TARGETS 1 Define and properly use the vocabulary

TARGETS 1. Define and properly use the vocabulary. 2. Describe three phases (states) of matter. 3. Identify phase and temperature changes as exothermic or endothermic. 4. Convert between the common energy units of Joules and calories. 5. Explain the relationship between temperature, energy, and heat.
THERMOCHEMISTRY Definition: Thermochemistry is the study of heat that is released or absorbed during chemical reactions, phase changes or other chemical activitiy.

THERMOCHEMISTRY LAW OF CONSERVATION OF ENERGY STATES THAT ENERGY CANNOT BE CREATED OR DESTROYED, BUT RATHER IT CHANGES FORMS. THE STUDY OF HEAT IN CHEMICAL REACTIONS APPLIES THIS LAW TO DEMONSTRATE THE CHANGE IN HEAT BEFORE AND AFTER THE REACTION.

THERMOCHEMISTRY ENERGY IN CHEMICAL EQUATIONS

SOLID, LIQUID, AND GAS PARTICLE ARRANGEMENT A substance has a particle arrangement based on the kinetic energy contained in the particles. The temperature of the substance is a measure of the kinetic energy of the particles. The particles with the least kinetic energy are in the solid phase, and the most kinetic energy particles are in the gas phase. https: //www. youtube. com/watch? v=Un. Bo. Qe 2 rsgo https: //www. youtube. com/watch? v=v 12 x. G 80 Kc. Zw

SOLID, LIQUID, AND GAS PARTICLE ARRANGEMENT § § Particle arrangement determines state of matter. State of matter is determined by temperature. Temperature is a measure of the particles’ kinetic energy. Kinetic energy determines the particle arrangement. §
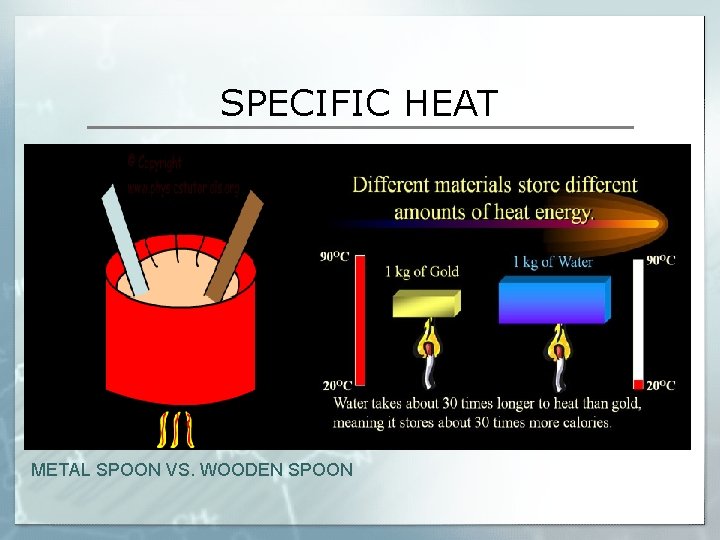
SPECIFIC HEAT METAL SPOON VS. WOODEN SPOON
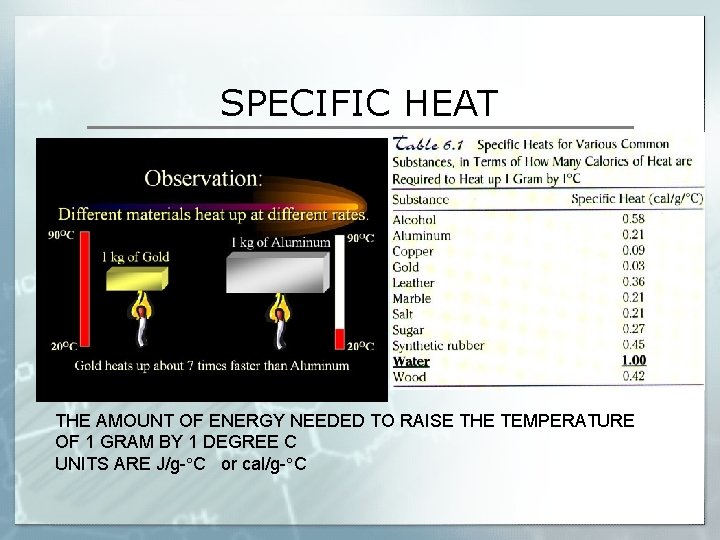
SPECIFIC HEAT THE AMOUNT OF ENERGY NEEDED TO RAISE THE TEMPERATURE OF 1 GRAM BY 1 DEGREE C UNITS ARE J/g- C or cal/g- C

