TAVR Unresolved Issues for 2016 Josep RodsCabau MD

TAVR: Unresolved Issues for 2016 Josep Rodés-Cabau, MD Quebec Heart & Lung Institute, Quebec City, Canada

Josep Rodés-Cabau, MD <Type of Relationship>: Research grants <Companies: Edwards Lifesciences, St Jude Medical, Medtronic
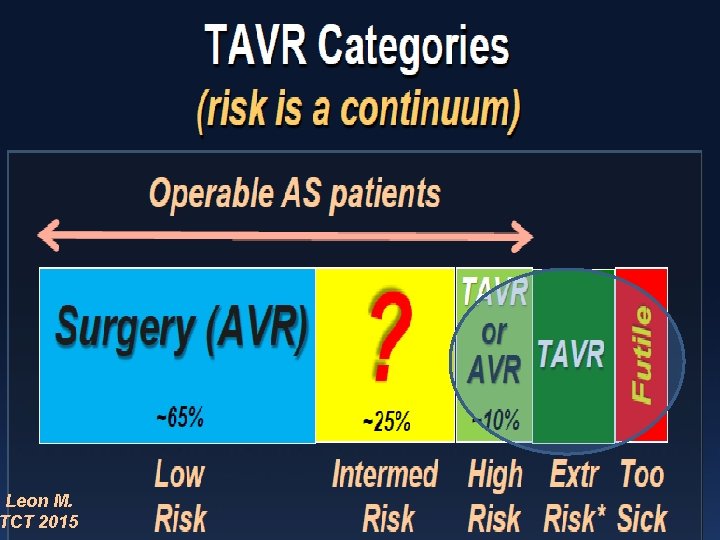
Leon M. TCT 2015
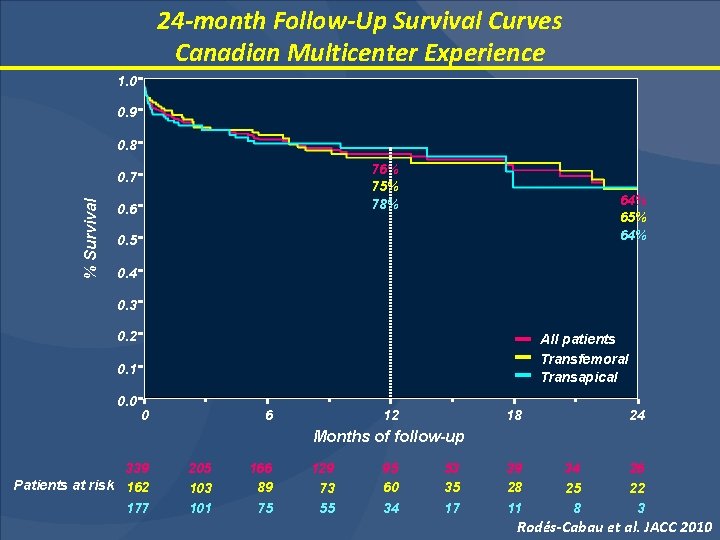
24 -month Follow-Up Survival Curves Canadian Multicenter Experience 1. 0 0. 9 0. 8 76% 75% 78% % Survival 0. 7 0. 6 64% 65% 64% 0. 5 0. 4 0. 3 0. 2 All patients Transfemoral Transapical 0. 1 0. 0 0 6 12 18 24 Months of follow-up 339 Patients at risk 162 177 205 103 101 166 89 75 129 73 55 95 60 34 53 35 17 39 28 11 34 25 8 26 22 3 Rodés-Cabau et al. JACC 2010
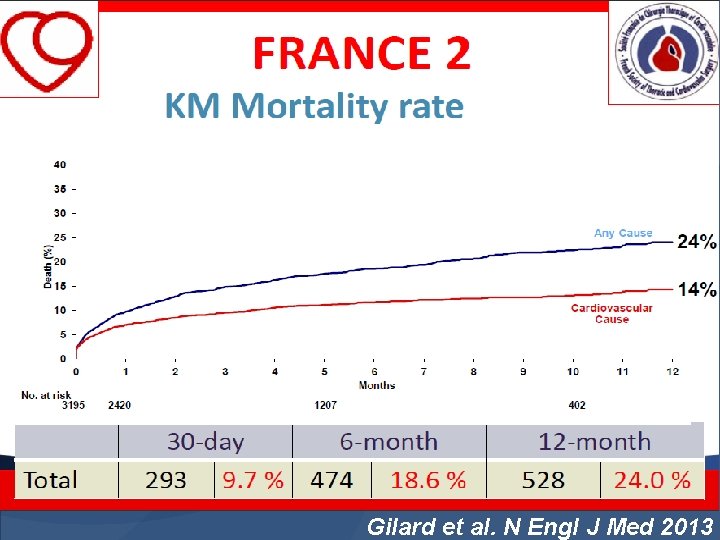
Gilard et al. N Engl J Med 2013
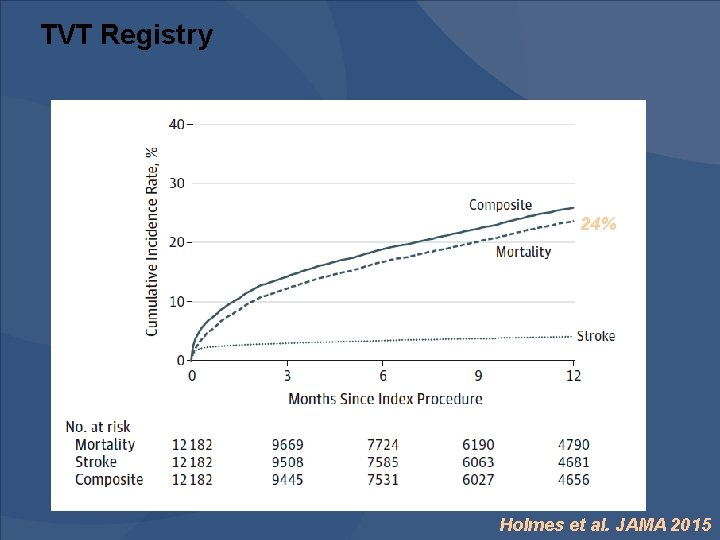
TVT Registry 24% Holmes et al. JAMA 2015
