid exchange factor) ile birleşerek, SOS (GEF)’un plazma membranında Ras proteini ile ilişki kurabileceği yere yerleşmesini sağlar. Ras proteini COOH ucuyla lipidlere bağlanarak iç tabakasına tutnur. SOS (GEF)’un etkisiyle inaktif ras proteini, GDP yerine GTP bağlayarak ve sistemdeki raf protein kinazla birleşir. Raf protein kinaz da, bir çeşit protein kinaz olan MEK kinazı fosforlayarak aktifleştirir. Aktif MEK kinaz ise, ERK’nin hem tirozin hem de treonin rezidülerini fosforlayarak onu aktive eder. ERK, aktive olur olmaz bir taraftan hücredeki protein kinazları aktive eder, bir taraftan da nükleus içine geçerek transkripsiyon faktörlerini fosforlayarak aktive eder. ERK, aktive olur olmaz bir taraftan hücredeki protein kinazları aktive eder, bir taraftan da nükleus içine geçerek transkripsiyon faktörlerini uyarır. Transkripsiyon sonucu oluşan m. RNA’lar nükleus ve sitoplazma için gerekli proteinlerin sentezlenmesinde kalıp görevi yaparlar.
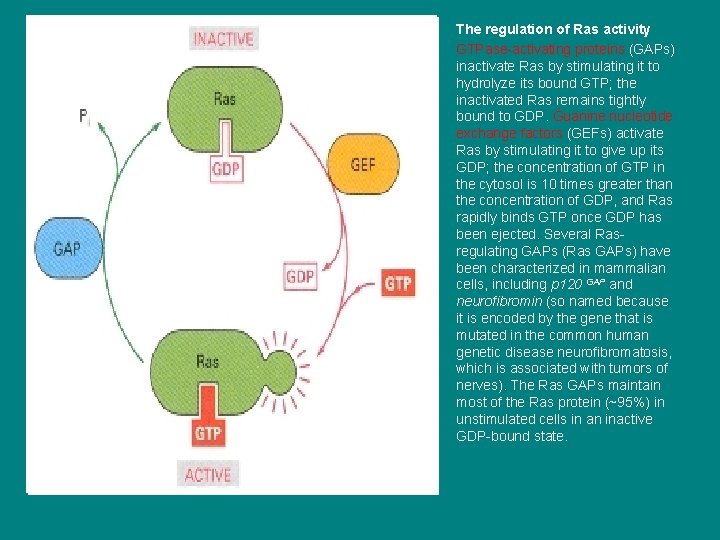
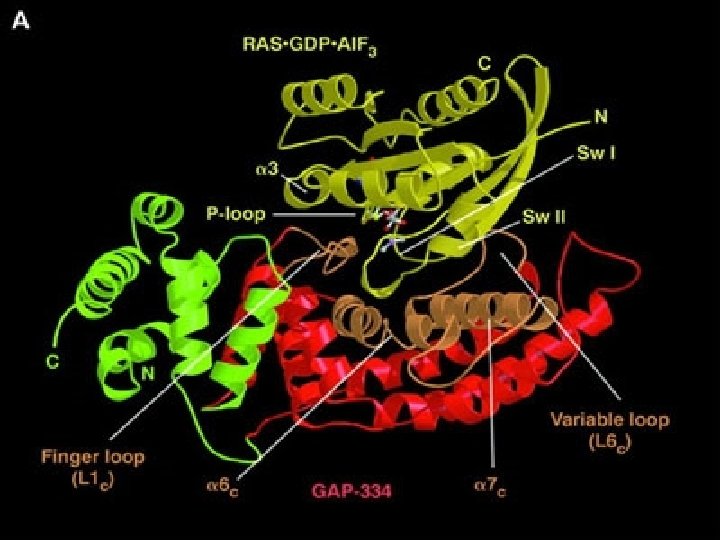
• • The regulation of Ras activity GTPase-activating proteins (GAPs) inactivate Ras by stimulating it to hydrolyze its bound GTP; the inactivated Ras remains tightly bound to GDP. Guanine nucleotide exchange factors (GEFs) activate Ras by stimulating it to give up its GDP; the concentration of GTP in the cytosol is 10 times greater than the concentration of GDP, and Ras rapidly binds GTP once GDP has been ejected. Several Rasregulating GAPs (Ras GAPs) have been characterized in mammalian cells, including p 120 GAP and neurofibromin (so named because it is encoded by the gene that is mutated in the common human genetic disease neurofibromatosis, which is associated with tumors of nerves). The Ras GAPs maintain most of the Ras protein (~95%) in unstimulated cells in an inactive GDP-bound state.
