Tannins q Definition Polyphenolic secondary metabolities with relatively
Tannins q Definition: - Polyphenolic secondary metabolities with relatively higher molecular weight (> 500) found in higher plants

q General properties: 1. Tannins are non-crystallizable compounds that form colloidal acidic solutions with distinct "puckering" taste. 2. They are capable of precipitating proteins from their respective solutions. 3. Tannins precipitate gelatin and alkaloids from their solutions. 4. They form dark blue to greenish-black soluble compounds with Fe. Cl 3. They also produce a deep red colour with K. ferricyanide and ammonia. 5. They have an astringent effect. 6. Most tannins are glycosides. 7. Most true tannins have very high molecular weights.
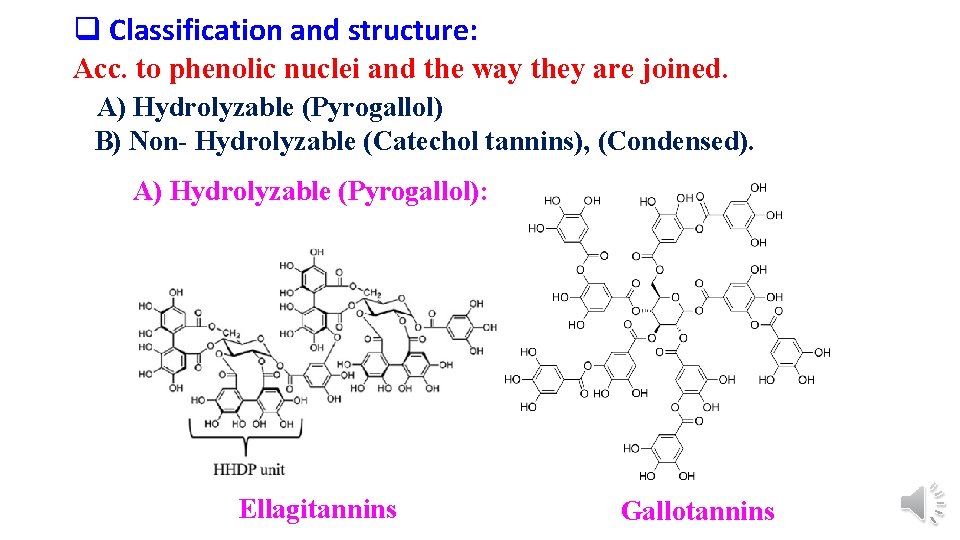
q Classification and structure: Acc. to phenolic nuclei and the way they are joined. A) Hydrolyzable (Pyrogallol) B) Non- Hydrolyzable (Catechol tannins), (Condensed). A) Hydrolyzable (Pyrogallol): Ellagitannins Gallotannins
