T Cell Effector Function and Cytokine Biology Georgina

T Cell Effector Function and Cytokine Biology Georgina Xanthou, Ph. D Cellular Immunology Laboratory Center for Basic Research, Biomedical Research Foundation of the Academy of Athens

Adaptive Immunity Ø Recognizes non self & self antigens (in pathological cases) Ø Responds to a foreign invader Ø Distinguishes between invaders (potential pathogen) Recognition of a non-self presence causes: Humoral responses – based upon the presence of antibodies Cellular responses – based upon the activity of T lymphocytes Works on the basis of: Ø Specificity Ø Diversity Ø Memory
T cell-mediated immunity Ø There are only a few lymphocytes with antigen receptors for any particular epitope. Ø In the lymph nodes, an antigen is exposed to a steady stream of lymphocytes until a match with the TCR is made. Ø This binding of a mature lymphocyte to an antigen initiates events that activate the lymphocyte. Ø Clusters of Differentiation (CD): An internationally-recognized systematic nomenclature for cell surface molecules used to discriminate between hematopoietic cells
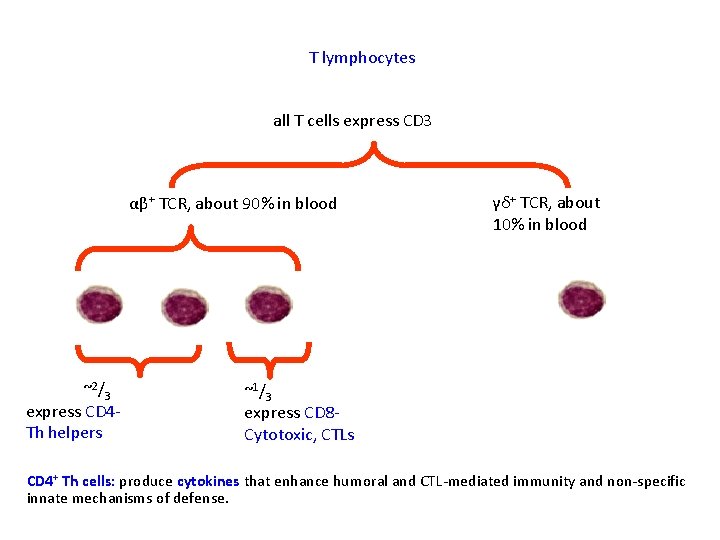
T lymphocytes all T cells express CD 3 αβ+ TCR, about 90% in blood ~2/3 express CD 4 Th helpers γδ+ TCR, about 10% in blood ~1/3 express CD 8 Cytotoxic, CTLs CD 4+ Th cells: produce cytokines that enhance humoral and CTL-mediated immunity and non-specific innate mechanisms of defense.
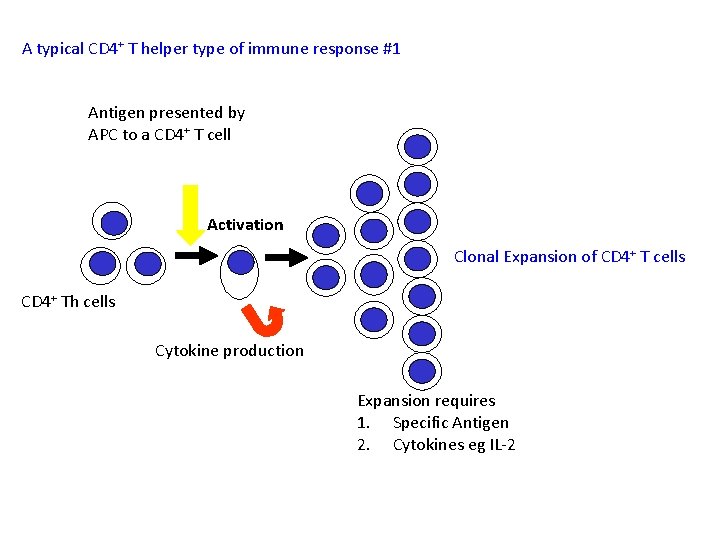
A typical CD 4+ T helper type of immune response #1 Antigen presented by APC to a CD 4+ T cell Activation Clonal Expansion of CD 4+ T cells CD 4+ Th cells Cytokine production Expansion requires 1. Specific Antigen 2. Cytokines eg IL-2
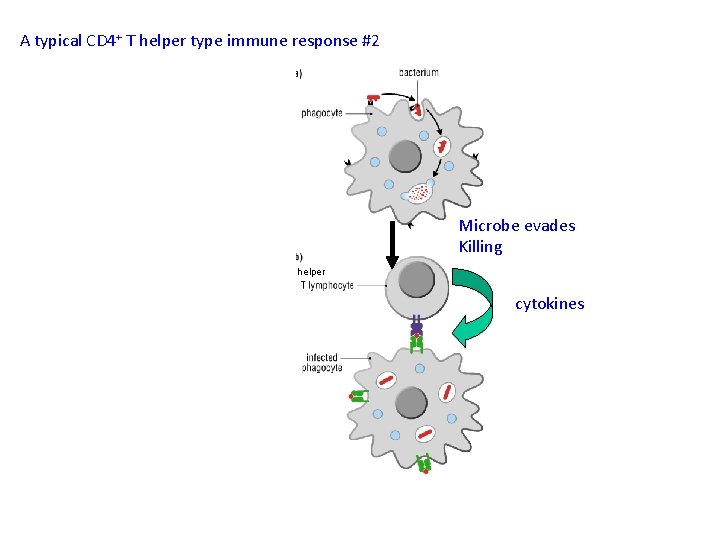
A typical CD 4+ T helper type immune response #2 Microbe evades Killing helper cytokines
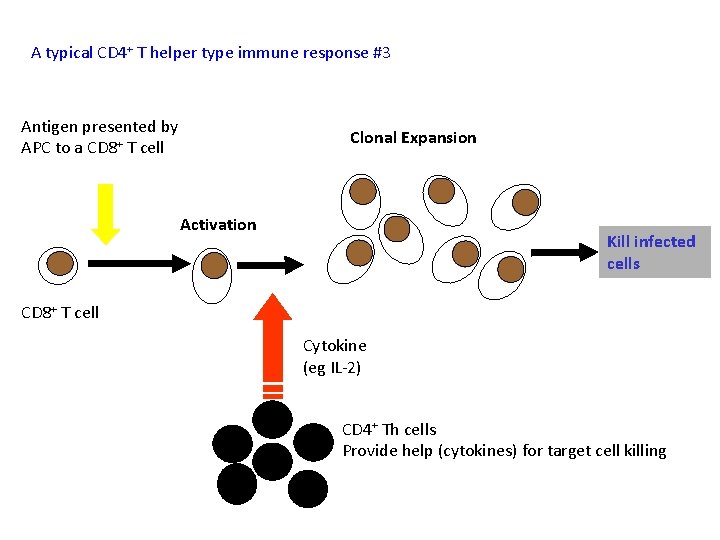
A typical CD 4+ T helper type immune response #3 Antigen presented by APC to a CD 8+ T cell Clonal Expansion Activation Kill infected cells CD 8+ T cell Cytokine (eg IL-2) CD 4+ Th cells Provide help (cytokines) for target cell killing
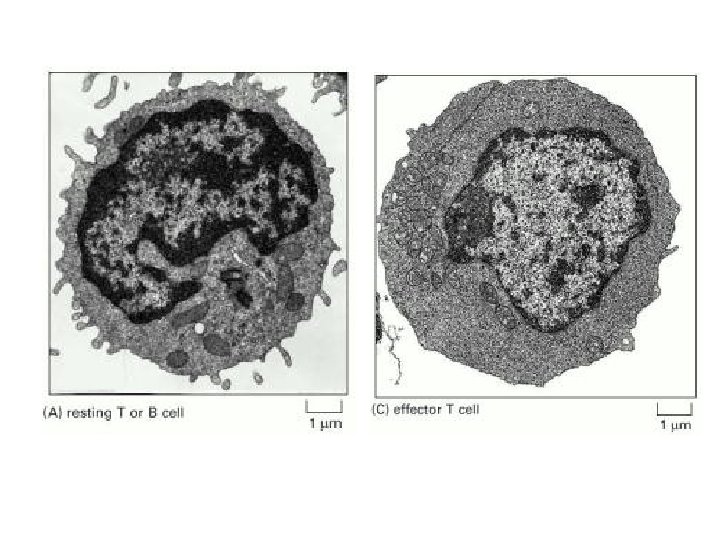
T lymphocytes

CD 8+ Cytotoxic T Cells (CTLs) Ø CTLs are the effector cells in cell-mediated adaptive responses Ø Defense against intracellular pathogens, including bacteria and viruses Ø CTLs recognize fragments of foreign proteins produced by infected cells and possess an accessory protein that binds to class I MHC molecules Ø Activated CTLs secrete proteins that disrupt the membranes of target cells and trigger apoptosis.