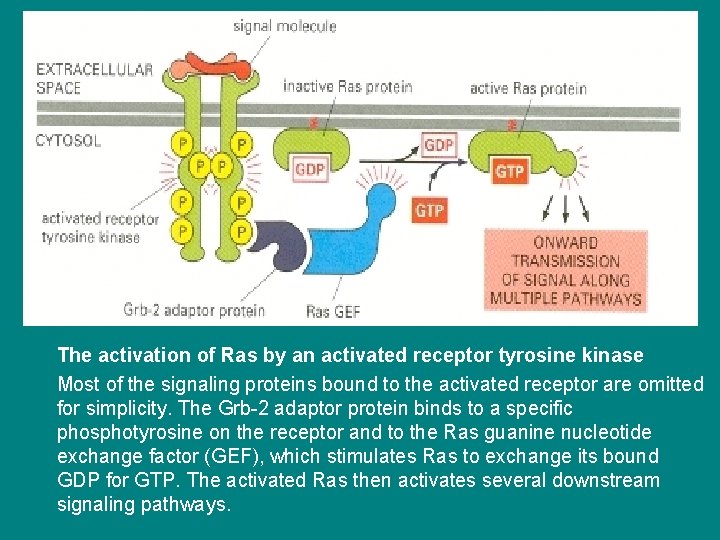
The activation of Ras by an activated receptor tyrosine kinase Most of the signaling proteins bound to the activated receptor are omitted for simplicity. The Grb-2 adaptor protein binds to a specific phosphotyrosine on the receptor and to the Ras guanine nucleotide exchange factor (GEF), which stimulates Ras to exchange its bound GDP for GTP. The activated Ras then activates several downstream signaling pathways.
12/28/2021 11
12/28/2021 12
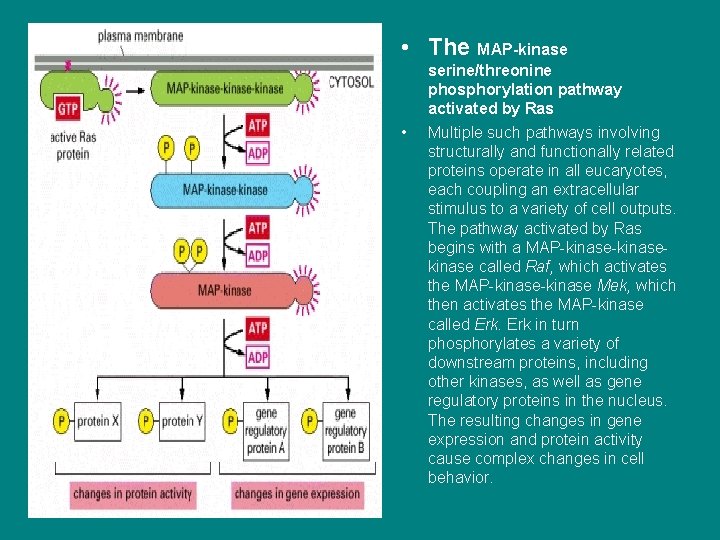
nn • The MAP-kinase • serine/threonine phosphorylation pathway activated by Ras Multiple such pathways involving structurally and functionally related proteins operate in all eucaryotes, each coupling an extracellular stimulus to a variety of cell outputs. The pathway activated by Ras begins with a MAP-kinasekinase called Raf, which activates the MAP-kinase Mek, which then activates the MAP-kinase called Erk in turn phosphorylates a variety of downstream proteins, including other kinases, as well as gene regulatory proteins in the nucleus. The resulting changes in gene expression and protein activity cause complex changes in cell behavior.