SPECIFIC HEAT PROBLEMS Q=mc� T Q= heat energy m = mass of substance in grams c = specific heat constant for the substance � T = Tfinal- Tinitial How much heat energy is contained in a 100 gram sample of water at 75. 0 C? https: //www. youtube. com/watch? v=Wf 3 w. Cjpmz. H 4

SPECIFIC HEAT PROBLEM c for water = 4. 18 J/g- C Q = 100 g x 4. 18 J/g- C x 75 C Q = 31, 350 J What is the specific heat of a substance containing 300 J of energy at a temperature of 25. 5 C with a mass of 131 g? 300 J = 131 g x c x 25. 5 0. 090 J/g-C

TARGETS 6. Describe the motion and arrangement of molecules and their kinetic and potential energy on the heating/cooling curve. 7. Identify the location of each phase, phase changes and the temperature changes on a heating/cooling curve. 8. Calculate the energy requirements for the temperature changes and phase changes. 9. Identify phase and temperature changes as exothermic or endothermic.
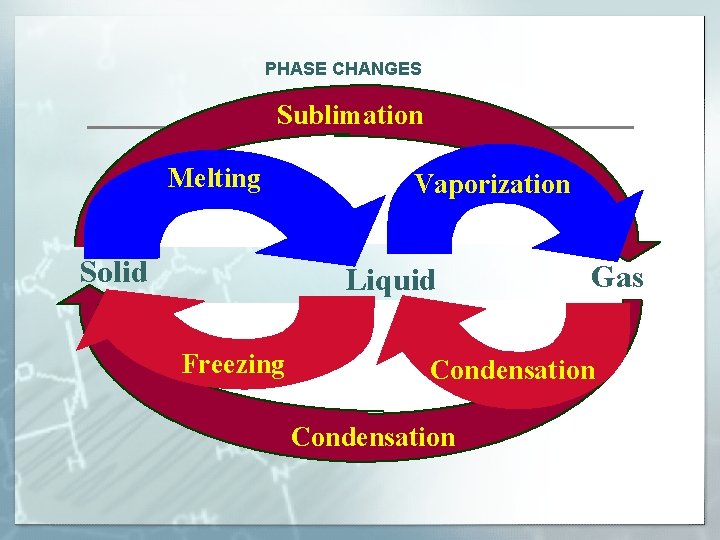
PHASE CHANGES Sublimation Melting Solid Vaporization Liquid Freezing Gas Condensation
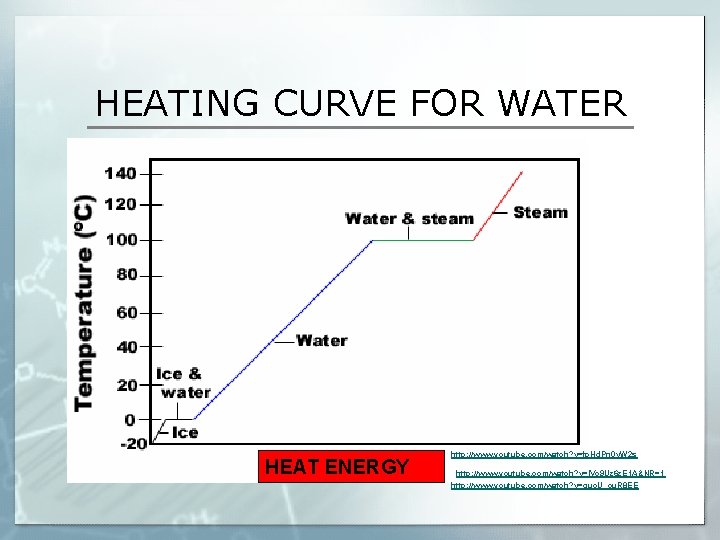
HEATING CURVE FOR WATER HEAT ENERGY http: //www. youtube. com/watch? v=fp. Hd. Pn 0 v. W 2 s http: //www. youtube. com/watch? v=IVc 9 Uz 6 z. E 1 A&NR=1 http: //www. youtube. com/watch? v=guo. U_cu. R 8 EE
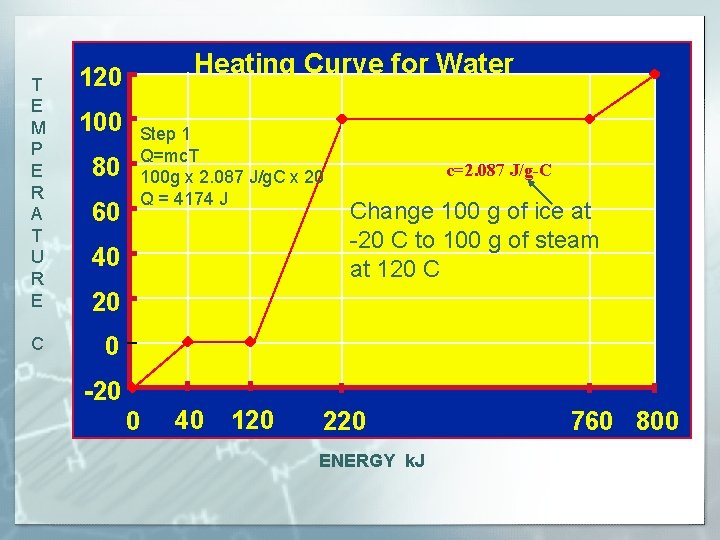
T E M P E R A T U R E C Heating Curve for Water 120 100 80 60 Steam Step 1 Q=mc. T 100 g x 2. 087 J/g. C x 20 Q = 4174 J 40 Water and Steam c=2. 087 J/g-C Change 100 g of ice at -20 C to 100 g of steam Water at 120 C 20 0 Water and Ice -20 0 40 120 220 ENERGY k. J 760 800