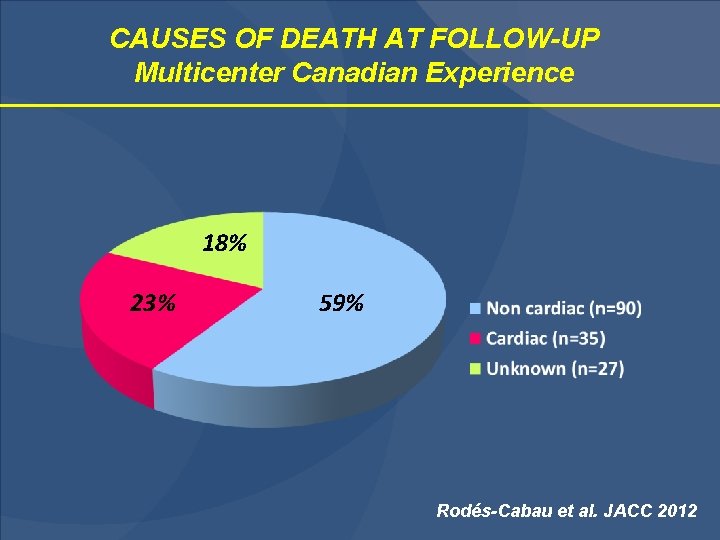
CAUSES OF DEATH AT FOLLOW-UP Multicenter Canadian Experience 18% 23% 59% Rodés-Cabau et al. JACC 2012



Leon M. TCT 2015
Results will be presented at ACC 2016 Leon M. TCT 2015

PARTNER III Trial • Prospective randomized trial: TAVR vs. SAVR in low risk patients (STS <4%) • SAPIEN 3 valve • N~1, 300 elderly (≥ 65 years) patients • 4 -D CT substudy (400 patients)
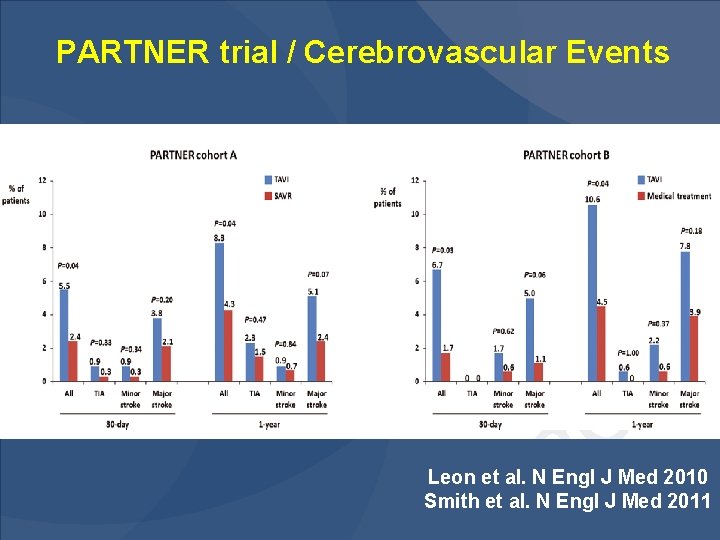
PARTNER trial / Cerebrovascular Events Leon et al. N Engl J Med 2010 Smith et al. N Engl J Med 2011
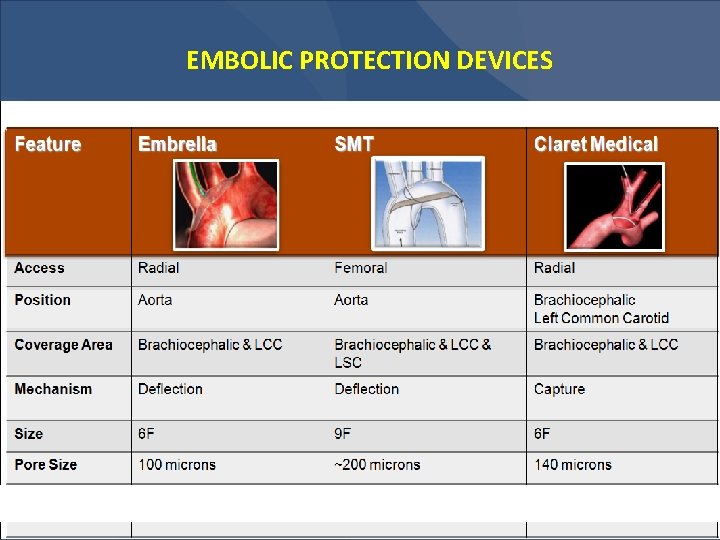
EMBOLIC PROTECTION DEVICES
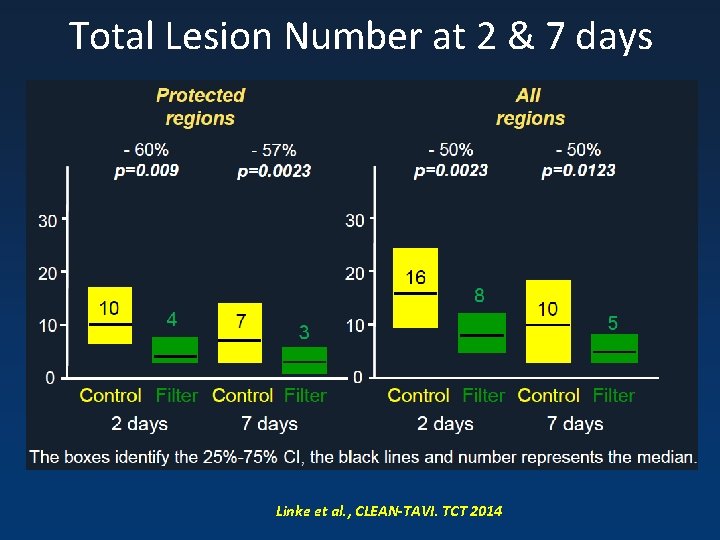
Total Lesion Number at 2 & 7 days Linke et al. , CLEAN-TAVI. TCT 2014
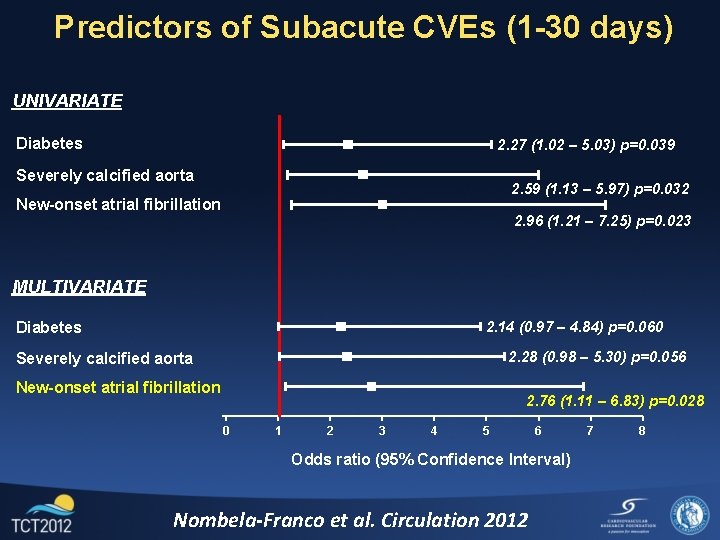
Predictors of Subacute CVEs (1 -30 days) UNIVARIATE Diabetes 2. 27 (1. 02 – 5. 03) p=0. 039 Severely calcified aorta 2. 59 (1. 13 – 5. 97) p=0. 