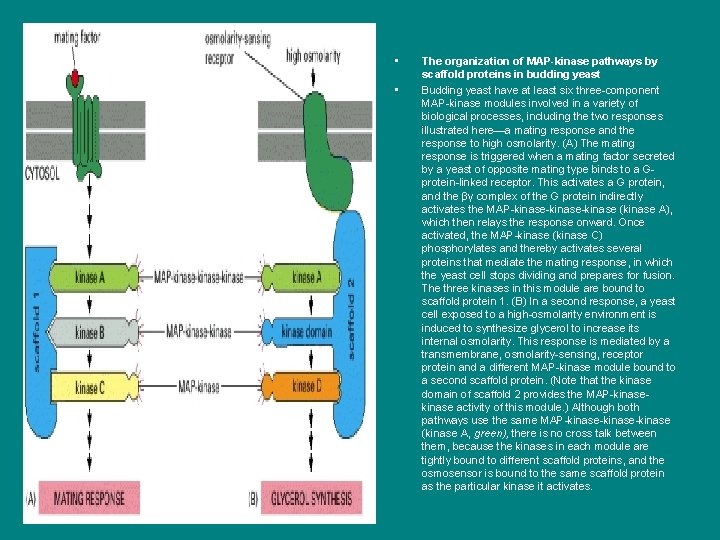
ii • • The organization of MAP-kinase pathways by scaffold proteins in budding yeast Budding yeast have at least six three-component MAP-kinase modules involved in a variety of biological processes, including the two responses illustrated here—a mating response and the response to high osmolarity. (A) The mating response is triggered when a mating factor secreted by a yeast of opposite mating type binds to a Gprotein-linked receptor. This activates a G protein, and the βγ complex of the G protein indirectly activates the MAP-kinase-kinase (kinase A), which then relays the response onward. Once activated, the MAP-kinase (kinase C) phosphorylates and thereby activates several proteins that mediate the mating response, in which the yeast cell stops dividing and prepares for fusion. The three kinases in this module are bound to scaffold protein 1. (B) In a second response, a yeast cell exposed to a high-osmolarity environment is induced to synthesize glycerol to increase its internal osmolarity. This response is mediated by a transmembrane, osmolarity-sensing, receptor protein and a different MAP-kinase module bound to a second scaffold protein. (Note that the kinase domain of scaffold 2 provides the MAP-kinase activity of this module. ) Although both pathways use the same MAP-kinase-kinase (kinase A, green), there is no cross talk between them, because the kinases in each module are tightly bound to different scaffold proteins, and the osmosensor is bound to the same scaffold protein as the particular kinase it activates.
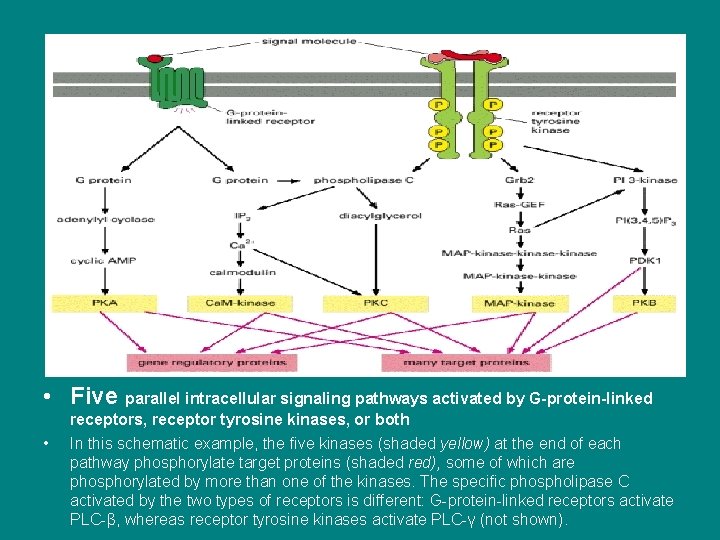
• Five parallel intracellular signaling pathways activated by G-protein-linked • receptors, receptor tyrosine kinases, or both In this schematic example, the five kinases (shaded yellow) at the end of each pathway phosphorylate target proteins (shaded red), some of which are phosphorylated by more than one of the kinases. The specific phospholipase C activated by the two types of receptors is different: G-protein-linked receptors activate PLC-β, whereas receptor tyrosine kinases activate PLC-γ (not shown).