Heating Curve for Water 100 g 120 Steam 100 80 60 40 20 0. 333 k. J/g=Heat of Fusion Step 2 Q = Hf x m 334 J/g x 100 = 33300 J Step 3 Water Q=mc. T 100 x 4. 18 x 100 = 41800 J 0 Ice -20 0 Water and Heat of fusion is Steam the number of J per g needed to melt ice Water and Ice 40 120 220 760 800
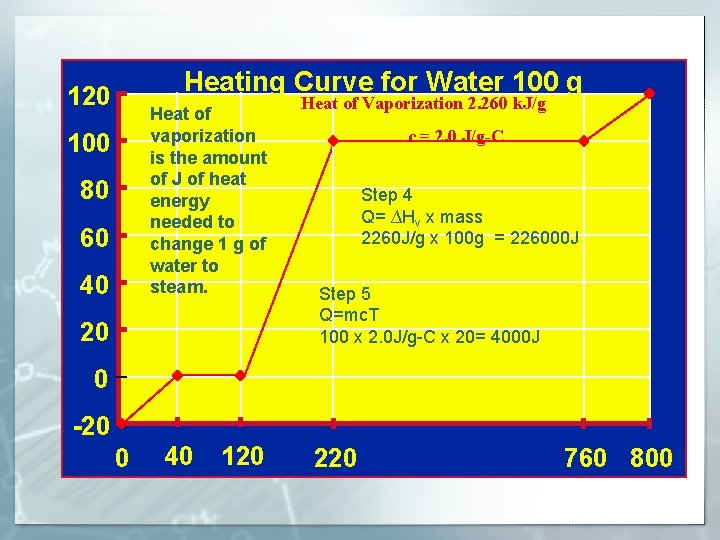
Heating Curve for Water 100 g 120 Heat of vaporization is the amount of J of heat energy needed to change 1 g of water to steam. 100 80 60 40 20 0 Heat of Vaporization 2. 260 k. J/g Steam Water and c = 2. 0 J/g-C Steam Step 4 Q= ∆Hv x mass 2260 J/g x 100 g = 226000 J Water Step 5 Q=mc. T 100 x 2. 0 J/g-C x 20= 4000 J Water and Ice -20 0 40 120 220 760 800
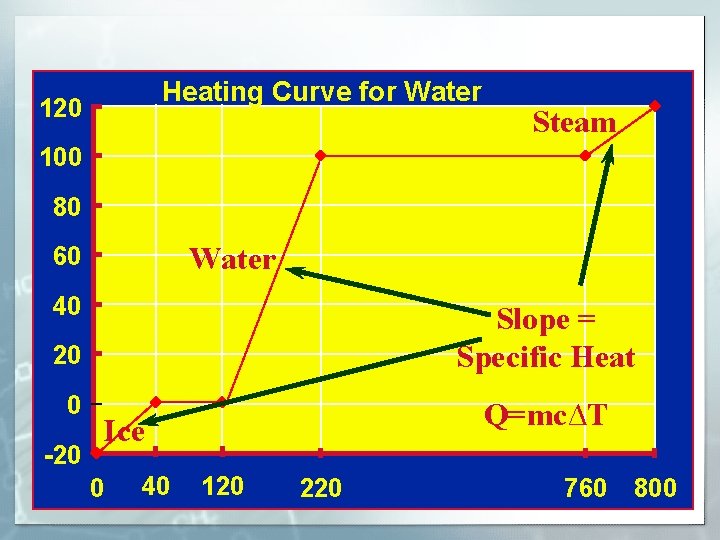
Heating Curve for Water 120 Steam Water and Steam 100 80 Water 60 Water 40 20 0 Ice -20 0 40 Water and Ice 120 220 Slope = Specific Heat Q=mc∆T 760 800