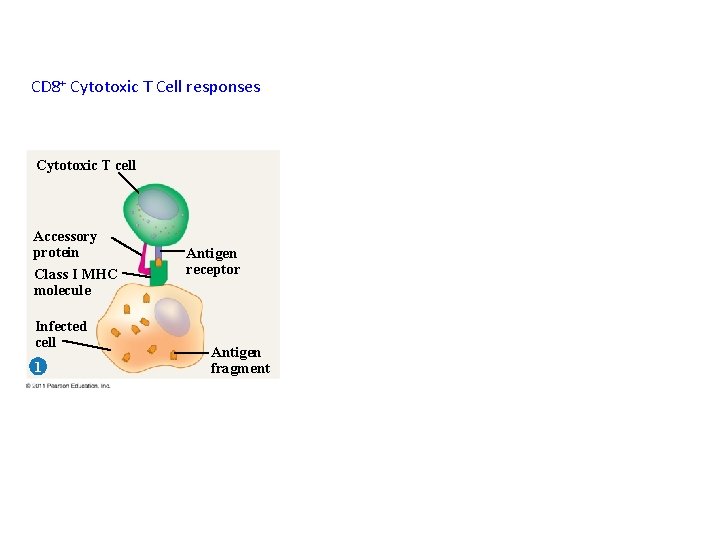
CD 8+ Cytotoxic T Cell responses Cytotoxic T cell Accessory protein Class I MHC molecule Infected cell 1 Antigen receptor Antigen fragment
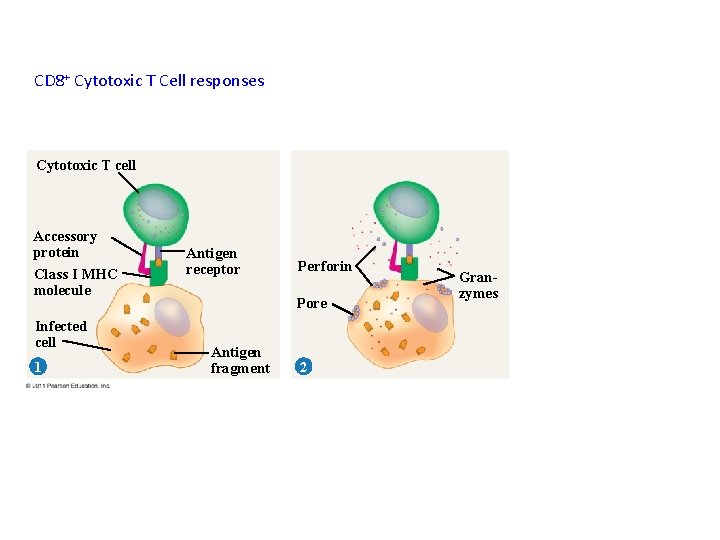
CD 8+ Cytotoxic T Cell responses Cytotoxic T cell Accessory protein Class I MHC molecule Infected cell 1 Antigen receptor Perforin Pore Antigen fragment 2 Granzymes
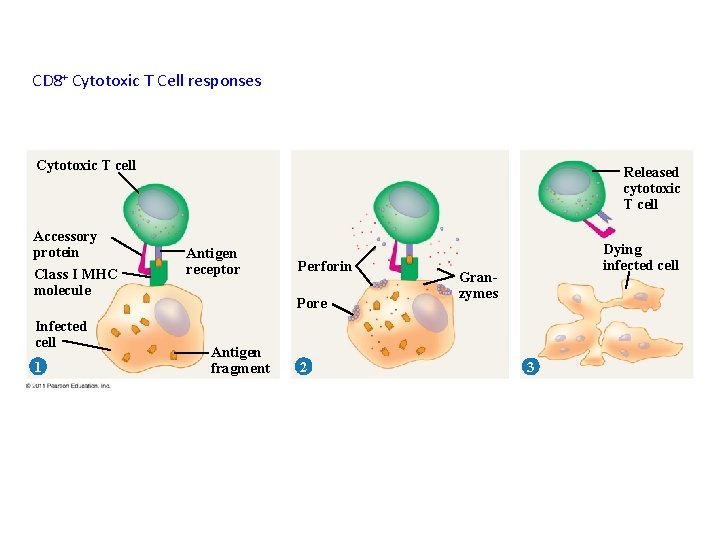
CD 8+ Cytotoxic T Cell responses Cytotoxic T cell Accessory protein Class I MHC molecule Infected cell 1 Released cytotoxic T cell Antigen receptor Perforin Pore Antigen fragment 2 Dying infected cell Granzymes 3
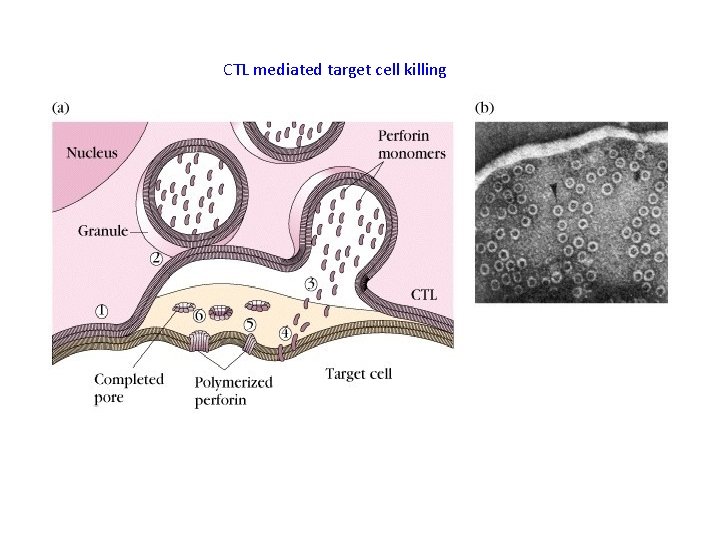
CTL mediated target cell killing
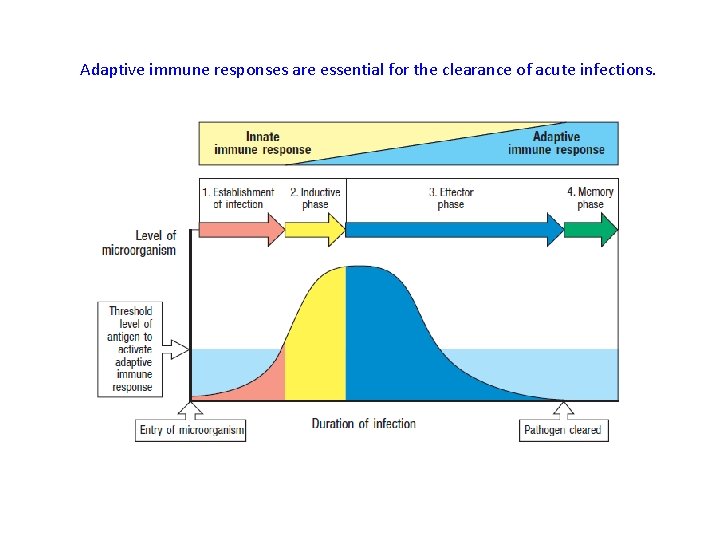
Adaptive immune responses are essential for the clearance of acute infections.