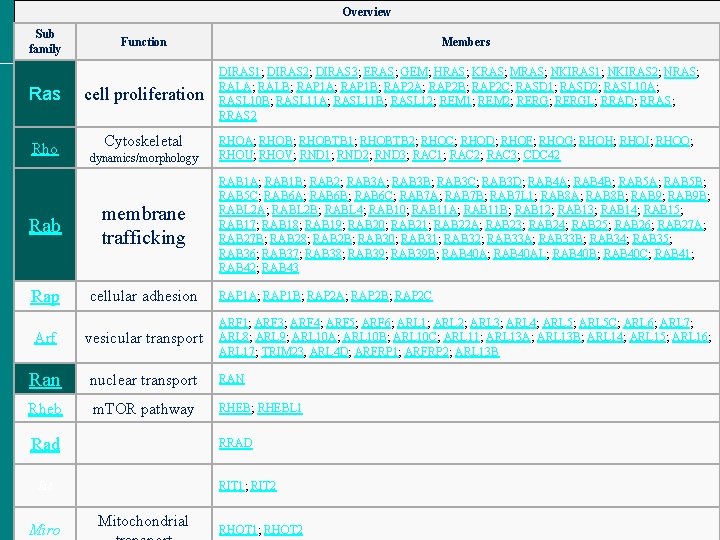
Overview Sub family Function Ras cell proliferation Rho Cytoskeletal dynamics/morphology Rab membrane trafficking Rap cellular adhesion Members DIRAS 1; DIRAS 2; DIRAS 3; ERAS; GEM; HRAS; KRAS; MRAS; NKIRAS 1; NKIRAS 2; NRAS; RALA; RALB; RAP 1 A; RAP 1 B; RAP 2 A; RAP 2 B; RAP 2 C; RASD 1; RASD 2; RASL 10 A; RASL 10 B; RASL 11 A; RASL 11 B; RASL 12; REM 1; REM 2; RERGL; RRAD; RRAS 2 RHOA; RHOBTB 1; RHOBTB 2; RHOC; RHOD; RHOF; RHOG; RHOH; RHOJ; RHOQ; RHOU; RHOV; RND 1; RND 2; RND 3; RAC 1; RAC 2; RAC 3; CDC 42 RAB 1 A; RAB 1 B; RAB 2; RAB 3 A; RAB 3 B; RAB 3 C; RAB 3 D; RAB 4 A; RAB 4 B; RAB 5 A; RAB 5 B; RAB 5 C; RAB 6 A; RAB 6 B; RAB 6 C; RAB 7 A; RAB 7 B; RAB 7 L 1; RAB 8 A; RAB 8 B; RAB 9 B; RABL 2 A; RABL 2 B; RABL 4; RAB 10; RAB 11 A; RAB 11 B; RAB 12; RAB 13; RAB 14; RAB 15; RAB 17; RAB 18; RAB 19; RAB 20; RAB 21; RAB 22 A; RAB 23; RAB 24; RAB 25; RAB 26; RAB 27 A; RAB 27 B; RAB 28; RAB 2 B; RAB 30; RAB 31; RAB 32; RAB 33 A; RAB 33 B; RAB 34; RAB 35; RAB 36; RAB 37; RAB 38; RAB 39 B; RAB 40 AL; RAB 40 B; RAB 40 C; RAB 41; RAB 42; RAB 43 RAP 1 A; RAP 1 B; RAP 2 A; RAP 2 B; RAP 2 C ARF 1; ARF 3; ARF 4; ARF 5; ARF 6; ARL 1; ARL 2; ARL 3; ARL 4; ARL 5 C; ARL 6; ARL 7; ARL 8; ARL 9; ARL 10 A; ARL 10 B; ARL 10 C; ARL 11; ARL 13 A; ARL 13 B; ARL 14; ARL 15; ARL 16; ARL 17; TRIM 23, ARL 4 D; ARFRP 1; ARFRP 2; ARL 13 B Arf vesicular transport Ran nuclear transport RAN Rheb m. TOR pathway RHEB; RHEBL 1 Rad RRAD Rit Miro RIT 1; RIT 2 Mitochondrial RHOT 1; RHOT 2
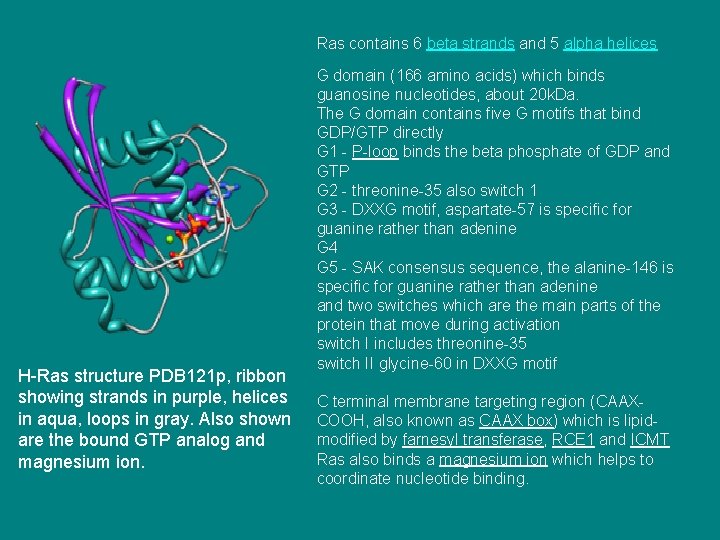
Ras contains 6 beta strands and 5 alpha helices H-Ras structure PDB 121 p, ribbon showing strands in purple, helices in aqua, loops in gray. Also shown are the bound GTP analog and magnesium ion. G domain (166 amino acids) which binds guanosine nucleotides, about 20 k. Da. The G domain contains five G motifs that bind GDP/GTP directly G 1 - P-loop binds the beta phosphate of GDP and GTP G 2 - threonine-35 also switch 1 G 3 - DXXG motif, aspartate-57 is specific for guanine rather than adenine G 4 G 5 - SAK consensus sequence, the alanine-146 is specific for guanine rather than adenine and two switches which are the main parts of the protein that move during activation switch I includes threonine-35 switch II glycine-60 in DXXG motif C terminal membrane targeting region (CAAXCOOH, also known as CAAX box) which is lipidmodified by farnesyl transferase, RCE 1 and ICMT Ras also binds a magnesium ion which helps to coordinate nucleotide binding.