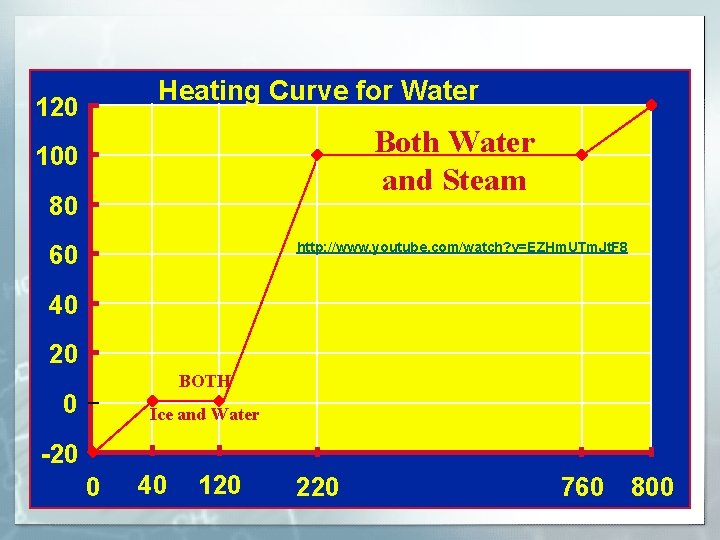
Heating Curve for Water 120 Water and Both Water Steam and Steam 100 80 Steam http: //www. youtube. com/watch? v=EZHm. UTm. Jt. F 8 60 Water 40 20 BOTH Water and Ice Ice and Water 0 -20 0 40 120 220 760 800
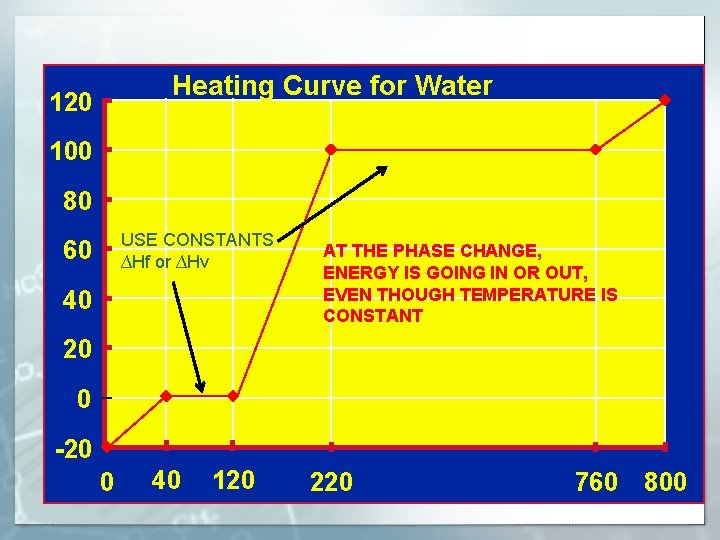
Heating Curve for Water 120 Steam Water and Steam 100 80 USE CONSTANTS ∆Hf or ∆Hv 60 AT THE PHASE CHANGE, ENERGY IS GOING IN OR OUT, Water EVEN THOUGH TEMPERATURE IS CONSTANT 40 20 0 Water and Ice -20 0 40 120 220 760 800
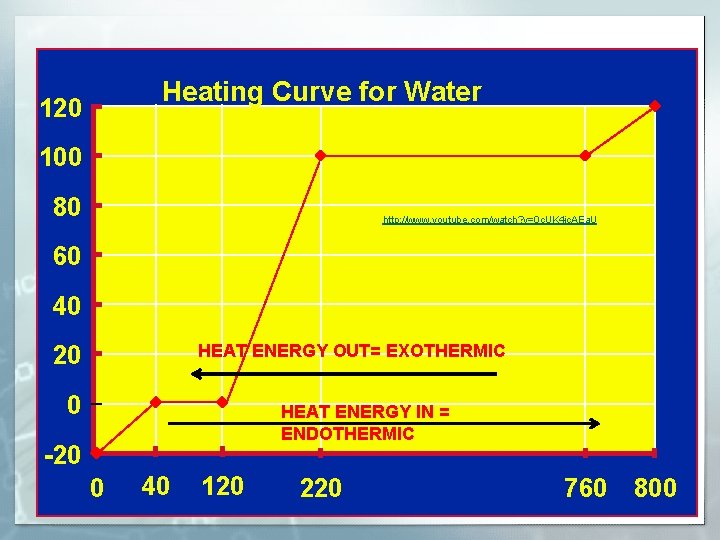
Heating Curve for Water 120 Steam Water and Steam 100 80 http: //www. youtube. com/watch? v=0 c. UK 4 jc. AEa. U 60 Water 40 HEAT ENERGY OUT= EXOTHERMIC 20 0 Water and Ice HEAT ENERGY IN = ENDOTHERMIC Ice -20 0 40 120 220 760 800