Immunological Memory: Ø Immunological memory is responsible for long-term protections against diseases, due to either a prior infection or vaccination. Ø The first exposure to a specific antigen represents the primary immune response. Ø During this time, selected B and T cells give rise to their effector forms. Ø In the secondary immune response, memory cells facilitate a faster, more efficient response.
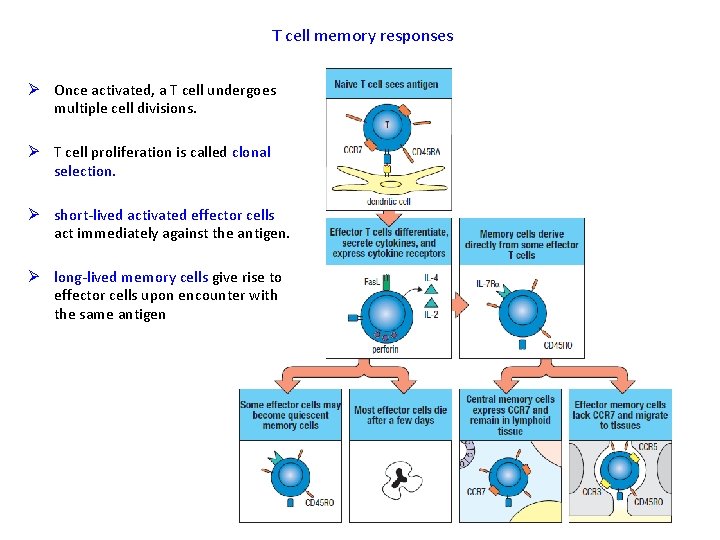
T cell memory responses Ø Once activated, a T cell undergoes multiple cell divisions. Ø T cell proliferation is called clonal selection. Ø short-lived activated effector cells act immediately against the antigen. Ø long-lived memory cells give rise to effector cells upon encounter with the same antigen
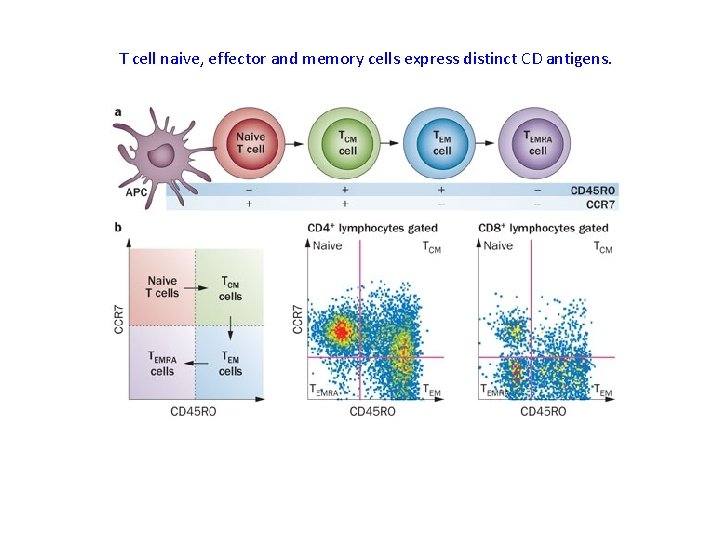
T cell naive, effector and memory cells express distinct CD antigens.
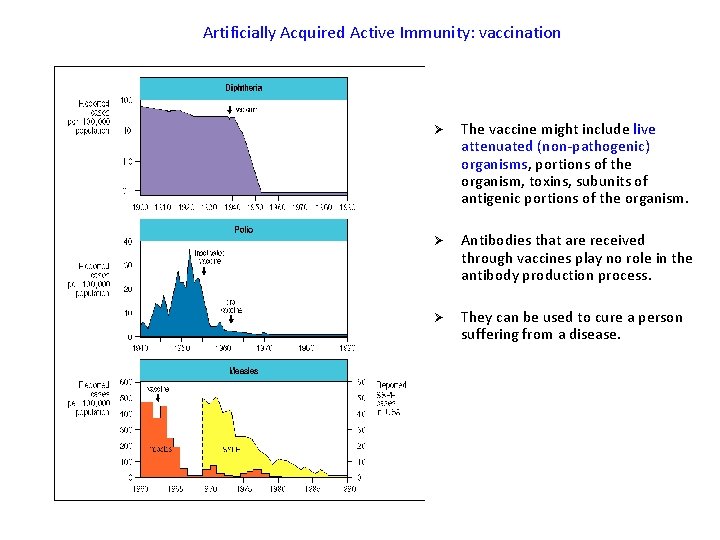
Artificially Acquired Active Immunity: vaccination Ø The vaccine might include live attenuated (non-pathogenic) organisms, portions of the organism, toxins, subunits of antigenic portions of the organism. Ø Antibodies that are received through vaccines play no role in the antibody production process. Ø They can be used to cure a person suffering from a disease.
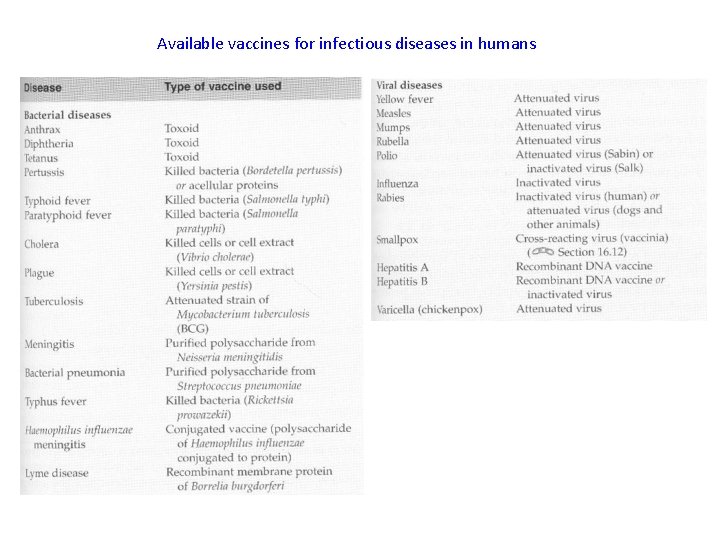
Available vaccines for infectious diseases in humans
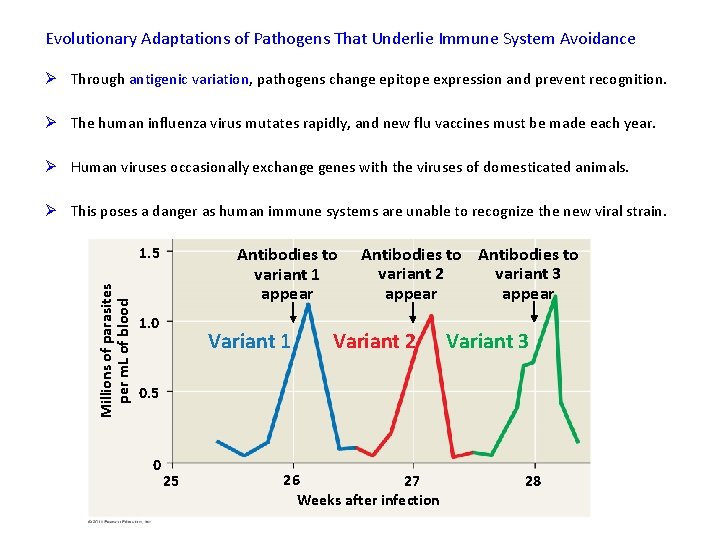