İşlevi • Apopitoz • Hc içi sinyal ağlarını kontrol eden ikili anahtar. • Ras bağımlı sinyal yolları, aktin hcre iskeleti bütünlüğü, proliferasyon, farklılaşma, hc adhezyonu, apopitoz ve hücre göçü. proteins function as binary molecular switches that controlü • Ras ve ras bağımlı proteinlerin regülasyonu kanserde bozulmakta ve artan invazyon ve metastazla, azalan apopitozla ilişkilendirilmekte • Ras aktive ettiği yollar – mitogen-activated protein (MAP) kinaz kaskadı • Hc büyümesi ve bölünmesinde işlevi olan genlerin transkripsiyonuna neden olur – AKT pathway • Apopitozu inhibe eder

Membran ataçmanı • Ras’ın hc zarına ataçmanı prenilasyon ile sağlanır. is attached to the cell membrane by prenylation, and in health is a key component in many pathways which couple growth factor receptors to downstream mitogenic effectors involved in cell proliferation or differentiation. • The C-terminal Caa. X box of Ras first gets farnesylated at its Cys residue in the cytosol and then inserted into the membrane of the endoplasmatic reticulum. The Tripeptide (aa. X) is then cleaved from the C-terminus by a specific prenyl-protein specific endoprotease, the new C-terminus is then methylated by a methyltransferase. The so processed Ras is now transported to the plasma membrane. Most Ras forms are now further palmitoylated, while K-Ras with its long positively charged stretch interacts electrostaticly with the membrane.

Ras in cancer • Mutations in the Ras family of proto-oncogenes (comprising H-Ras, N-Ras and K-Ras) are very common, being found in 20% to 30% of all human tumours. It is reasonable to speculate that a pharmacological approach that curtails Ras activity may represent a possible method to inhibit certain cancer types. • Ras point mutations are the single most common abnormality of human proto-oncogenes. Ras inhibitor trans-farnesylthiosalicylic acid (FTS, Salirasib) exhibits profound anti-oncogenic effects in many cancer cell lines.