032 New-onset atrial fibrillation 2. 96 (1. 21 – 7. 25) p=0. 023 MULTIVARIATE 2. 14 (0. 97 – 4. 84) p=0. 060 Diabetes 2. 28 (0. 98 – 5. 30) p=0. 056 Severely calcified aorta New-onset atrial fibrillation 2. 76 (1. 11 – 6. 83) p=0. 028 0 1 2 3 4 5 6 Odds ratio (95% Confidence Interval) Nombela-Franco et al. Circulation 2012 7 8

Current recommendations for antithrombotic therapy following TAVI Iung B, Rodes-Cabau J. Eur Heart J 2014
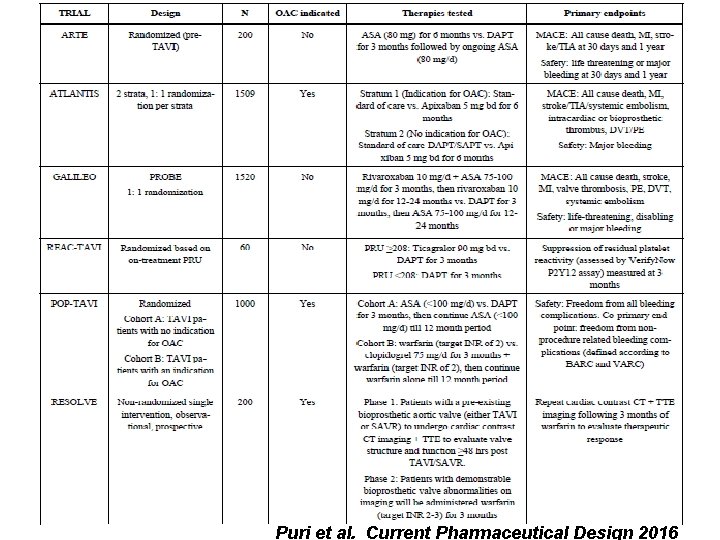
Puri et al. Current Pharmaceutical Design 2016
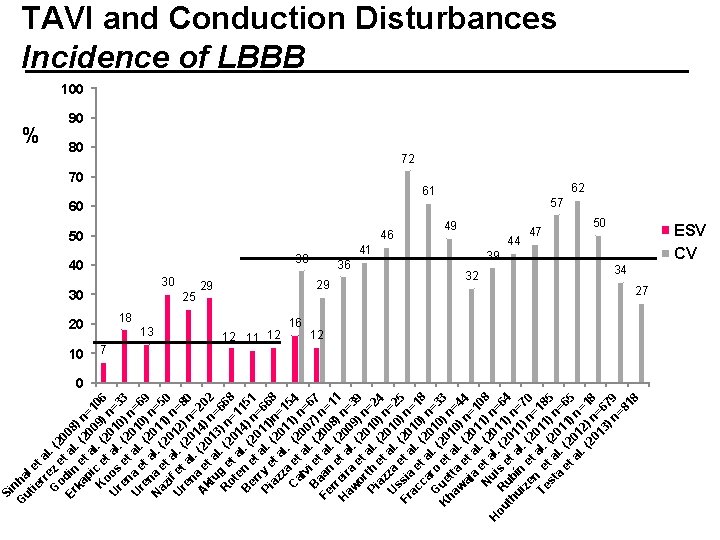
nh ut al ie et rr a G ez e l. (2 o 0 Er din t al 08 ka e. (2 ) n pi t a 00 =1 K c e l. (2 9) 06 o t U os al. 010 n=3 re e ( n t 2 ) 3 U a e al. 010 n=6 re t (2 ) 9 n N a al. 01 n=5 az et (2 1) 0 i a 0 U f et l. ( 12) n=8 re a 2 n l. 0 n= 0 A a e (2 14) 20 kt t 01 n 2 u al 3 = R g e. (2 ) n 668 ot t = e a 01 1 B n e l. ( 4) 151 er t 20 n= Pi ry e al. 