Evolutionary Adaptations of Pathogens That Underlie Immune System Avoidance Ø Through antigenic variation, pathogens change epitope expression and prevent recognition. Ø The human influenza virus mutates rapidly, and new flu vaccines must be made each year. Ø Human viruses occasionally exchange genes with the viruses of domesticated animals. Ø This poses a danger as human immune systems are unable to recognize the new viral strain. Millions of parasites per m. L of blood 1. 5 Antibodies to variant 1 appear 1. 0 Variant 1 Antibodies to variant 2 variant 3 appear Variant 2 Variant 3 0. 5 0 25 26 27 Weeks after infection 28

Th cell differentiation and effector function in protective immunity
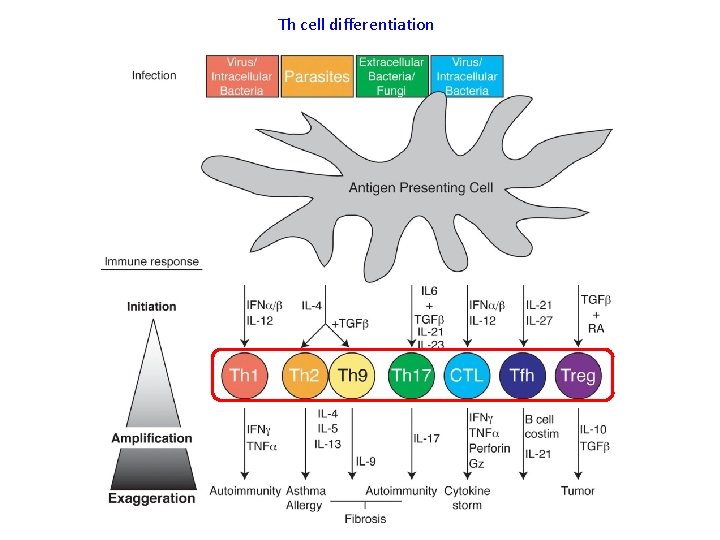
Th cell differentiation
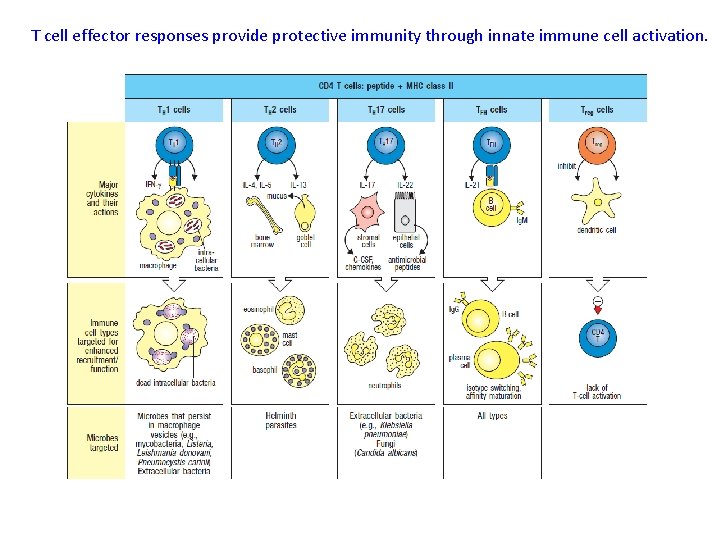
T cell effector responses provide protective immunity through innate immune cell activation.
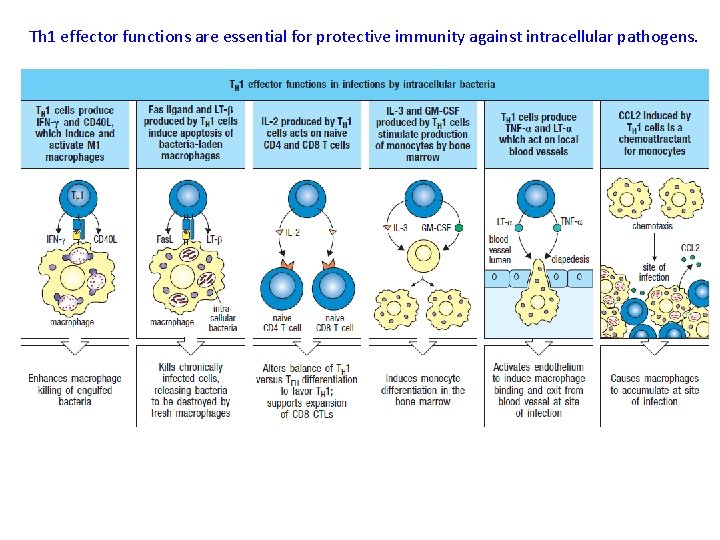
Th 1 effector functions are essential for protective immunity against intracellular pathogens.
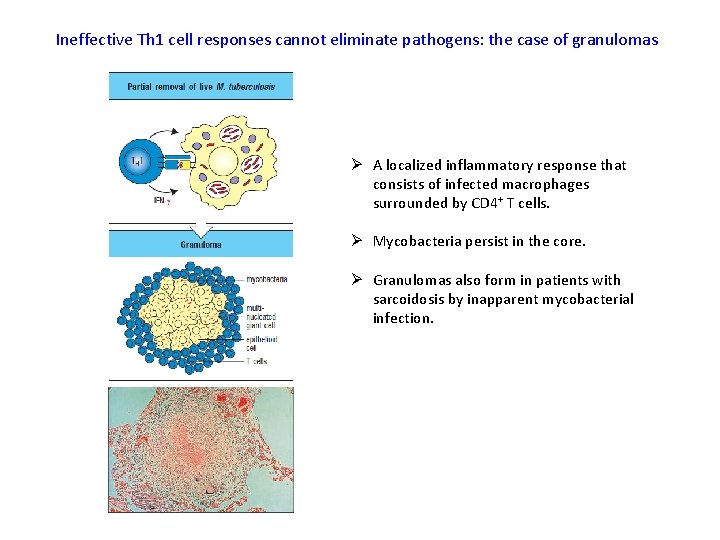
Ineffective Th 1 cell responses cannot eliminate pathogens: the case of granulomas Ø A localized inflammatory response that consists of infected macrophages surrounded by CD 4+ T cells. Ø Mycobacteria persist in the core. Ø Granulomas also form in patients with sarcoidosis by inapparent mycobacterial infection.
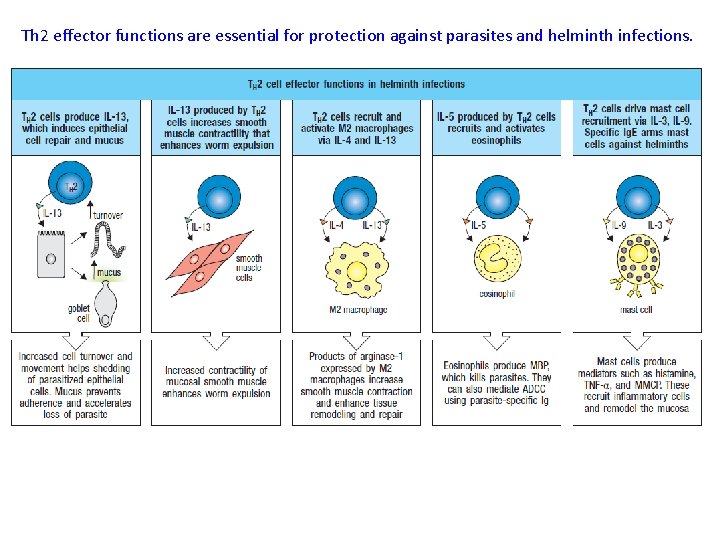
Th 2 effector functions are essential for protection against parasites and helminth infections.
