TAXUS IV Trial Slowrate release polymerbased paclitaxeleluting stent

TAXUS IV Trial Slow-rate release polymer-based paclitaxel-eluting stent compared with bare stent in patients with single de novo coronary lesions Presented at TCT 2003

TAXUS IV Trial 1, 326 patients with single de novo lesions Lesions 10 -28 mm in length and 2. 5 -3. 75 mm in diameter; Double blind, randomized trial Paclitaxel-eluting stent § Slow-release formulation § n=662 Bare Stent § n= 652 Endpoints (follow-up 1 year): g g Primary – Ischemic driven target vessel revascularization, adjudicated by CEC Secondary – MACE at 30 days and 1 year, defined as cardiac death, MI, or TVR; angiographic restenosis at 9 months; IVUS at 9 months www. Clinical trial results. org TCT 2003
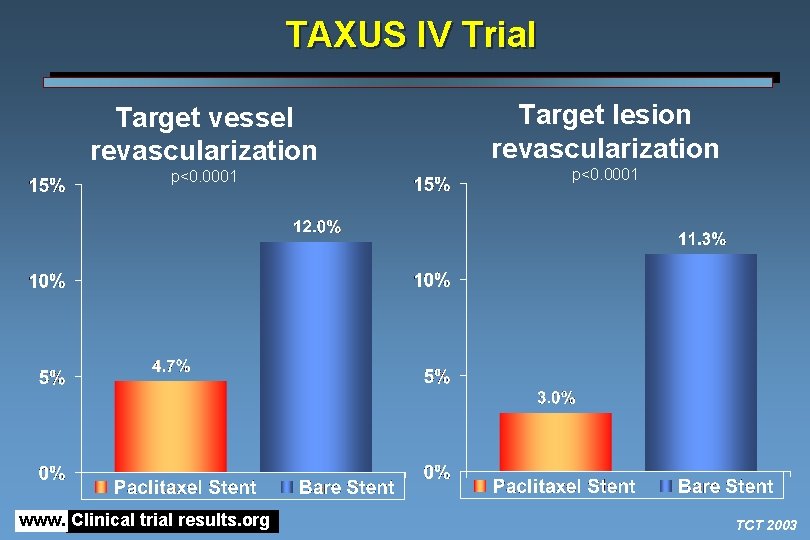
TAXUS IV Trial Target vessel revascularization p<0. 0001 www. Clinical trial results. org Target lesion revascularization p<0. 0001 TCT 2003
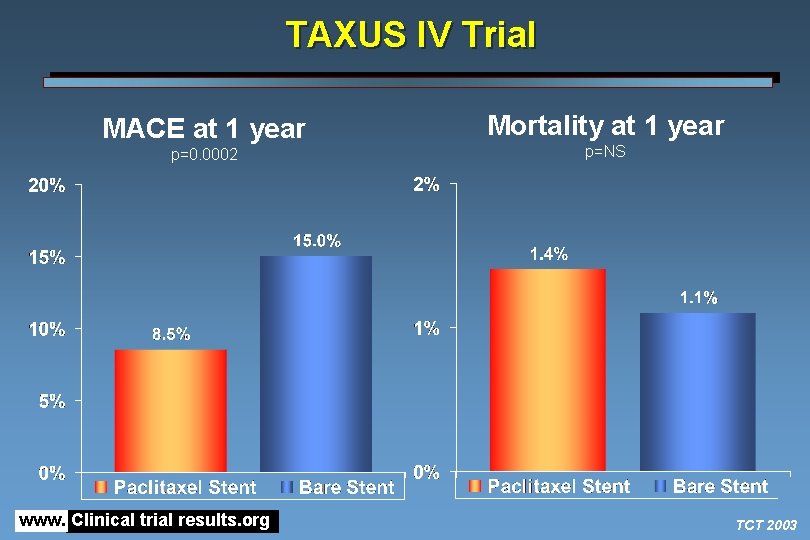
TAXUS IV Trial MACE at 1 year p=0. 0002 www. Clinical trial results. org Mortality at 1 year p=NS TCT 2003
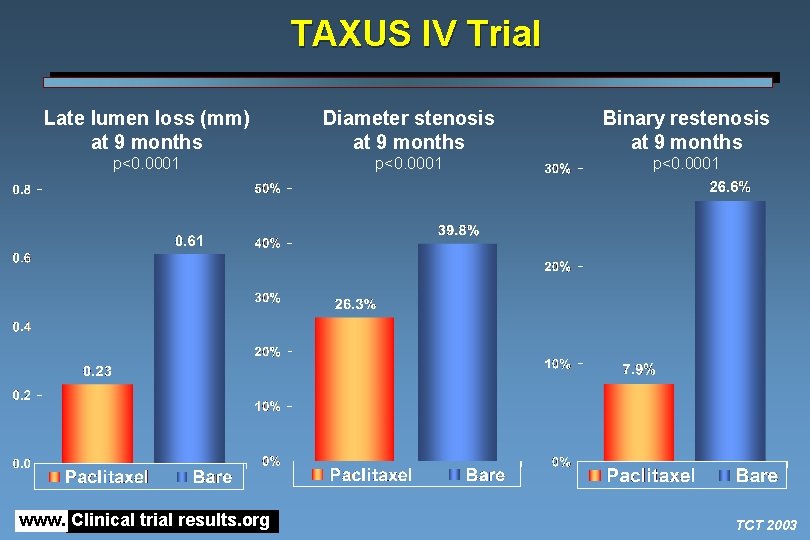
TAXUS IV Trial Late lumen loss (mm) at 9 months Diameter stenosis at 9 months Binary restenosis at 9 months p<0. 0001 www. Clinical trial results. org TCT 2003

TAXUS IV Trial • Among patients with single de novo coronary lesions, treatment with the slow-rate release polymer-based paclitaxel-eluting stent was associated with a reduction in the primary endpoint of target lesion revascularization at 1 year compared with bare stent • Results of the present trial are consistent with results from the SIRIUS trial, which compared sirolimus-eluting stents with bare stents and also showed a significant reduction in TVR with the drug-eluting stent • Neither trial has shown a reduction in the clinical endpoints of death or MI • The on-going REALITY trial is comparing the two drug-eluting stents in a head-to-head trial www. Clinical trial results. org
- Slides: 6