11) 66 az t (2 n= 8 z al 01 1 C a e. (2 1) 54 al t a 0 n v 0 = B i e l. (2 7) 67 Fe aan t al 00 n= 8 11 r e. H reir t a (20 ) n= aw a l. 09 3 or et (20 ) n 9 Pi th al. 10 =2 az et (2 ) n 4 U za al. 010 =2 s Fr sia et a (20 ) n 5 ac e l. 10 =1 ca t a (2 ) n 8 G ro l. ( 010 =3 K ue et 20 ) n 3 ha tt a 10 = w a e l. ( ) n 44 aj t 20 = a al 1 1 N et a. (2 1) n 08 u 0 H ou R is l. (2 11 =6 e th ub t 01 ) n 4 ui ín al. 1 =7 ze e (2 ) n 0 n t = Te e al. 011 18 st t a (20 ) n 5 a l. et (2 11) =65 al 01 n=. ( 2) 1 20 n 8 13 =6 ) n 79 =8 18 G Si TAVI and Conduction Disturbances Incidence of LBBB 100 % 90 80 10 72 70 61 60 50 46 40 38 30 30 25 20 13 7 29 18 12 11 12 16 36 29 57 62 49 41 44 32 47 50 39 ESV CV 34 27 12 0
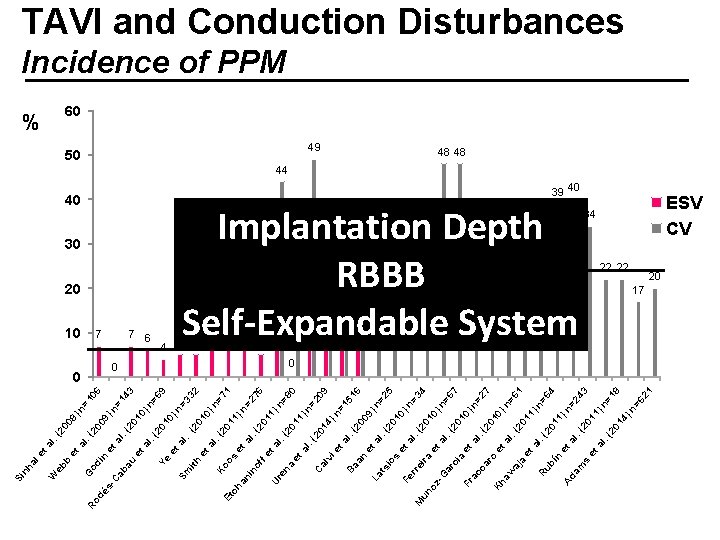
b et et al. ( 20 % 08 )n al =1. ( G Ro od 06 20 dé 09 in s)n et Ca =1 al ba. ( 43 2 u 01 et 0) al n=. ( 69 20 Ye 10 et )n al Sm =3. 