Energy and Phase Change § Heat of vaporization energy required to change one gram of a substance from liquid to gas. For water 540 cal/g OR 2. 260 k. J/g, 2260 J/g Heat of fusion energy required to change one gram of a substance from solid to liquid. For water 80 cal/g or 334 J/g

How to do it § The total heat = the sum of all the heats you have to use to go from first temperature to last temperature. Heat Ice Melt Ice 0 °C At 0 °C + Below + Heat Water Boil Water 100 °C At 100 °C + 0 °C - + Heat Steam Above 100 °C

Calculating Energy § Three equations § Heat = mass x specific heat x DT Q= mc. DT § Heat = heat of fusion x mass Q= Hfus x mass § Heat = heat of vaporization x mass Q = Hvap x mass

CONSTANTS AND SYMBOLS c = specific heat Q = heat energy m = mass in grams cal= calories J= Joules ◦C= degree Celcius △ T = Tf-Ti (Final Tem – Initial Temp) § For ice c. = § For water c = § For steam c = § Heat of vaporization= § Heat of fusion = CALORIES 0. 50 cal/g°C 1 cal/g°C 0. 50 cal/g°C 540 cal/g 80 cal/g JOULES 2. 09 J /g x ºC. 4. 18 J/g 2. 0 J /g x ºC. 2260 J/g 334 J /g

CHANGE IN HEAT ENERGY § EXOTHERMIC: HEAT OUT, -△H = heat out of the substance § http: //www. youtube. com/watch? v=i 3 m. YWB 2 f. Np 4 § ENDOTHERMIC : HEAT IN +△H= heat into the substance § http: //www. youtube. com/watch? v=c. By 4 Q 6 A 8 Hfw

