TAKS Review Objective 3 The student will demonstrate

TAKS Review Objective 3 The student will demonstrate an understanding of the structures and properties of matter.

Physical or Chemical Property or Change • • Red rock PP • Paper can burn CP • Sharpen a pencil PC • Rusting CC CP Na can react with H 20 • Copper tarnishing CC Candle melting PC • Candle burning CC Boiling point of H 2 O PP Dissolving C 6 H 12 O 6 in H 2 O PC Substance can react CP with another substance Substance forms new substances CC Substance’s form is altered PC ~ KEY ~ PP = physical property CP = chemical property PC = physical change CC = chemical change

Do you know? • Element is a substance that cannot be broken down into simpler substances. • Compound is two or more elements combined to form a new substance whose characteristics are different than the original elements. • Mixture is two or more substances that are not chemically combined and can be separated.

Common Elements & Compounds… • • • He Helium N Nitrogen C Carbon Ca Calcium Cl Chlorine Si Silicon • • • H 2 O Water CO 2 Carbon Dioxide Na. Cl Sodium Chloride (salt) C 6 H 12 O 6 Sugar CO Carbon Monoxide HCl Hydrochloric Acid
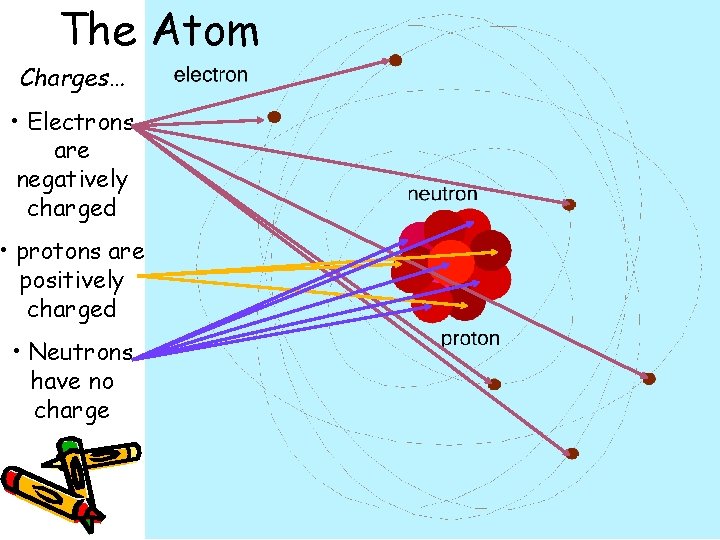
The Atom Charges… • Electrons are negatively charged • protons are positively charged • Neutrons have no charge
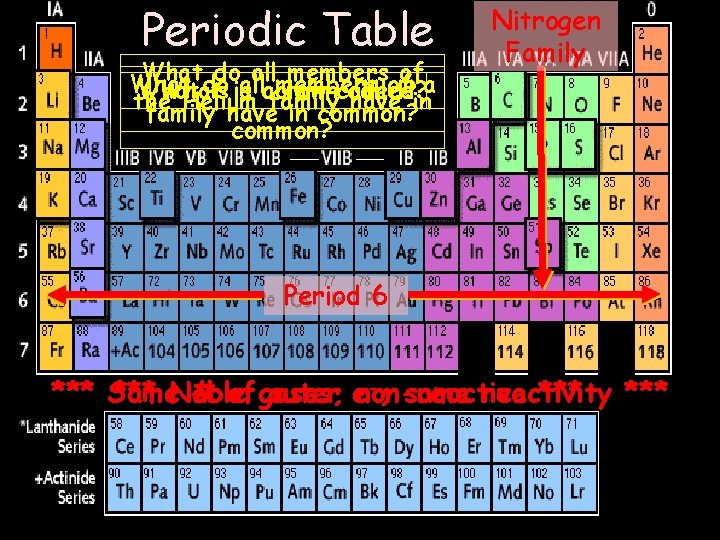
Periodic Table Whatdo doall allmembersof ofa What is is a a column row called? the Helium family have in common? Nitrogen Family Period 6 -; same reactivity *** Same *** Noble # of gases; outer enon-reactive ***