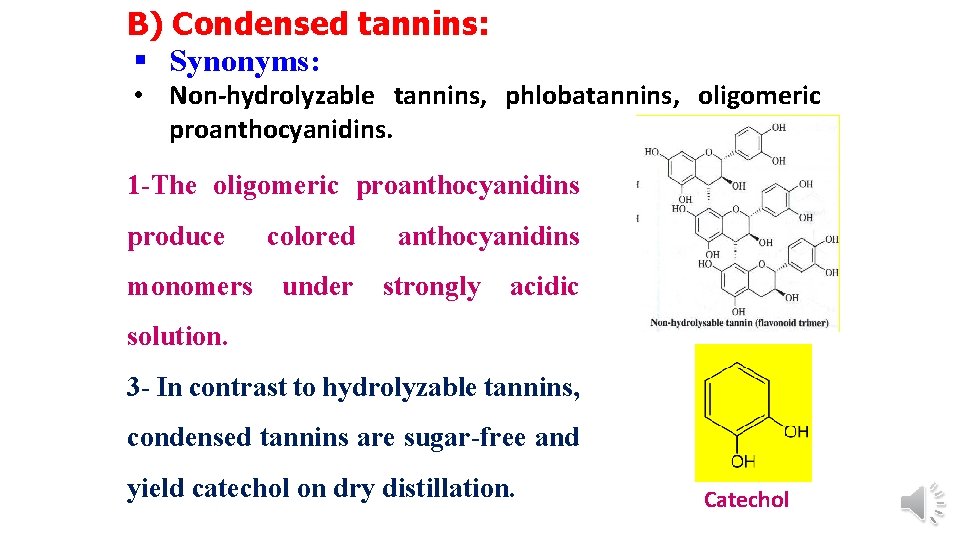
B) Condensed tannins: § Synonyms: • Non-hydrolyzable tannins, phlobatannins, oligomeric proanthocyanidins. 1 -The oligomeric proanthocyanidins produce monomers colored under anthocyanidins strongly acidic solution. 3 - In contrast to hydrolyzable tannins, condensed tannins are sugar-free and yield catechol on dry distillation. Catechol

2 - Under the effect of hydrolytic agents, they polymerize giving an insoluble, usually red-coloured product (Phlobaphenes). - Phlobaphenes (tannin red) are insoluble in most solvents and impart the characteristic red colour to many drugs e. g. red Cinchona bark. Phlobaphene is the red pigment present in the pericarp of certain maize varieties
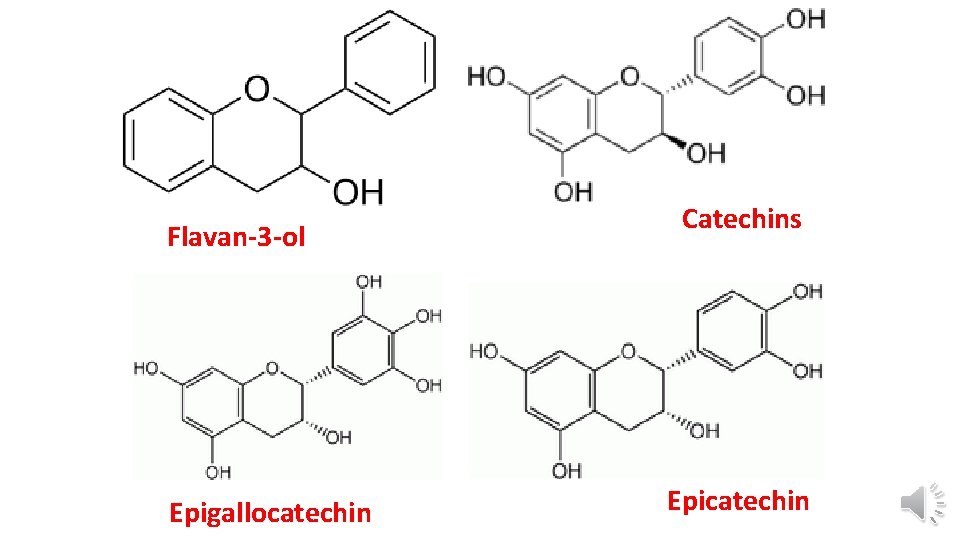
Flavan-3 -ol Epigallocatechin Catechins Epicatechin
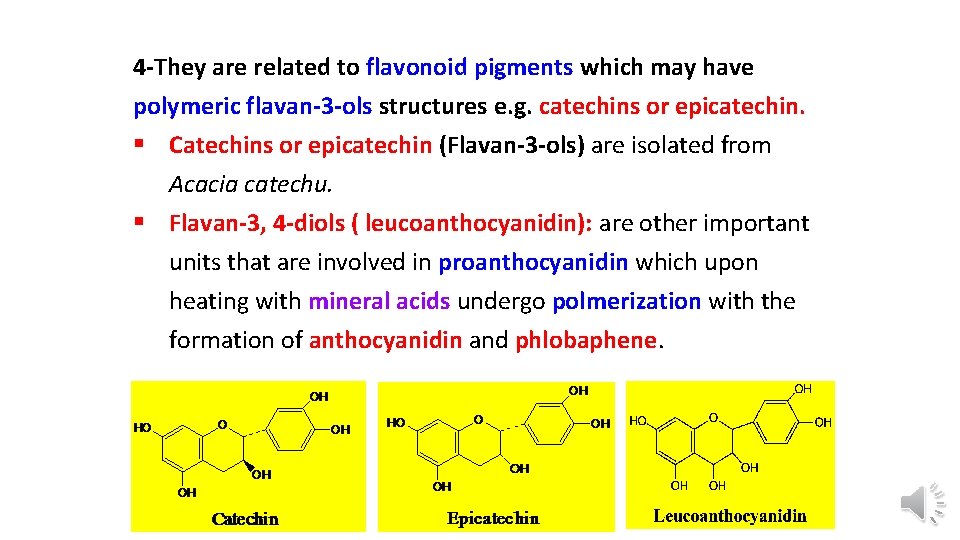