32 (2 ith 0 1 et 0) al n=. Ko (2 71 01 Et os ch 1 )n et an =2 al in. 76 of (2 01 fe 1) ta Ur n= l. en ( 80 20 a 11 et )n al. ( =2 20 Ca 09 14 lv )n ie ta =1 l. 51 Ba ( 6 20 an 09 et )n La al =2. ( ts 5 20 io s 10 e )n ta Fe =3 l. rr M ei (2 4 un ra 01 oz 0) et -G n= al. ( ar 67 20 ci a 1 0) et Fr n= ac al. ( ca 27 2 ro 01 Kh 0) et aw n= al. ( 61 aj 20 a 11 et )n al. ( Ru =6 20 4 bí 11 n )n et Ad =2 al am. ( 43 20 s 11 et )n al. ( =1 20 8 14 )n =6 21 eb W al Si nh TAVI and Conduction Disturbances Incidence of PPM 60 50 10 0 49 40 30 20 7 7 6 0 10 9 4 5 5 31 21 24 18 Implantation Depth RBBB Self-Expandable System 6 5 7 48 48 44 39 40 12 6 33 26 34 30 24 21 21 16 34 28 ESV CV 22 22 17 20 12 7 0
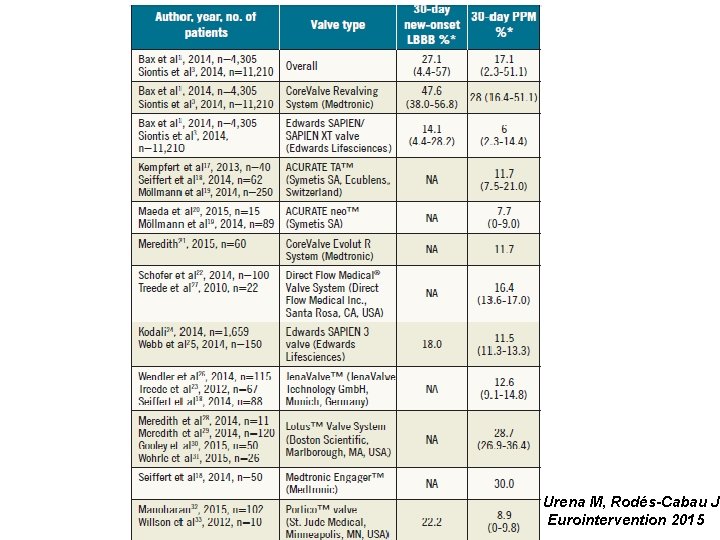
Urena M, Rodés-Cabau J Eurointervention 2015
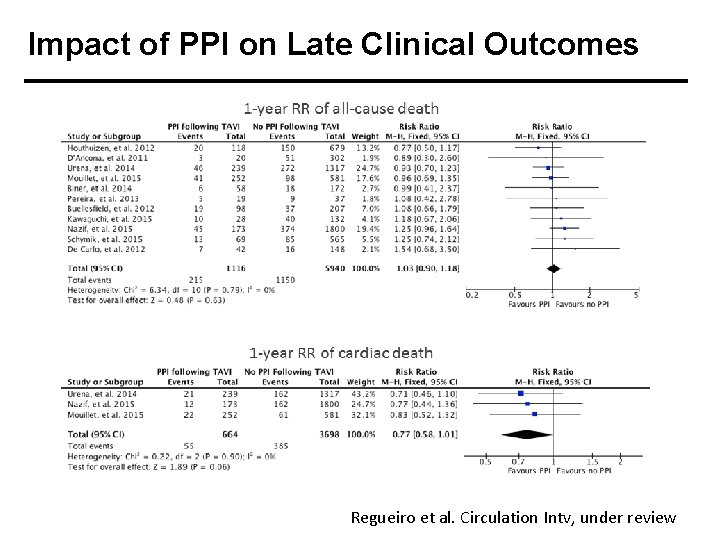
Impact of PPI on Late Clinical Outcomes Regueiro et al. Circulation Intv, under review