Example I § I. How much heat does it take to heat 12 g of ice at -6°C to 25°C water? § 1. solve for Q for ice changing from -6 ◦C to 0◦C Q= mc△T 12 g x 2. 09 J/g-C x (0 - (-6)=150. 48 J § 2. solve for Q = Hfus x mass (this is melting at 0) 334 J/g x 12 g = 4008 J § 3. solve for Q =mc△T from 0◦C to 25 ◦C 12 g x 4. 184 J/g-C x (25 -0) = 1255. 2 J § 4. Add all three answers to determine the total amount of heat energy needed. § 150. 48 J + 4008 J + 1255. 2 J = 5413. 68 J

EXAMPLE II II How much heat does it take to heat 35 g of ice at -10°C to steam at 100 °C? 1. solve for Q for ice changing from -10 ◦C to 0◦C Q= mc△T 2. solve for Q = Hfus x mass (this is melting at 0) 3. solve for Q =mc△T from 0◦C to 100 ◦C 4. Solve for Q= Hvap x mass (this is vaporization) 5. Add all four answers to determine the total amount of heat energy needed.

Problem Steps 1. Q=mcΔT Q = 35. 0 g x 2. 09 J/g-C x (0 - -10) = 731. 5 J 2. Q = ΔH x m Q = 334 J/g x 35 = 11690 J 3. Q=mcΔT Q = 35. 0 g x 4. 18 J/g-C x (100 -0) = 14630 J 4 Q = △H x m Q = 35 g x 2260 J/g = 79100. 03 731. 5 J + 11, 690 J + 14, 630 J + 79, 100. 03 J Total = 106151. 5 J
§ https: //www. youtube. com/watch? v=TDOj. ELi-m 8 w https: //www. youtube. com/watch? v=Ju. Wt. BR-r. DQk

HEAT OF FORMATION § § § § § Calculate ∆H for the following reaction: 8 Al(s) + 3 Fe 3 O 4(s) --> 4 Al 2 O 3(s) + 9 Fe(s) Solution ∆H for a reaction is equal to the sum of the heats of formation of the product compounds minus the sum of the heats of formation of the reactant compounds: ∆H = ∑ ∆ Hf products - ∑ ∆ Hf reactants Elements are removed because you cannot form an element. ∆H = 4 ∆Hf Al 2 O 3(s) - 3 ∆ Hf Fe 3 O 4(s) The values for ∆Hf may be found in the Heats of Formation of Compounds table. Plugging in these numbers: Al 2 O 3 =-1669. 8 k. J/mol Al 2 O 3 = -1120. 9 k. J ∆ H = 4(-1669. 8 k. J) - 3(-1120. 9 k. J) ∆ H = -3316. 5 k. J

HEAT OF FORMATION EQUATION ∆H = ∑∆Hp - ∑∆ Hr Hess’ Law: Total heat of reaction is equal to the sum of the products minus the sum of the reactants.

HEAT OF REACTIONS C 3 H 8 (g) +5 O 2 (g) 3 CO 2 (g) + 4 H 2 O (g) + 2044. 0 k. J The heat of combustion of propane is – 2044. 0 k. J/mol. HOW MUCH ENERGY IS RELEASED WHEN 350. 0 GRAMS OF PROPANE IS BURNED? Identify the givens: -2044 k. J, 350 g C 3 H 8 (g) Understand that 1 mole gives off 2044 k. J of energy. 350 g x 1 mole = 7. 95 mole 44 g Q = mass x ∆Hc Q = mole x ∆Hc 7. 95 mol x -2044 k. J/mol = -16249. 8 k. J

HEATS OF REACTION PRACTICE PROBLEM CH 4 + 2 O 2 CO 2 + 2 H 2 O + 890 k. J How much heat energy is given off when 425 grams of CH 4 are burned? The equation tells us that 890 k. J of energy are give off when 1 mole of CH 4 are burned. 1. Change grams to moles: 425 g x 1 mole/16 = 26. 56 2. Multiply moles times heat: 26. 56 x -890 k. J = -2. 364 x 104 k. J