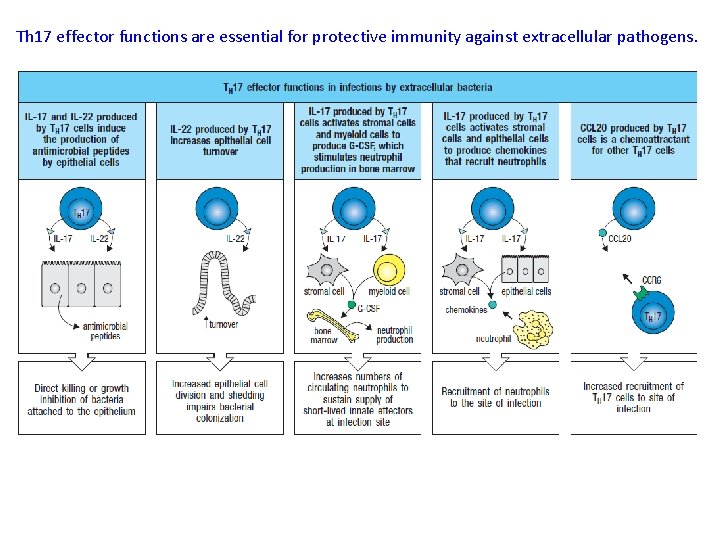
Th 17 effector functions are essential for protective immunity against extracellular pathogens.
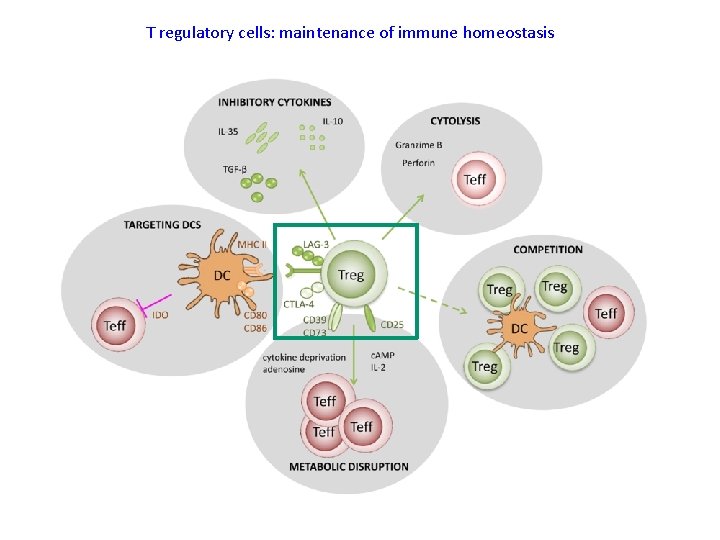
T regulatory cells: maintenance of immune homeostasis
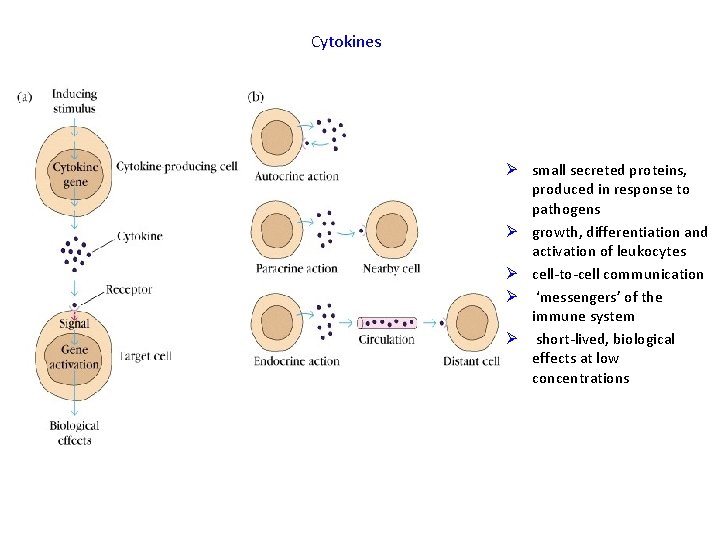
Cytokines Ø small secreted proteins, produced in response to pathogens Ø growth, differentiation and activation of leukocytes Ø cell-to-cell communication Ø ‘messengers’ of the immune system Ø short-lived, biological effects at low concentrations
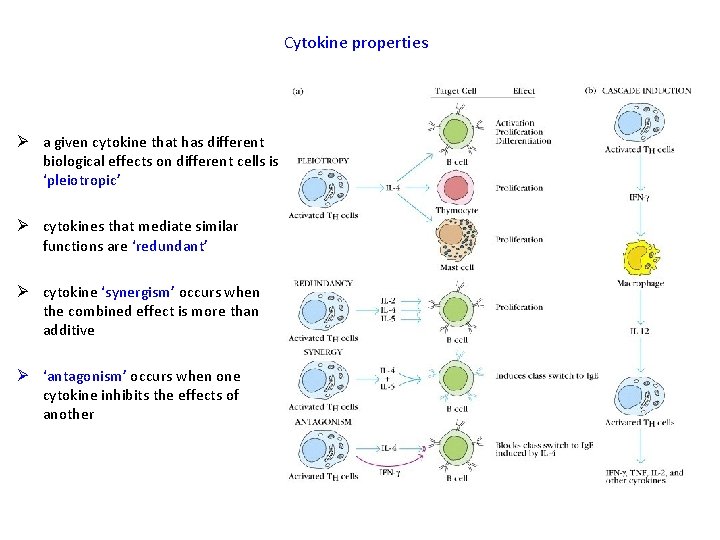
Cytokine properties Ø a given cytokine that has different biological effects on different cells is ‘pleiotropic’ Ø cytokines that mediate similar functions are ‘redundant’ Ø cytokine ‘synergism’ occurs when the combined effect is more than additive Ø ‘antagonism’ occurs when one cytokine inhibits the effects of another

Major cytokines involved in T cell responses Ø IL-1β, TNF-α, IL-6, proinflammatory and cytotoxic role Ø IL-2, Th proliferation, NK proliferation and activation Ø IL-4, B cell Ig class switching, Th proliferation and differentiation Ø IL-5, eosinophil proliferation and differentiation Ø IL-10, TGF-β, immunosupression, inhibit effector cell proliferation Ø IFN-α/β, IFN-γ, anti-viral immunity, macrophage/DC activation, B cell Ig class switching Ø IL-12, directs adaptive immunity, activates NK cells
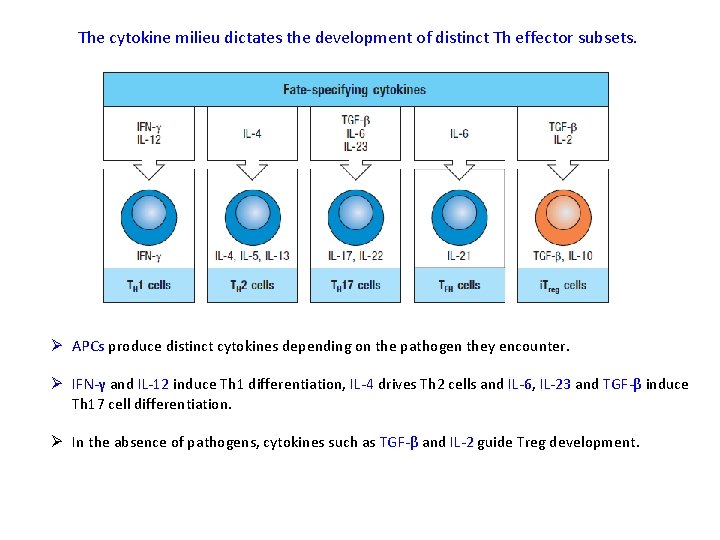
The cytokine milieu dictates the development of distinct Th effector subsets. Ø APCs produce distinct cytokines depending on the pathogen they encounter. Ø IFN-γ and IL-12 induce Th 1 differentiation, IL-4 drives Th 2 cells and IL-6, IL-23 and TGF-β induce Th 17 cell differentiation. Ø In the absence of pathogens, cytokines such as TGF-β and IL-2 guide Treg development.