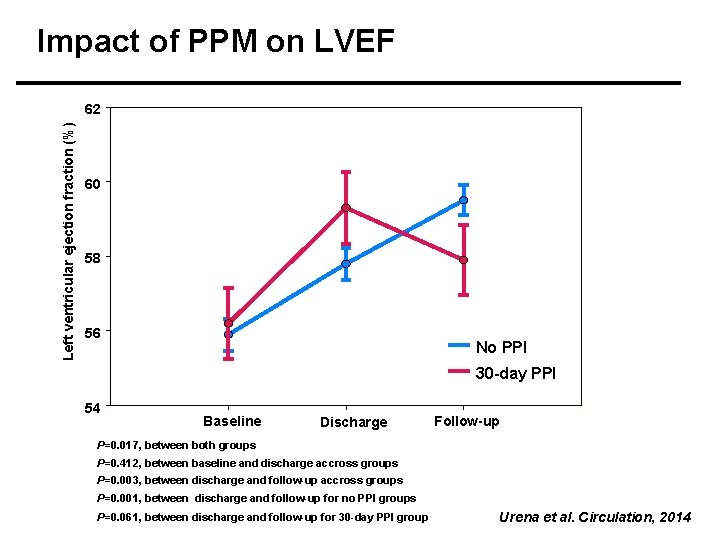
Impact of PPM on LVEF Left ventricular ejection fraction (%) 62 60 58 56 No PPI 30 -day PPI 54 Baseline Discharge Follow-up P=0. 017, between both groups P=0. 412, between baseline and discharge accross groups P=0. 003, between discharge and follow-up accross groups P=0. 001, between discharge and follow-up for no PPI groups P=0. 061, between discharge and follow-up for 30 -day PPI group Urena et al. Circulation, 2014
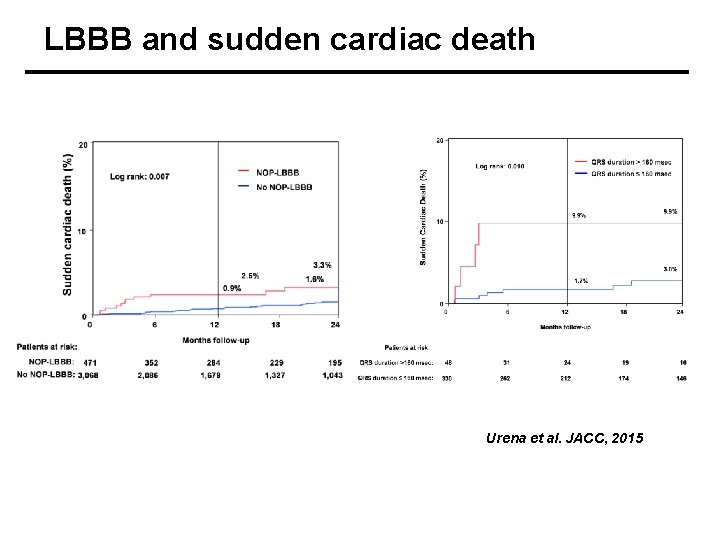
LBBB and sudden cardiac death Urena et al. JACC, 2015
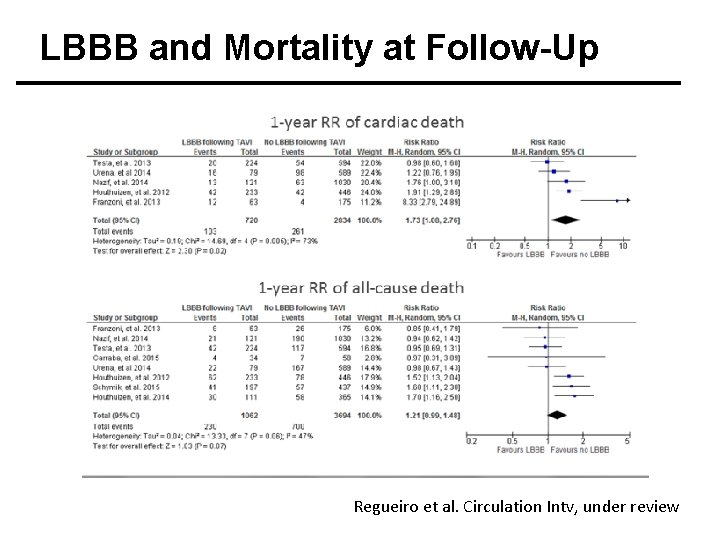
LBBB and Mortality at Follow-Up Regueiro et al. Circulation Intv, under review
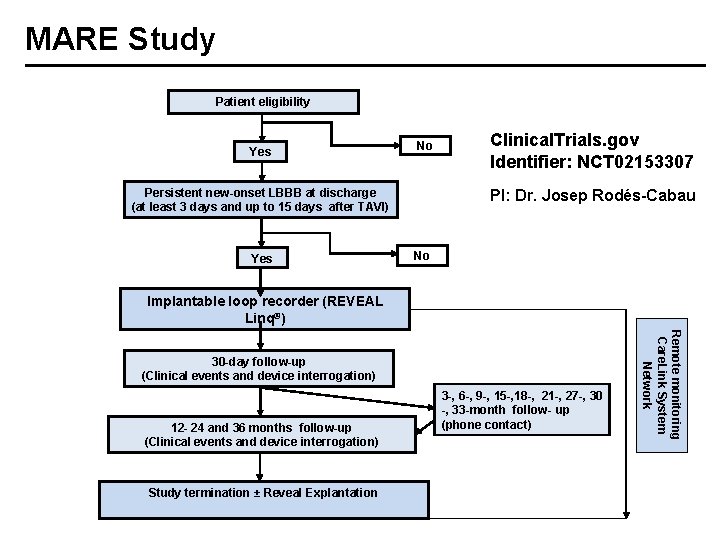
MARE Study Patient eligibility Yes No Persistent new-onset LBBB at discharge (at least 3 days and up to 15 days after TAVI) Yes Clinical. Trials. gov Identifier: NCT 02153307 PI: Dr. Josep Rodés-Cabau