Inappropriate activation • Inappropriate activation of the gene has been shown to play a key role in signal transduction, proliferation and malignant transformation. • Mutations in a number of different genes as well as RAS itself can have this effect. Oncogenes such as p 210 BCR-ABL or the growth receptor erb. B are upstream of Ras, so if they are constitutively activated their signals will transduce through Ras. • The tumour suppressor gene NF 1 encodes a Ras-GAP – its mutation in neurofibromatosis will mean that Ras is less likely to be inactivated. Ras can also be amplified, although this only occurs occasionally in tumours. • Finally, Ras oncogenes can be activated by point mutations so that its GTPase reaction can no longer be stimulated by GAP – this increases the half life of active Ras-GTP mutants.

Constitutively active Ras • Constitutively active Ras (Ras. D) is one which contains mutations that prevent GTP hydrolysis, thus locking Ras in a permanently 'On' state. • The most common mutations are found at residue G 12 in the P-loop and the catalytic residue Q 61. • The glycine to valine mutation at residue 12 renders the GTPase domain of Ras insensitive to inactivation by GAP and thus stuck in the "on state". Ras requires a GAP for inactivation as it is a relatively poor catalyst on its own, as opposed to other G-domain-containing proteins such as the alpha subunit of heterotrimeric G proteins. • Residue 61 is responsible for stabilizing the transition state for GTP hydrolysis. Because enzyme catalysis in general is achieved by lowering the energy barrier between substrate and product, mutation of Q 61 to K necessarily reduces the rate of intrinsic Ras GTP hydrolysis to physiologically meaningless levels.
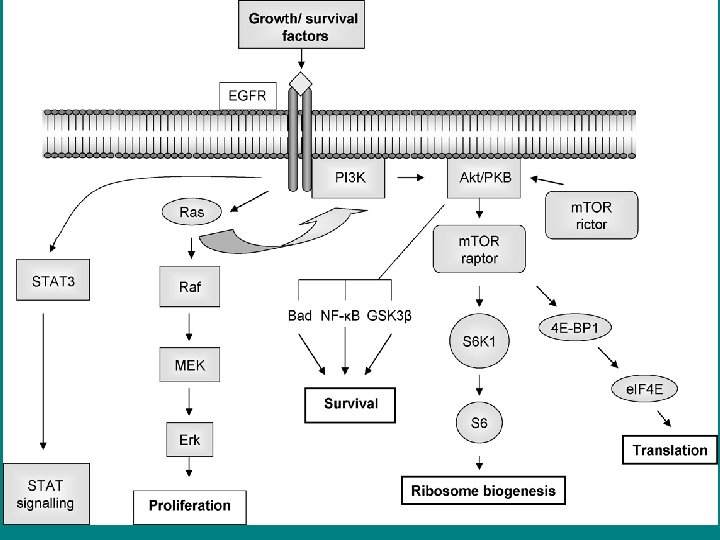
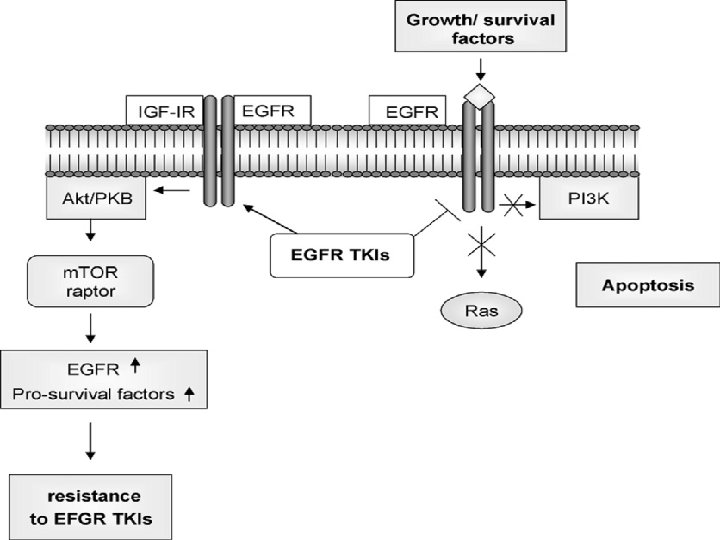