Cytokines signal through the STAT family of transcription factors to initiate Th cell differentiation. STATs are activated downstream of cytokine receptor signalling and induce key Th cell programming transcription factors, such as T-bet (Th 1), GATA-3 (Th 2), RORγT (Th 17), BCL 6 (Tfh) and Foxp 3 (Treg).
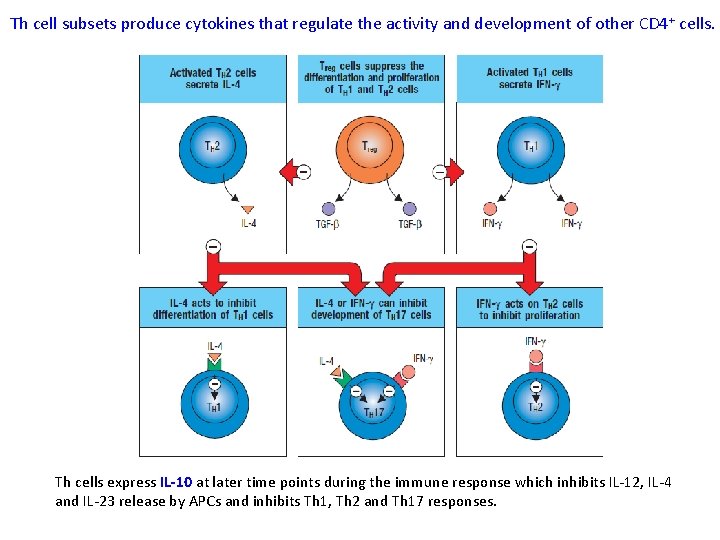
Th cell subsets produce cytokines that regulate the activity and development of other CD 4 + cells. Th cells express IL-10 at later time points during the immune response which inhibits IL-12, IL-4 and IL-23 release by APCs and inhibits Th 1, Th 2 and Th 17 responses.
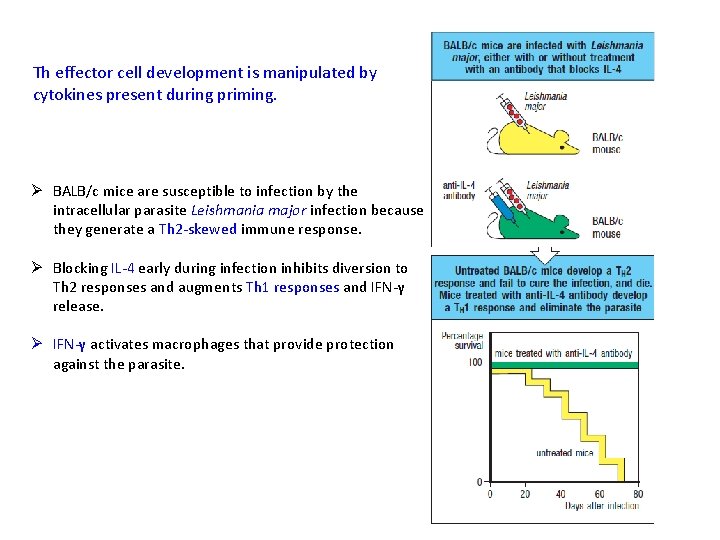
Th effector cell development is manipulated by cytokines present during priming. Ø BALB/c mice are susceptible to infection by the intracellular parasite Leishmania major infection because they generate a Th 2 -skewed immune response. Ø Blocking IL-4 early during infection inhibits diversion to Th 2 responses and augments Th 1 responses and IFN-γ release. Ø IFN-γ activates macrophages that provide protection against the parasite.
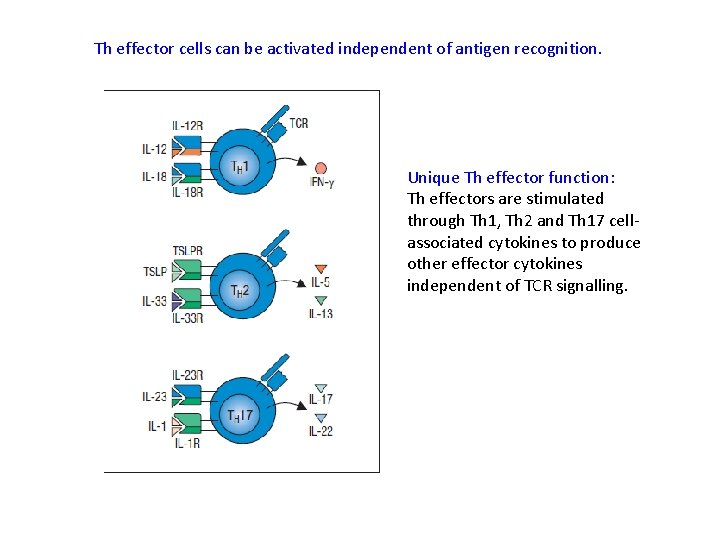
Th effector cells can be activated independent of antigen recognition. Unique Th effector function: Th effectors are stimulated through Th 1, Th 2 and Th 17 cellassociated cytokines to produce other effector cytokines independent of TCR signalling.