No Implantable loop recorder (REVEAL Linq®) 12 - 24 and 36 months follow-up (Clinical events and device interrogation) Study termination ± Reveal Explantation 3 -, 6 -, 9 -, 15 -, 18 -, 21 -, 27 -, 30 -, 33 -month follow- up (phone contact) Remote monitoring Care. Link System Network 30 -day follow-up (Clinical events and device interrogation)
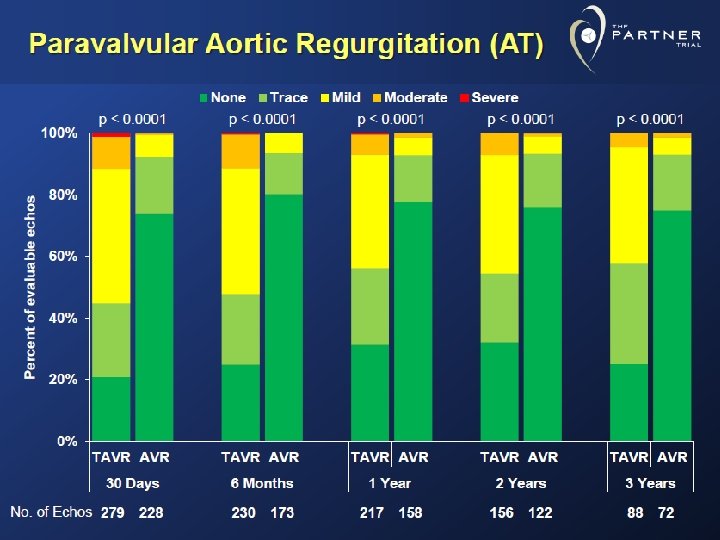
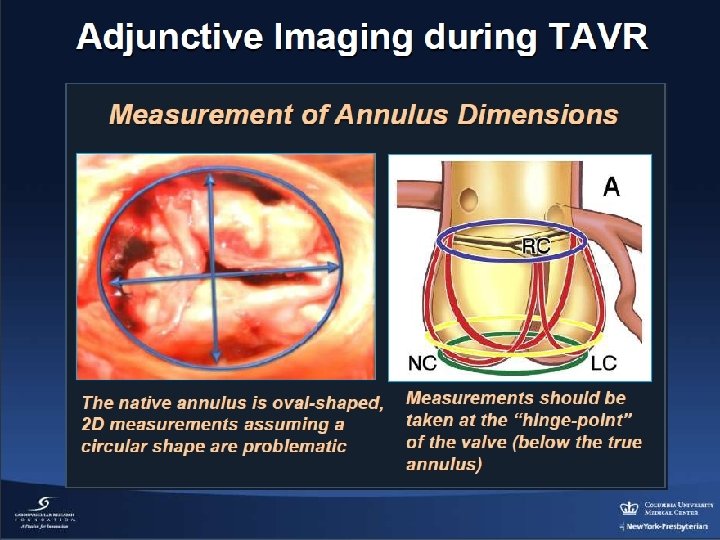
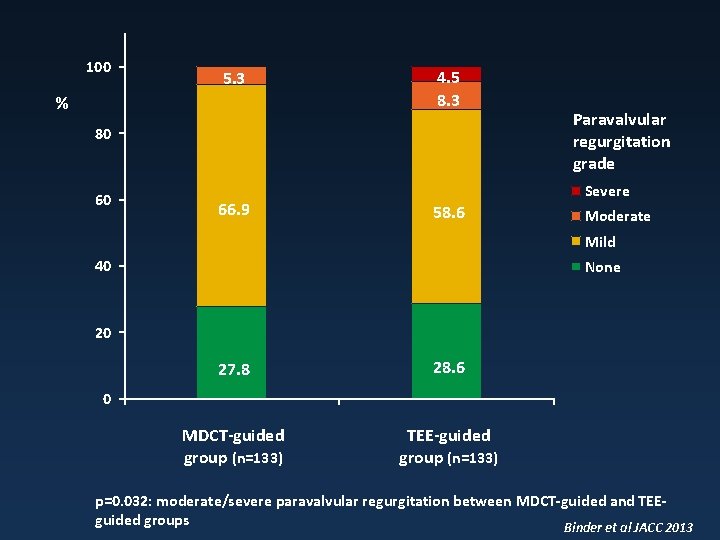
100 5. 3 % 4. 5 8. 3 80 60 66. 9 Paravalvular regurgitation grade Severe 58. 6 Moderate Mild 40 None 20 27. 8 28. 6 MDCT-guided group (n=133) TEE-guided group (n=133) 0 p=0. 032: moderate/severe paravalvular regurgitation between MDCT-guided and TEEguided groups Binder et al JACC 2013
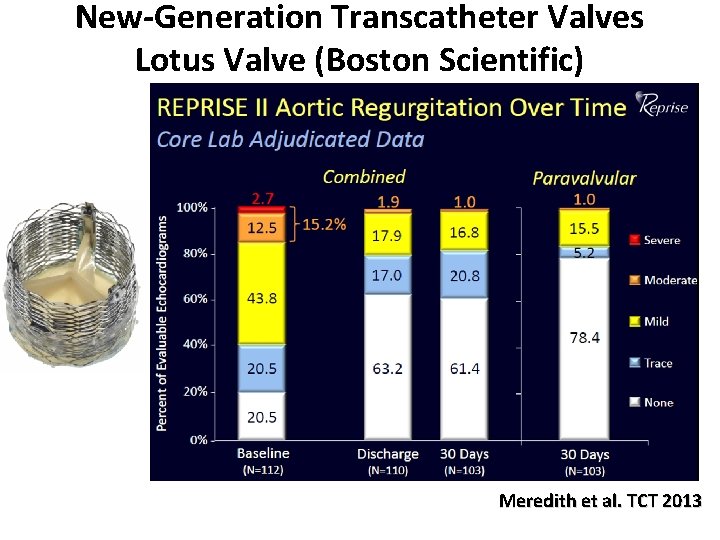
New-Generation Transcatheter Valves Lotus Valve (Boston Scientific) Meredith et al. TCT 2013
