Tale of Two Adolescent Vaccine Preventable Diseases Pertussis

Tale of Two Adolescent Vaccine. Preventable Diseases: Pertussis and Meningococcal Disease Amanda Cohn, MD National Center for Immunization and Respiratory Diseases Centers for Disease Control and Prevention

Presentation overview • Epidemiology and Transmission • Vaccine properties and immunogenicity • Advisory Committee on Immunization Practices (ACIP) recommendations • Next steps
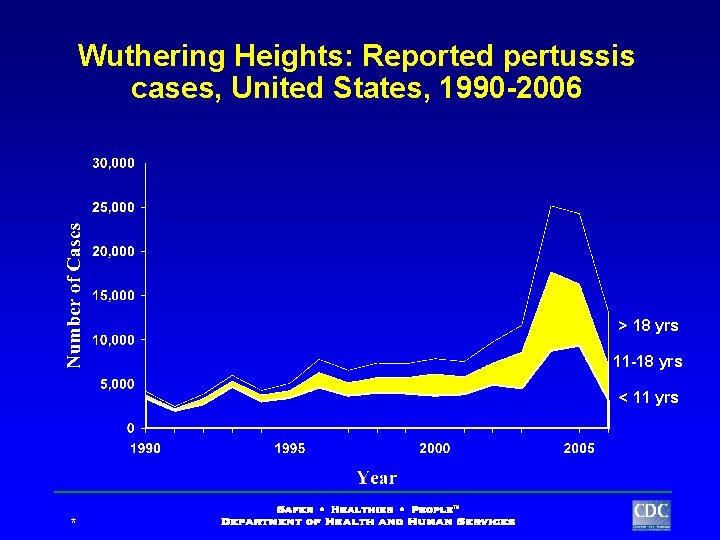
Wuthering Heights: Reported pertussis cases, United States, 1990 -2006 > 18 yrs 11 -18 yrs < 11 yrs *
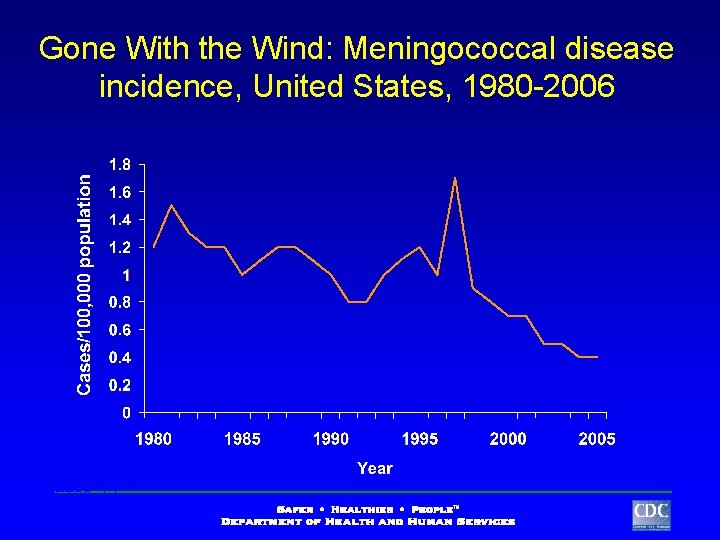
Gone With the Wind: Meningococcal disease incidence, United States, 1980 -2006 NETSS data
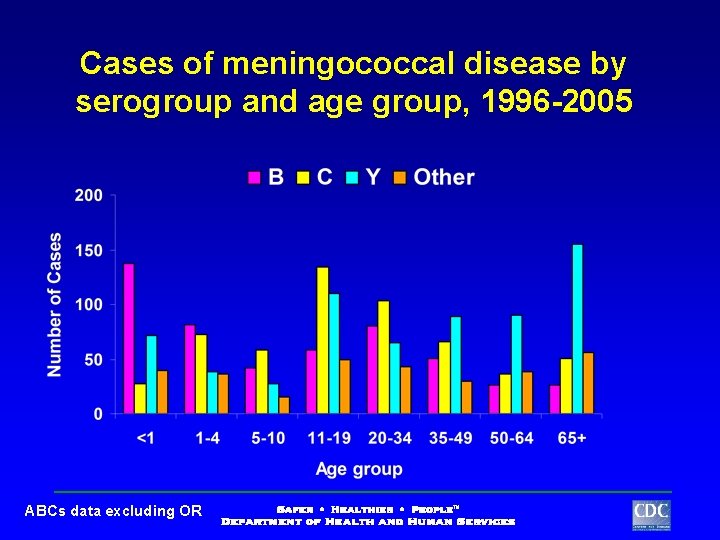
Cases of meningococcal disease by serogroup and age group, 1996 -2005 ABCs data excluding OR
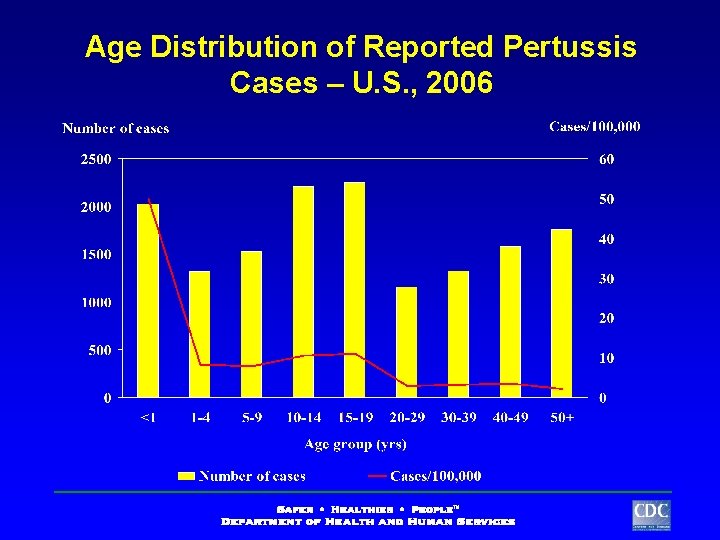
Age Distribution of Reported Pertussis Cases – U. S. , 2006