Chemical Reactions & Equations Hydrochloric Acid + Sodium Hydroxide Sodium Chloride + Water HCl + Na. OH Na. Cl + H 2 O Is this equation balanced? H=2 Cl = 1 Na = 1 O=1 YES, it is balanced.
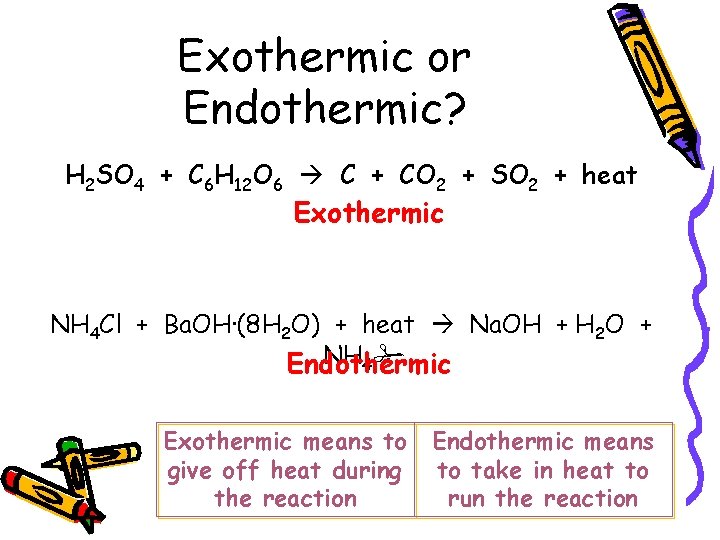
Exothermic or Endothermic? H 2 SO 4 + C 6 H 12 O 6 C + CO 2 + SO 2 + heat Exothermic NH 4 Cl + Ba. OH·(8 H 2 O) + heat Na. OH + H 2 O + NH 4 Endothermic Exothermic means to give off heat during the reaction Endothermic means to take in heat to run the reaction

Specific Heat • Also know as heat capacity • The amount of heat needed to raise the temperature of 1 g of a substance 1°C • Substances with low specific heat will heat very quickly • Substance with high specific heat values require larger amounts of heat to cause its temperature to raise the same amount

Which material will loses heat faster, Which material will stay hot the liquid water or solid heat longest? the fastest? water?

Specific Heat We have land sea breezes… …due to the differences in specific heat of the land of the water. Water has a much higher specific heat which means it takes more energy to heat the water than the land. Since the land heats faster during the day, the breeze blows in from the sea and vice versa.
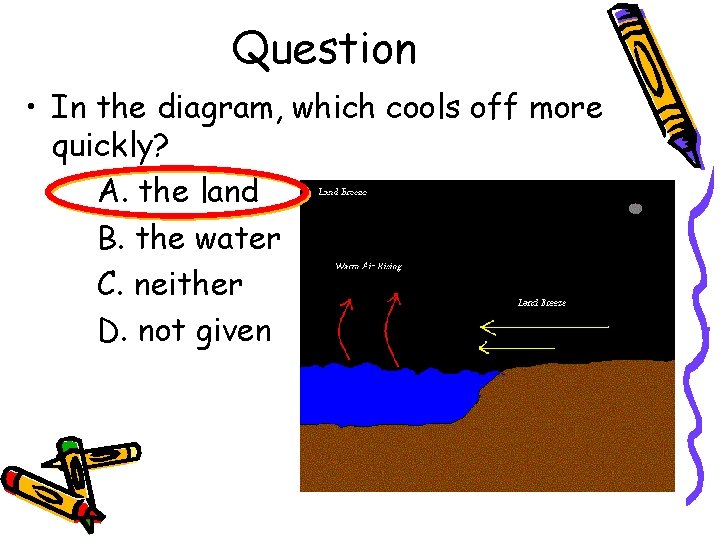
Question • In the diagram, which cools off more quickly? A. the land B. the water C. neither D. not given

Question • When a drain cleaner is added to a clogged sink, the drainpipe becomes warm to the touch. What type of chemical reaction is occurring to cause the pipe to become warm? Since heat is being given off as evidenced by the warming of the pipe, this chemical reaction must be exothermic.

Reminders… • We balance equations because of the law of Conservation of Mass • Know common element symbols including C, N, O, H, He, Na, Cl, Al, Si, &S • Specific heat affects the world around us including weather • Know that the atom is the smallest particle of a substance » Any questions? ? ?
- Slides: 14