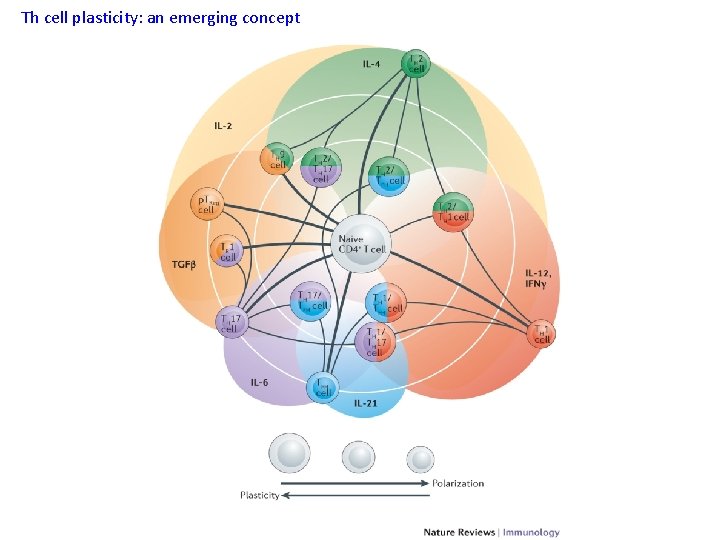
Th cell plasticity: an emerging concept
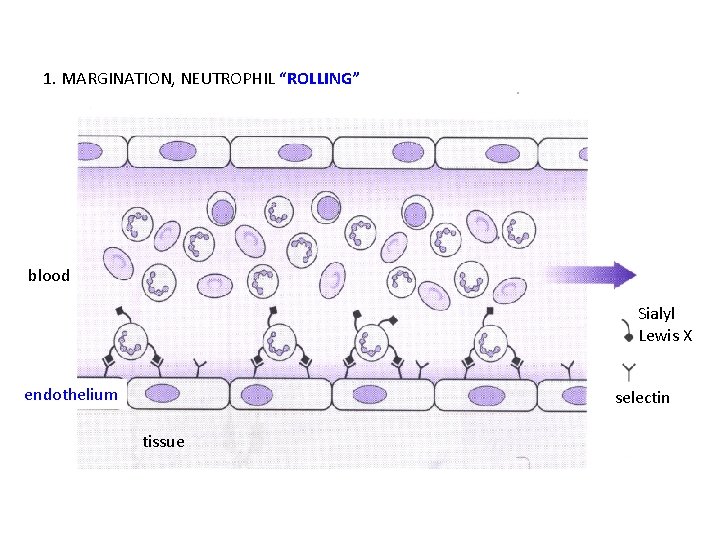
1. MARGINATION, NEUTROPHIL “ROLLING” blood Sialyl Lewis X endothelium selectin tissue
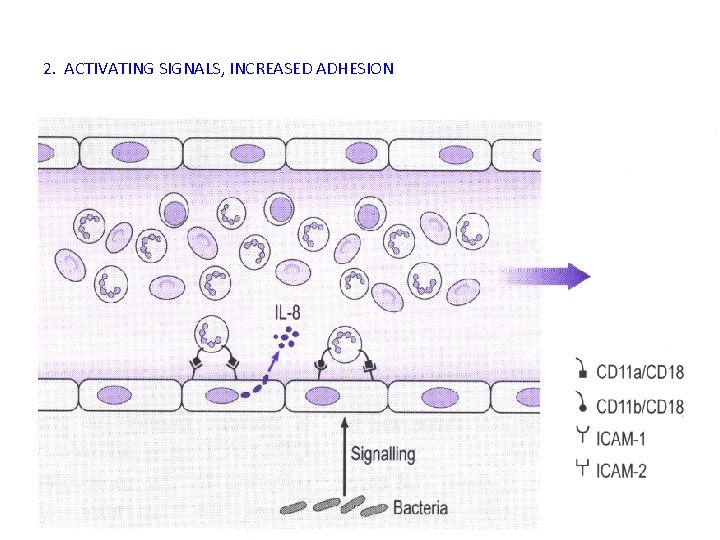
2. ACTIVATING SIGNALS, INCREASED ADHESION
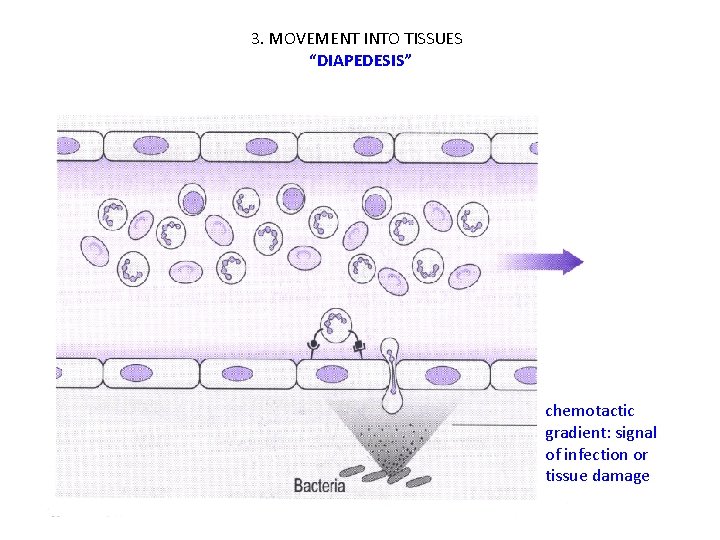
3. MOVEMENT INTO TISSUES “DIAPEDESIS” chemotactic gradient: signal of infection or tissue damage
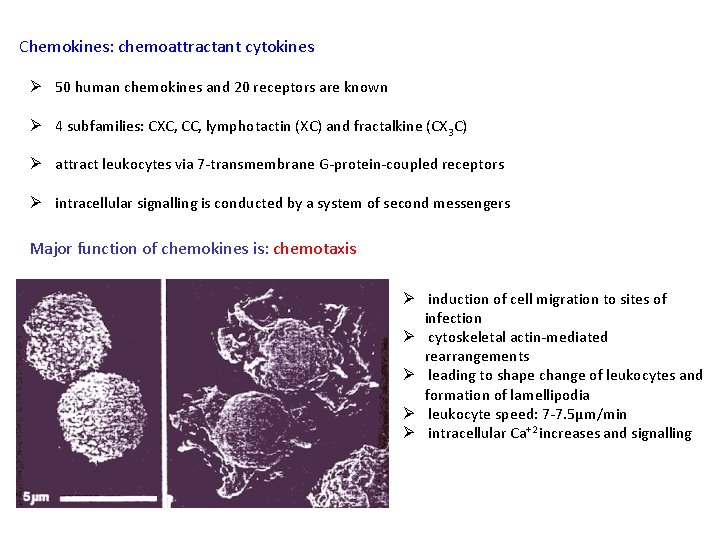
Chemokines: chemoattractant cytokines Ø 50 human chemokines and 20 receptors are known Ø 4 subfamilies: CXC, CC, lymphotactin (XC) and fractalkine (CX 3 C) Ø attract leukocytes via 7 -transmembrane G-protein-coupled receptors Ø intracellular signalling is conducted by a system of second messengers Major function of chemokines is: chemotaxis Ø induction of cell migration to sites of infection Ø cytoskeletal actin-mediated rearrangements Ø leading to shape change of leukocytes and formation of lamellipodia Ø leukocyte speed: 7 -7. 5μm/min Ø intracellular Ca+2 increases and signalling
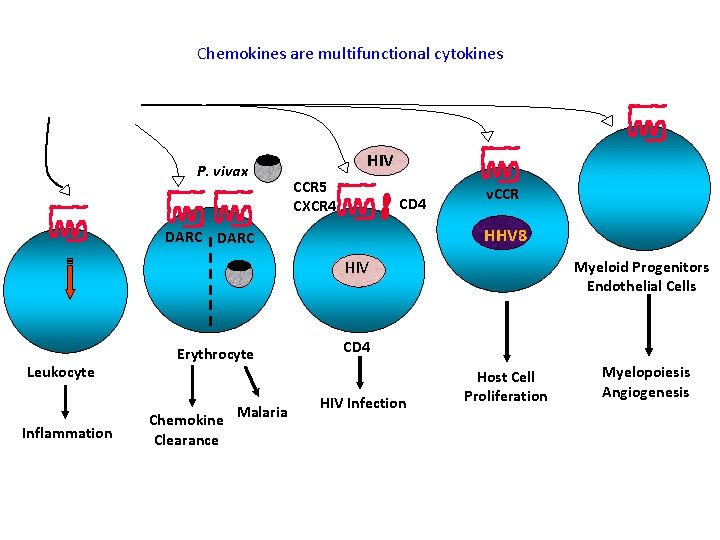
Chemokines are multifunctional cytokines P. vivax HIV CCR 5 CXCR 4 CD 4 v. CCR HHV 8 DARC HIV Leukocyte Inflammation Erythrocyte Chemokine Clearance Malaria Myeloid Progenitors Endothelial Cells CD 4 HIV Infection Host Cell Proliferation Myelopoiesis Angiogenesis

During Th effector cell differentiation chemokine receptor expression is altered. Ø Effector Th cell subsets lose CCR 7 and express chemokine receptors guided by cytokine and chemokine signals. Ø CXCR 3 & CCR 5 characterize Th 1 cells. Ø CRTH 2 & CCR 4 characterize Th 2 cells. Ø CCR 6 & CCR 9 characterize Th 17 cells.