Steps in reporting, meningococcal disease and pertussis Cough, Cough Medical encounter Hmmm, I just read an article about pertussis… Clinical Recognition What does this result mean? Reliable Diagnostics Hi, I’m calling from your health department Report to Health Department

Pertussis transmission Coughing Adolescent Lots of coughing adolescents + =

Meningococcal disease transmission Asymptomatic Carriers + Invasive Disease =

Meningococcal Disease and Pertussis Epidemiology • Endemic diseases • Cases in all age groups, higher rates in infants and adolescents • Clinical presentations lead to very different recognition and reporting patterns

Meningococcal Conjugate Vaccine (MCV 4) and Acellular Pertussis Vaccine (Tdap)

Meningococcal Conjugate Vaccine Composition • Serogroups A, C, Y, W-135 – 4μg of each capsular polysaccharide – Same as meningococcal polysaccharide vaccine (MPSV 4) • MCV 4 conjugated to 48μg diphtheria toxoid • Potential licensure in 2009 of Men. ACYW-Crm – Same age indication as MCV 4

Tdap Vaccine composition • Tetanus and diphtheria toxoids ~ Td • Acellular pertussis antigens same as DTa. P except some antigens present in lower quantity – BOOSTRIX® ~ INFANRIX®: PT, FHA, PRN – ADACEL® ~ DAPTACEL®: PT, FHA, PRN, Fimbrae 2/3
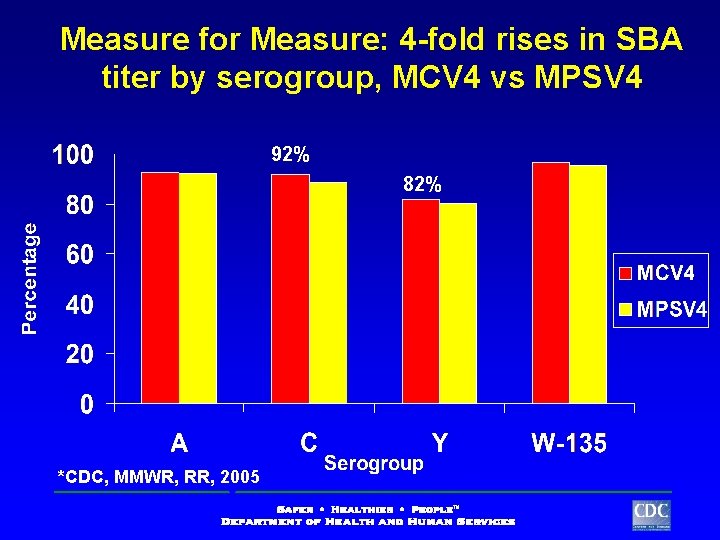
Measure for Measure: 4 -fold rises in SBA titer by serogroup, MCV 4 vs MPSV 4 92% 82% *CDC, RR, 2005 FDA MMWR, Clinical Briefing Document, VRBPAC 09/22/04 Study MTA-02

