Table of Contents Chapter 25 Nuclear Chemistry Nuclear

Table of Contents Chapter 25: Nuclear Chemistry Nuclear Radiation

Nuclear Chemistry: Basic Concepts Nuclear Radiation • Nuclear chemistry is the study of the structure of atomic nuclei and the changes they undergo. Radioactivity • 1895 - Wilhelm Roentgen – discovered invisible rays were emitted when electrons bombarded the surface of certain materials. • Henri Becquerel studied minerals that emit light after being exposed to sunlight, called phosphorescence – discovered some that are continually phosphorescent.

Nuclear Chemistry: Basic Concepts The Discovery of Radioactivity • Marie & Pierre Curie isolated the components emitting the rays. • Radioactivity: the process by which materials give off such rays. • Radiation: rays and particles emitted by a radioactive source. • Radioisotopes: Isotopes of atoms with unstable nuclei (emits radiation).

Nuclear Chemistry: Basic Concepts Types of Radiation • Radioactive decay: The process by which unstable nuclei emit radiation to attain more stable atomic configurations. • During radioactive decay, unstable atoms lose energy by emitting one of several types of radiation. • The three most common types of radioactive decay are • alpha (α) • beta (β) • gamma (γ) Decay = BREAKDOWN!
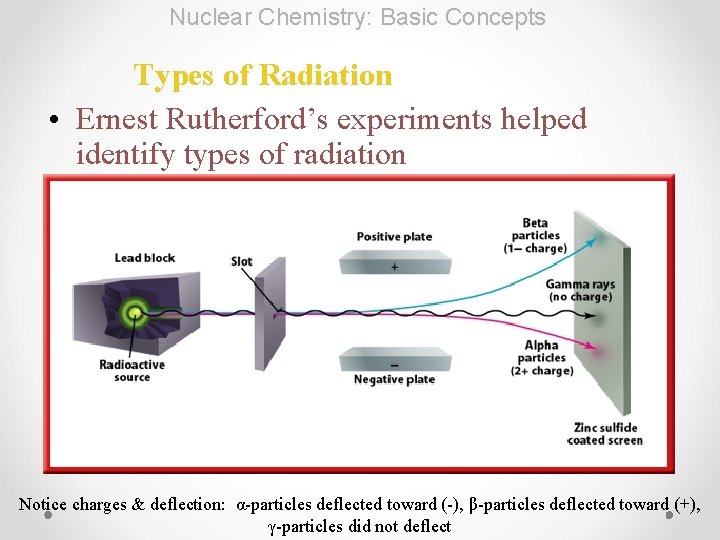
Nuclear Chemistry: Basic Concepts Types of Radiation • Ernest Rutherford’s experiments helped identify types of radiation Notice charges & deflection: α-particles deflected toward (-), β-particles deflected toward (+), γ-particles did not deflect

Nuclear Chemistry: Basic Concepts Types of Radiation • Alpha Decay • The release of a helium nucleus • 2 protons, 2 neutrons • Big and low energy • Change: Z decreases by 2 / A decreases by 4 Notice #s on right = #s on left

Nuclear Chemistry: Basic Concepts Try it out! • Write a balanced nuclear equation for the alpha decay of thorium-230 ( ). 230 90 Th → 226 88 Ra + 42 He What element is formed when polonium-214 radioisotope undergoes alpha decay? Give the atomic number and mass number of the element.

Nuclear Chemistry: Basic Concepts Types of Radiation • Beta Decay • A neutron becomes a proton, releasing an electron (called a beta particle) • Electron originating in the nucleus when a neutron becomes a proton • Small, high energy • Change: Z increases by 1 / A remains constant

Basic Assessment Questions Question 2 What element is formed when undergoes beta decay? Give the atomic number and mass number of the element.