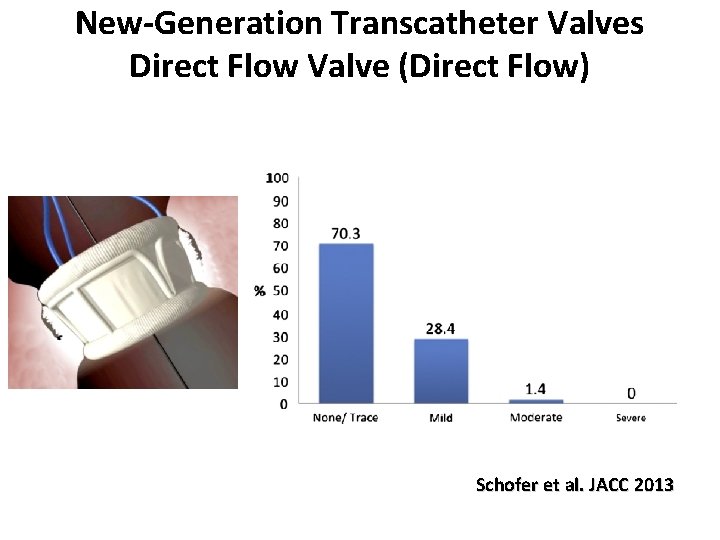
New-Generation Transcatheter Valves Direct Flow Valve (Direct Flow) Schofer et al. JACC 2013
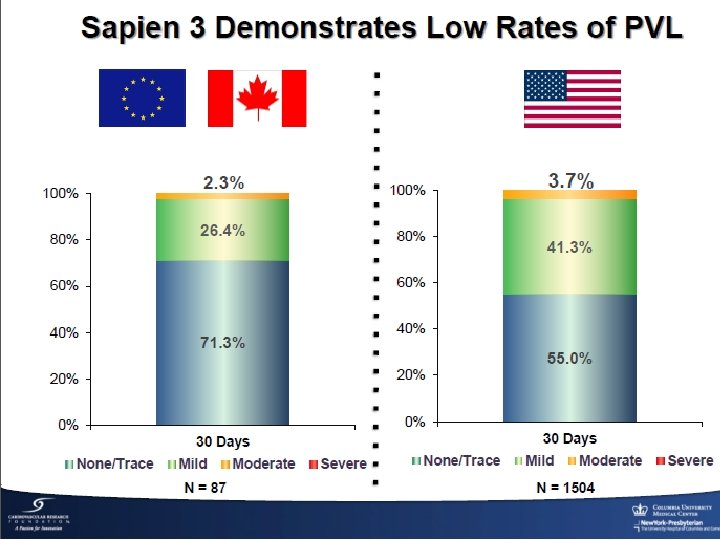
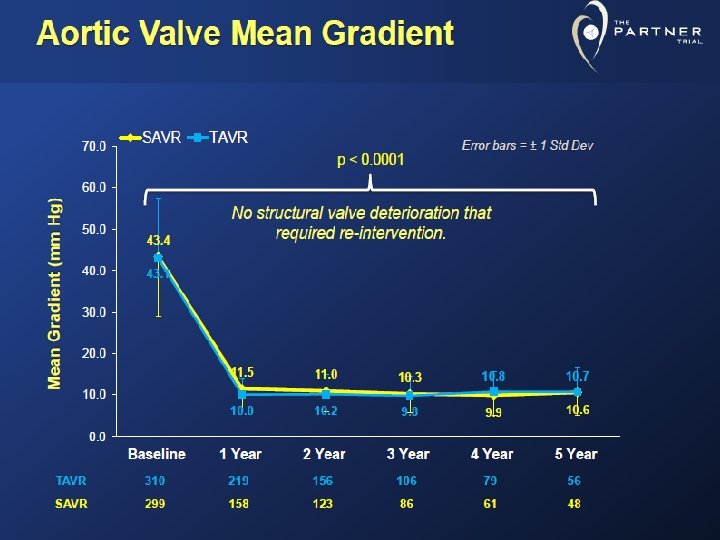
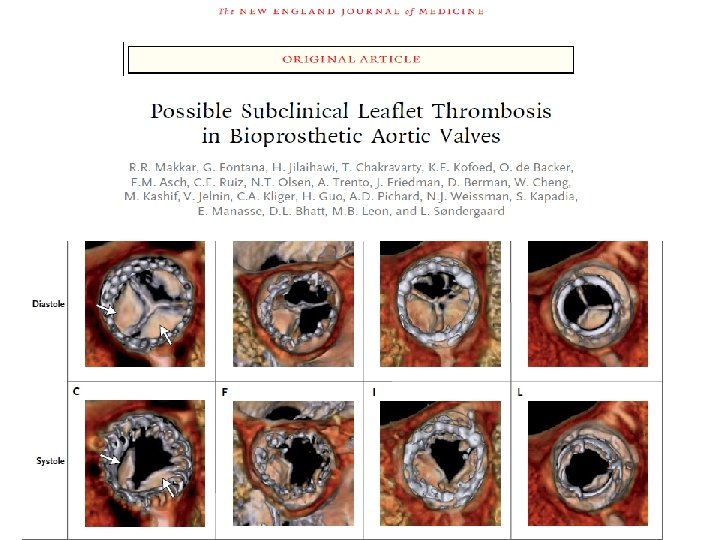
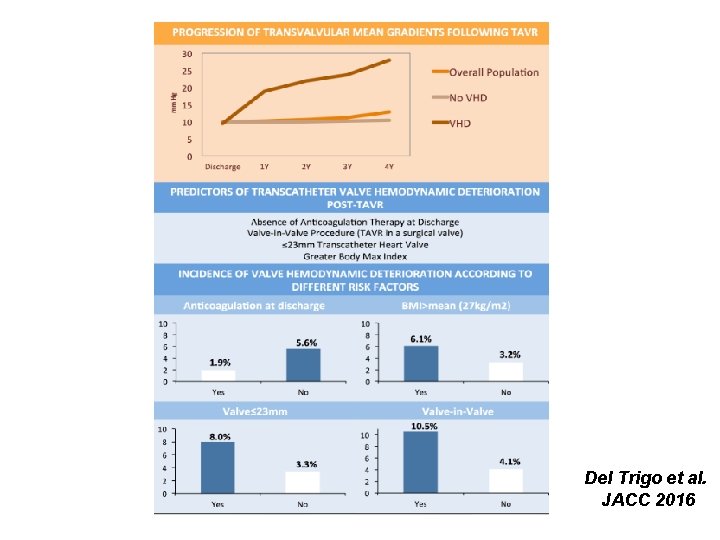
Del Trigo et al. JACC 2016

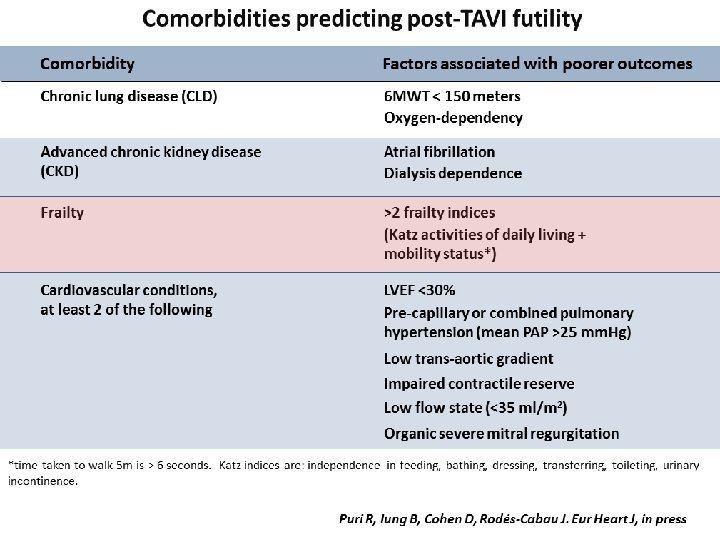
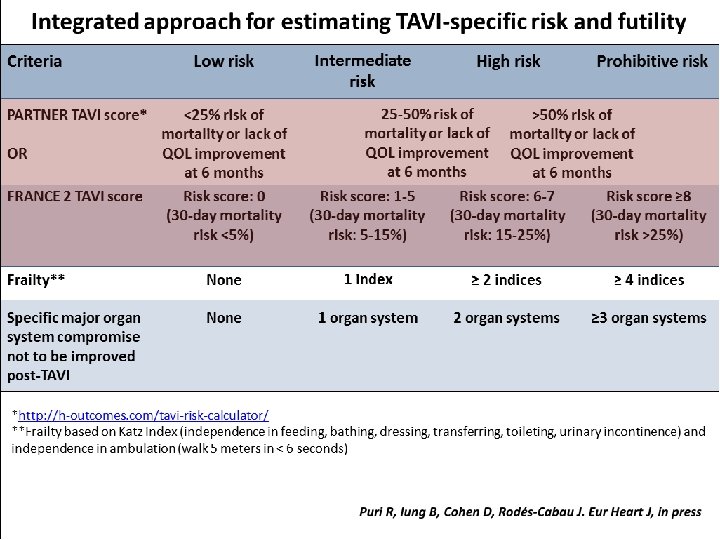
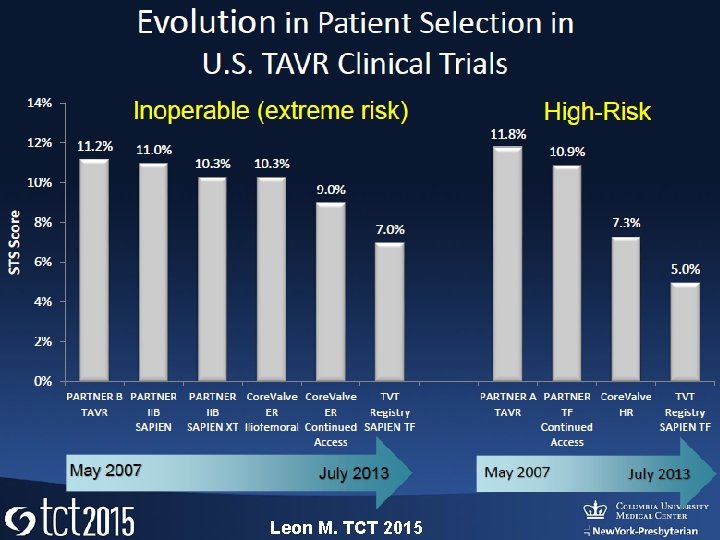
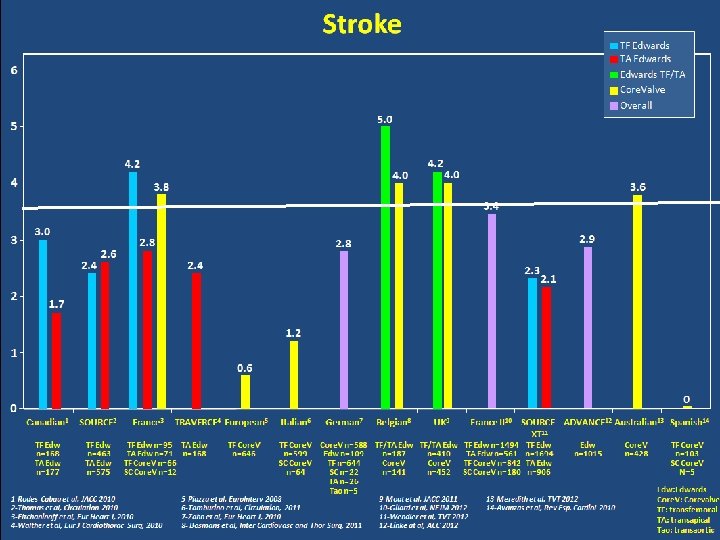
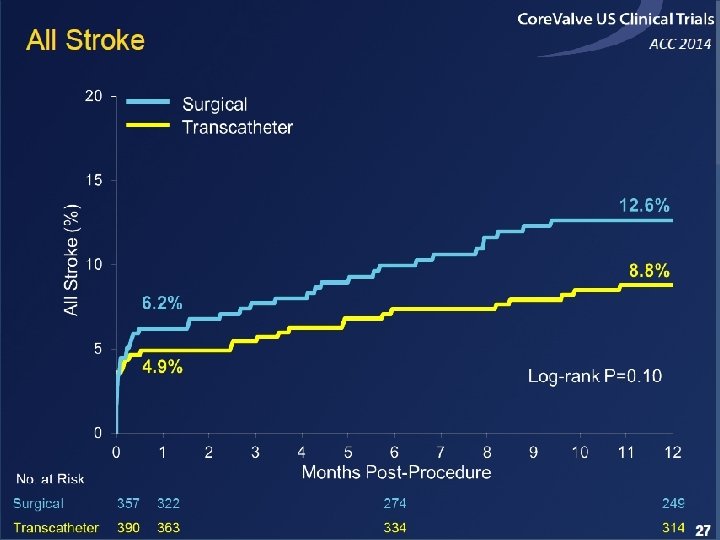
TAVR: Unresolved Issues for 2016 • Patient Selection (high-extreme risk) / TAVI risk scores / Frailty parameters • Remaining issues – Stroke: relatively high rate; role of embolic protection devices, antithrombotic treatment – Conduction disturbances: very high rate, clinical impact remain to be determined – Residual AR: better sizing, newer devices – Valve durability – thrombosis
- Slides: 41