Vaccine effectiveness of MPSV 4 • VE of MPSV 4 among 2 - to 29 -year-olds= 85% (95% CI, 27%-97%) against serogroup C disease • Presumed advantages of conjugate vaccines – – T cell-dependent response Longer duration of protection Primes for immunologic memory Protects from acquisition of carriage

The Bridge to Terabithia: Tdap Immunogenicity • Efficacy for licensure inferred from immungenicity data • Serologic bridge – Immune responses in adolescents and adults after 1 dose Tdap not inferior to response in infants after 3 doses DTa. P during pertussis efficacy trials • Infant vaccine efficacy approx. 85% – INFANFRIX®: 89% (95% CI: 77%, 95%)* – DAPTACEL®: 85% (95% CI: 80%, 89%)** *Schmitt HJ et al. JAMA 1996; 275: 37 -41 **Gustafsson LH et al. NEJM 1996; 334: 349 -355
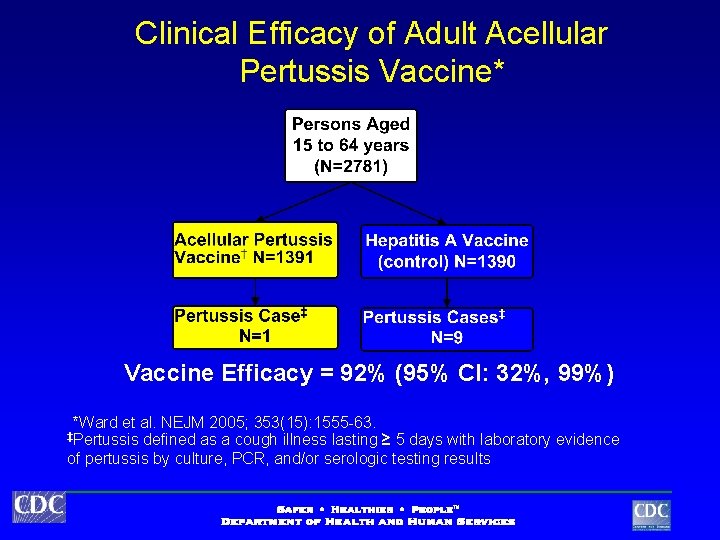
Clinical Efficacy of Adult Acellular Pertussis Vaccine* Vaccine Efficacy = 92% (95% CI: 32%, 99%) *Ward et al. NEJM 2005; 353(15): 1555 -63. defined as a cough illness lasting ≥ 5 days with laboratory evidence of pertussis by culture, PCR, and/or serologic testing results ‡Pertussis

MCV 4 and Tdap • Lack of known protective antibody level for both diseases • Good vaccines, but not perfect • We have a lot to learn – Ability to reduce transmission to unvaccinated groups – Duration of protection

ACIP Recommendations and Implementation Issues

The Three Musketeers: Recommended Adolescent Vaccines

Sense and Sensibility: The art of risk-benefit decisions • Based on little data about risk or benefit – Intervals between Td and Tdap – Use of Tdap in pregnant adolescents • Increasing evidence about risk – Guillian-Barre Syndrome (GBS) and MCV 4

Intervals between Td and Tdap • A 5 year interval between Td and Tdap is encouraged to reduce risk for local and systemic reactions • Theoretical risk with multiple doses of tetanus and diphtheria toxoids (including MCV 4) • Shorter intervals may be used if the benefit outweighs the risk of vaccination