HEATS OF REACTION PRACTICE PROBLEM CH 4 + 2 O 2 CO 2 + 2 H 2 O + 890 k. J How much heat energy is given off when 2, 500 grams of CH 4 are burned? 2, 500 g x 1 mol/16 = 156. 25 mol x -890 k. J = -1. 39063 x 105 k. J

HEATS OF REACTION 3 CO(g) + Fe 2 O 3(s) 2 Fe(s) + 3 CO 2(g) + 24. 7 k. J, How much heat is released when 235. 0 g of CO react? First: change Grams to moles of CO Look at the equation: 3 mol of CO= 24. 7 k. J (You need to divide 24. 7 by 3 mol) Second: Multiply mol CO by the k. J per mole 235 g x 1 mol/28= 8. 393 mol (-24. 7 k. J/3 mol)= -8. 233 k. J 8. 393 X -8. 233 = -69. 10

PRACTICE PROBLEMS Q=m∆H How many moles of water were frozen if 1. 334 x 106 J of energy were used? Divide J/J-mol for fusion 1. 334 x 106 J/6009 J-mol = 222 How many moles of water were changed to steam if 5. 0 x 105 J were used? Divide J/J-mol for vaporization 5. 0 x 105 J/40700 J-mol = 12. 29 mol If 2950 J were used to change liquid to solid ice, how many grams froze? Divide J/J-g 2950 J/ 334 J-g =8. 832 Quantity Heat of fusion per gram Per mole 334 J/g 6009 J/mole Heat of 2260 J/g 40700 vaporization J/mole Specific heat of 2. 087 solid H 2 O (ice) J/(g·°C) * Specific heat of 4. 184 liquid H 2 O J/(g·°C) * (water) Specific heat of 2. 000 gaseous H 2 O J/(g·°C) * (steam)

WHAT TO KNOW FOR TEST § § VOCAB WORDS HOW TO SOLVE FOR Q=mc∆T (any variable) INTERPRET A PHASE CHANGE DIAGRAM KNOW THAT DURING A PHASE CHANGE, THERE IS NO TEMPERATURE CHANGE ONLY A CHANGE IN ENERGY. § HOW TO SOLVE FOR THE TOTAL Q WHEN ICE AT ANY TEMP CHANGES TO STEAM AT ANY TEMPERATURE.

WHAT TO KNOW FOR TEST IN AN EXOTHERMIC REACTION HEAT IS GIVEN OFF, CH 4 + 2 O 2 CO 2 + 2 H 2 O + 890 k. J, ∆H= -890 k. J IN AN ENDO THERMIC REACTION HEAT IS TAKEN IN, N 2 + O 2 +180. 5 k. J 2 NO, ∆H = + 180. 5 k. J Solve for ∆ H given the heat of reaction using stoichiometry

WHAT TO KNOW FOR TEST SOLVE CALORIMETRY PROBLEMS AND IDENTIFY THE METAL. Particle arrangement and relative energy in a solid, liquid and gas. LAW OF CONSERVATION OF ENERGY Freezing point and melting point are the same temp. The amount of heat energy transferred to a substance depends on: mass, specific heat of substance, and change in temp.

PROBLEMS Q=m∆H CH 4 + 2 O 2 CO 2 + 2 H 2 O + 890 k. J, ∆H= -890 k. J § How many k. J of energy were produced if 200 g of CH 4 were burned? CHANGE 200 g TO MOLES 200 X 1/16 =12. 50 mole 12. 5 m x -890 k. J = - 11, 125 k. J How many grams CH 4 were burned is the total energy output was 9. 9 x 104 k. J? Q=m x ∆H 9. 9 x 104 k. J= m x -890 k. J/mol = 111. 235 mol 111. 325 x 16/1 = 1781. 2 g If 15. 0 moles of water were produced, how many k. J were also produced? Divide ∆H by 2 Q = 15 mol x -890 k. J/2 mol =6675 k. J

PROBLEMS N 2 + O 2 +180. 5 k. J 2 NO, ∆H = + 180. 5 k. J § HOW MUCH ENERGY IS CHANGED IF 614. 0 g OF NO are produced? § EXIT SLIP: SHOW WORK AND ANSWER.

- Slides: 42