Nuclear Chemistry: Basic Concepts Types of Radiation • Gamma Decay • High energy electromagnetic waves (similar to light released as an atom relaxes) • Doesn’t change atomic number or mass of the element • Not a particle, just energy – a relaxation process • Never occurs on its own
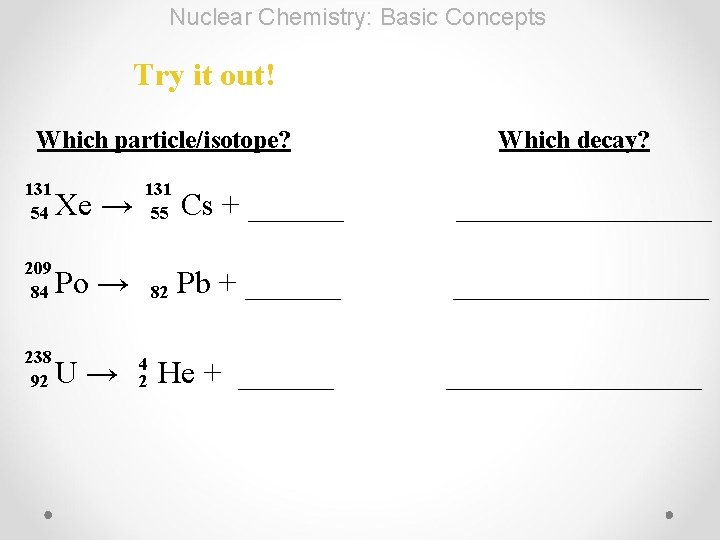
Nuclear Chemistry: Basic Concepts Try it out! Which particle/isotope? 131 54 Which decay? Xe → 131 55 Cs + ___________ 209 84 Po → 82 Pb + ___________ 238 92 U→ He + ___________ 4 2
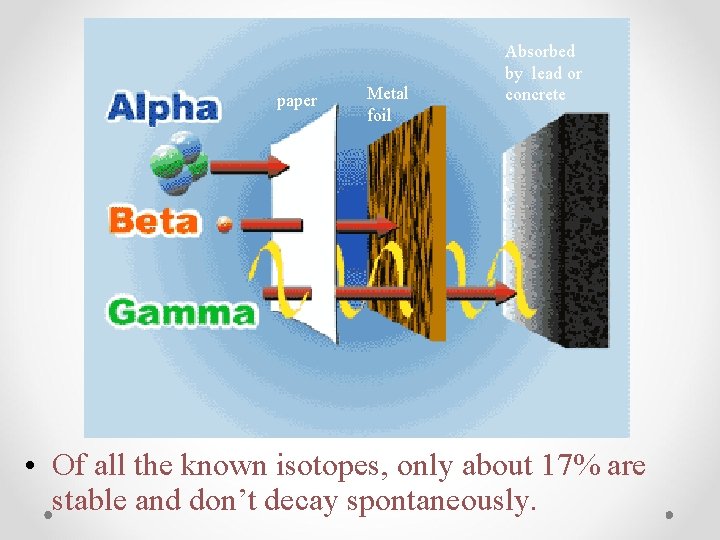
paper Metal foil Absorbed by lead or concrete • Of all the known isotopes, only about 17% are stable and don’t decay spontaneously.

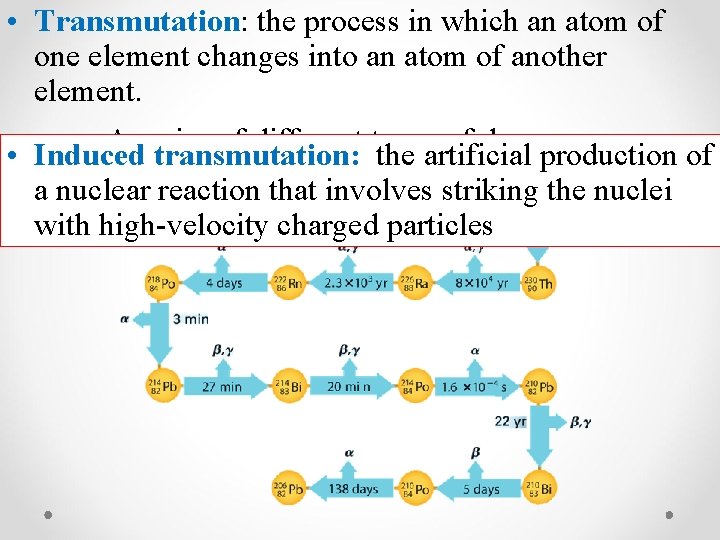
• Transmutation: the process in which an atom of one element changes into an atom of another element. • A series of different types of decay • Induced transmutation: the artificial production of a nuclear reaction that involves striking the nuclei with high-velocity charged particles
- Slides: 14