Tdap during pregnancy • No data on safety or effectiveness • ACIP does not recommend vaccination of pregnant women with Tdap – Not a contraindication, may be useful in certain situations • AAP recommends that pregnant adolescents be given the same considerations for immunization as nonpregnant adolescents. If Tdap or Td vaccine is indicated, administer in the second or third trimester

Guillian-Barre Syndrome (GBS) and MCV 4 • Vaccine Adverse Event Reporting system – 33 reported cases of GBS after MCV 4 – No increase in risk in 11 -19 year-olds – Possible small increase in risk in 15 -19 year-olds • Vaccine Safety Datalink (VSD) – One case of GBS pending confirmation, no other cases identified – Over 640, 000 doses in VSD <= 1 case expected • Ongoing risk of meningococcal disease • History of GBS a precaution to receiving MCV 4 ACIP, February 2009

ACIP Recommendations • All adolescents 11 -18 years should receive a dose of Tdap and MCV 4 – Preferably at 11 -12 year-old visit • More data on safety in special situations (pregnancy, intervals) needed and may come with time

Next Steps: Maximizing Prevention Strategies
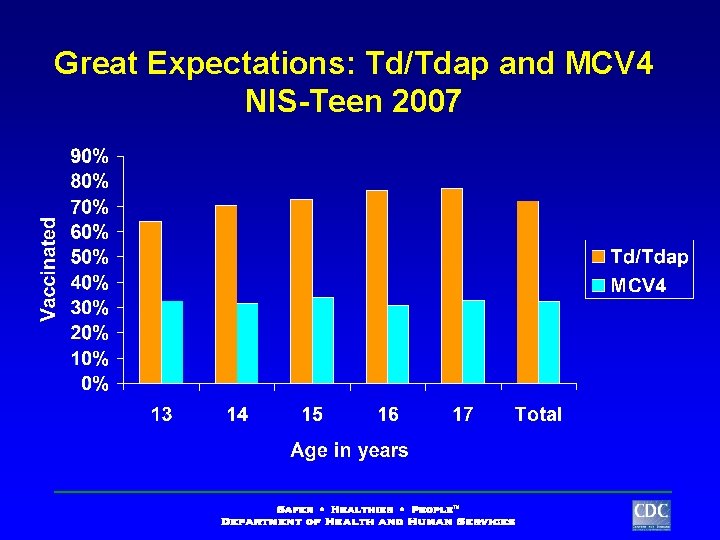
Great Expectations: Td/Tdap and MCV 4 NIS-Teen 2007

Outbreak Management • Susceptible group = 1 - (VE * Coverage) – 1 - (0. 85 * 0. 75)= 47% – 1 - (0. 85*0. 95)= 20% - herd immunity? • Response to vaccine takes 7 -14 days • Vaccination does not replace chemoprophylaxis of close contacts but may reduce transmission

Do you need to give chemoprophylaxis to a vaccinated close contact? • Meningococcal disease – Chemoprophylaxis often given before serogroup data available • Pertussis – Adolescents often exposed multiple times in school outbreaks – Long exposure period

Will we have to revaccinate with Tdap and/or MCV 4? • Duration of protection unknown – Waning immunity of pertussis vaccines – Memory response may not be enough to protect against meningooccal disease • Disease risk continues – Pre-teen girls will become mothers (hopefully after adolescence!) – Pre-teens will become college freshmen

All’s Well that Ends Well: Preventing Meningococcal Disease and Pertussis • Both endemic with transmission among adolescents, different disease patterns • Susceptible populations remain; increasing coverage will reduce disease in the target population and may increase herd immunity across all age groups, e. g. high-risk infants • Safety and effectiveness data are emerging and may help refine recommendations – Intervals, revaccination

Acknowledgements • • • Jessica Mac. Neil Stacey Martin Tami Skoff Stanley Wei Thomas Clark Nancy Messonnier • • • Nihdi Jain Shannon Stokely Elaine Miller Eric Weintraub John Iskander

Thank You! Contact Info: Amanda Cohn Centers for Disease Control and Prevention 1600 Clifton Rd MS C-09 Atlanta, GA 30333 Phone: (404) 639 -6039 E-mail: acohn@cdc. gov
- Slides: 33