4 -They are related to flavonoid pigments which may have polymeric flavan-3 -ols structures e. g. catechins or epicatechin. § Catechins or epicatechin (Flavan-3 -ols) are isolated from Acacia catechu. § Flavan-3, 4 -diols ( leucoanthocyanidin): are other important units that are involved in proanthocyanidin which upon heating with mineral acids undergo polmerization with the formation of anthocyanidin and phlobaphene.
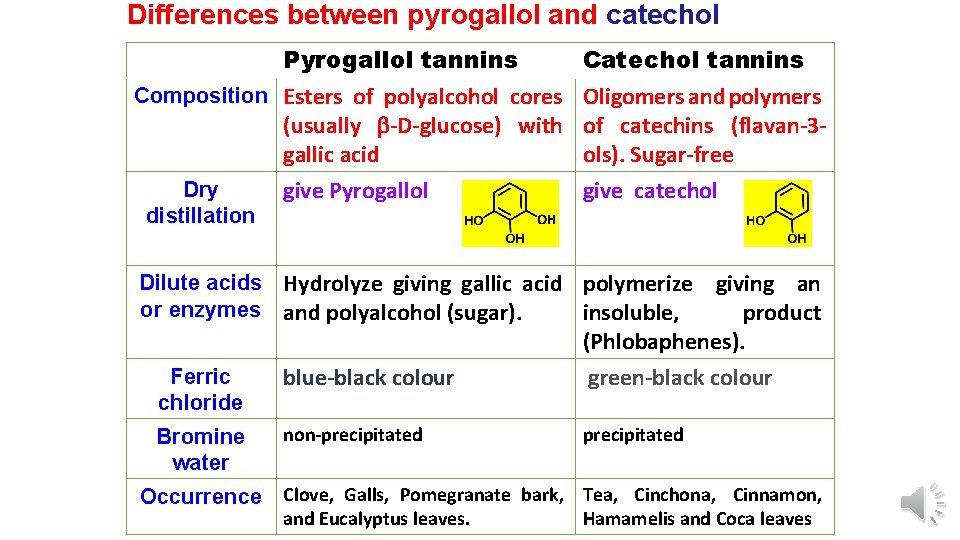
Differences between pyrogallol and catechol Pyrogallol tannins Composition Esters of polyalcohol cores (usually -D-glucose) with gallic acid Dry distillation give Pyrogallol Catechol tannins Oligomers and polymers of catechins (flavan-3 ols). Sugar-free give catechol Dilute acids Hydrolyze giving gallic acid polymerize or enzymes and polyalcohol (sugar). insoluble, giving an product (Phlobaphenes). Ferric chloride blue-black colour green-black colour Bromine water non-precipitated Occurrence Clove, Galls, Pomegranate bark, Tea, Cinchona, Cinnamon, and Eucalyptus leaves. Hamamelis and Coca leaves
- Slides: 8