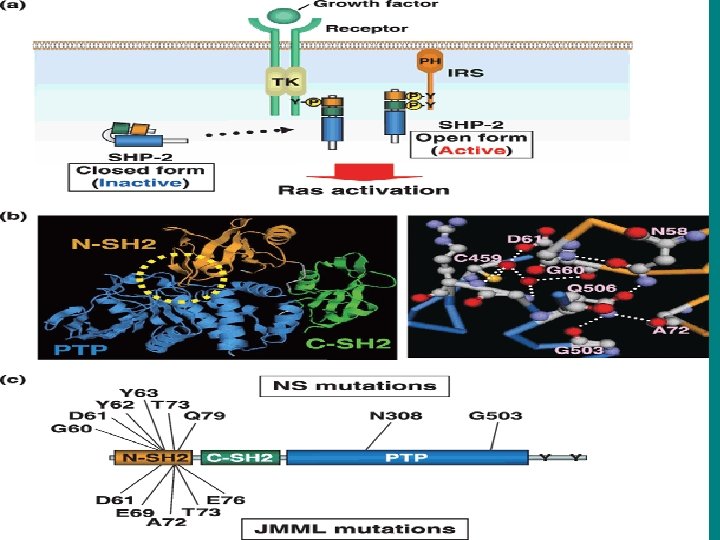
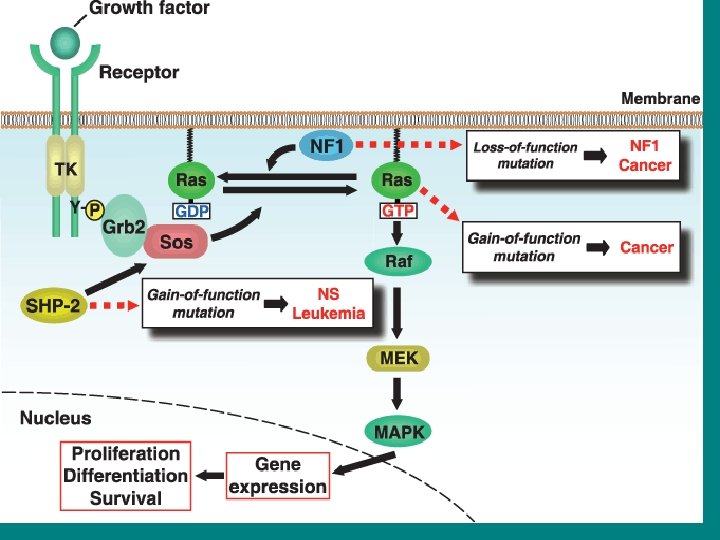
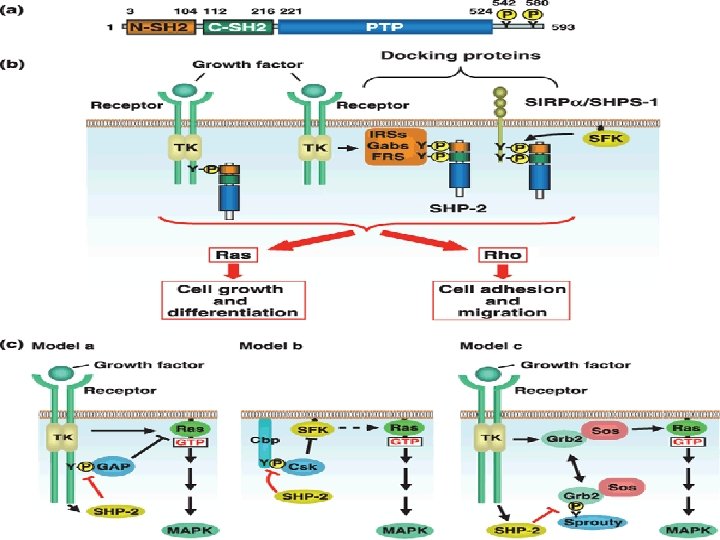
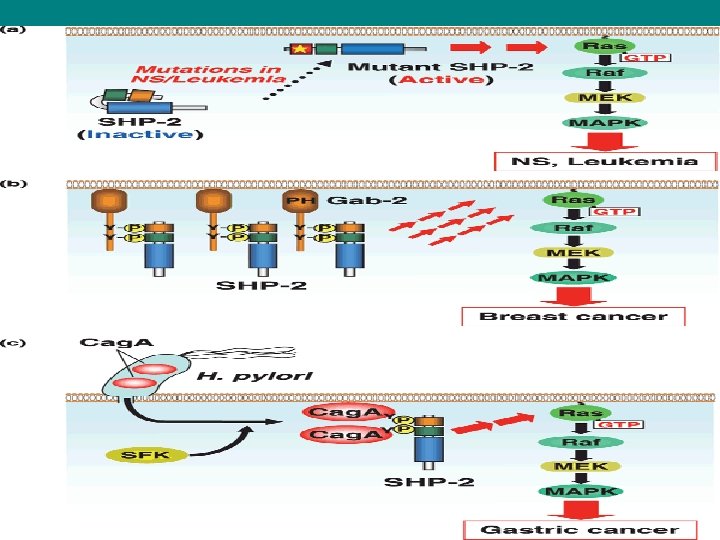
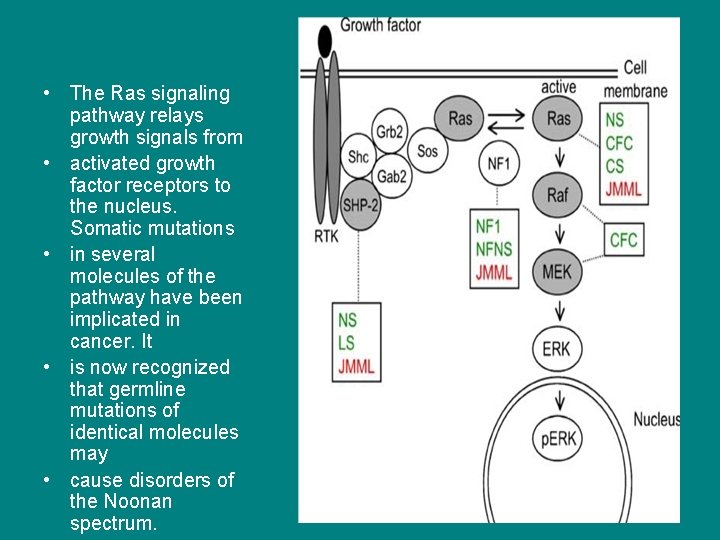
• The Ras signaling pathway relays growth signals from • activated growth factor receptors to the nucleus. Somatic mutations • in several molecules of the pathway have been implicated in cancer. It • is now recognized that germline mutations of identical molecules may • cause disorders of the Noonan spectrum.
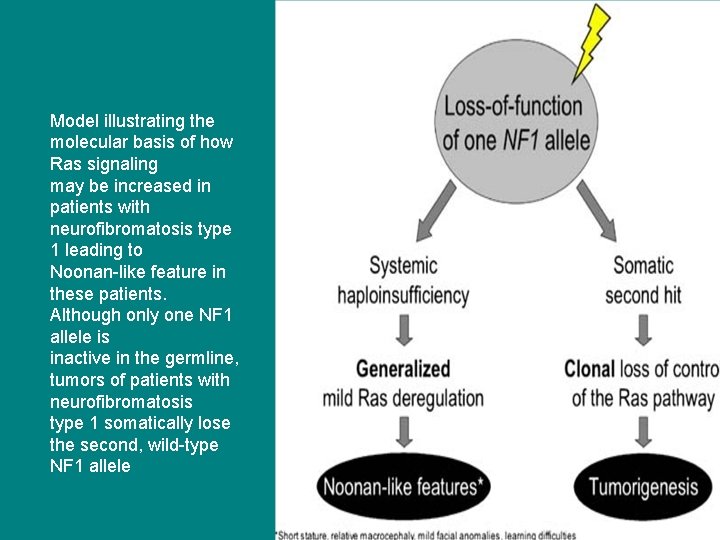
Model illustrating the molecular basis of how Ras signaling may be increased in patients with neurofibromatosis type 1 leading to Noonan-like feature in these patients. Although only one NF 1 allele is inactive in the germline, tumors of patients with neurofibromatosis type 1 somatically lose the second, wild-type NF 1 allele

TEŞEKKÜRLER
- Slides: 35