Th cell differentiation and effector function in human immune diseases
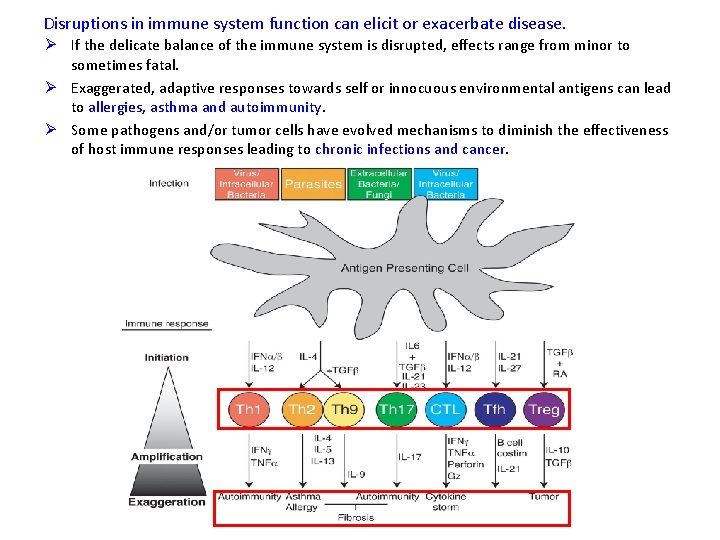
Disruptions in immune system function can elicit or exacerbate disease. Ø If the delicate balance of the immune system is disrupted, effects range from minor to sometimes fatal. Ø Exaggerated, adaptive responses towards self or innocuous environmental antigens can lead to allergies, asthma and autoimmunity. Ø Some pathogens and/or tumor cells have evolved mechanisms to diminish the effectiveness of host immune responses leading to chronic infections and cancer.
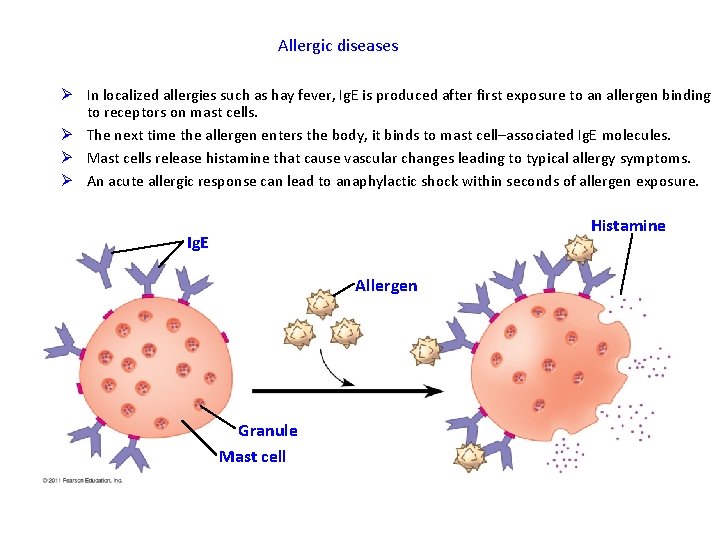
Allergic diseases Ø In localized allergies such as hay fever, Ig. E is produced after first exposure to an allergen binding to receptors on mast cells. Ø The next time the allergen enters the body, it binds to mast cell–associated Ig. E molecules. Ø Mast cells release histamine that cause vascular changes leading to typical allergy symptoms. Ø An acute allergic response can lead to anaphylactic shock within seconds of allergen exposure. Histamine Ig. E Allergen Granule Mast cell
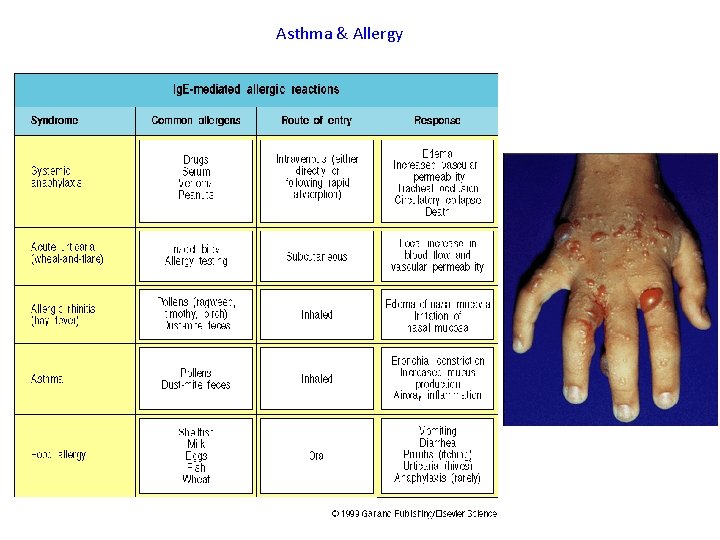
Asthma & Allergy
Origin of Self-Tolerance-autoimmune diseases Ø Antigen receptors are generated by random rearrangement of DNA. Ø Lymphocytes are tested for self-reactivity. Ø B and T cells with receptors specific for self antigens are destroyed by apoptosis, or programmed cell death or rendered nonfunctional. Ø In autoimmune diseases, self-tolerance is broken and the immune system attacks antigens in the body. Ø Examples: systemic lupus erythematosus, rheumatoid arthritis, insulin-dependent diabetes mellitus, and multiple sclerosis.
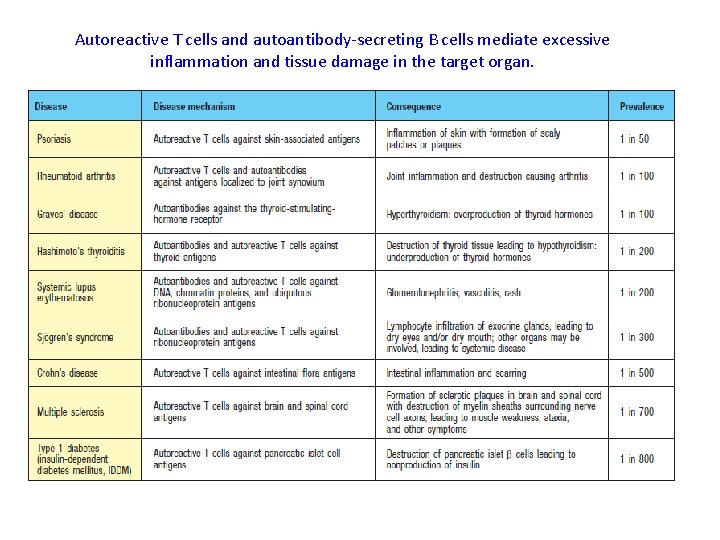
Autoreactive T cells and autoantibody-secreting B cells mediate excessive inflammation and tissue damage in the target organ.
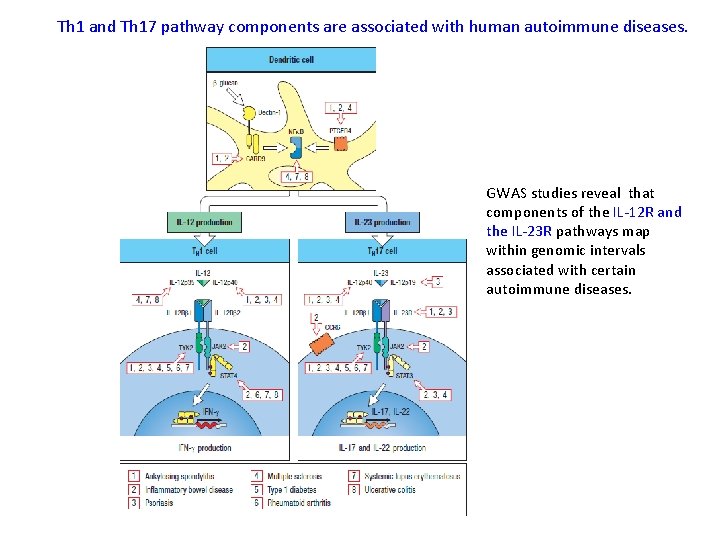
Th 1 and Th 17 pathway components are associated with human autoimmune diseases. GWAS studies reveal that components of the IL-12 R and the IL-23 R pathways map within genomic intervals associated with certain autoimmune diseases.
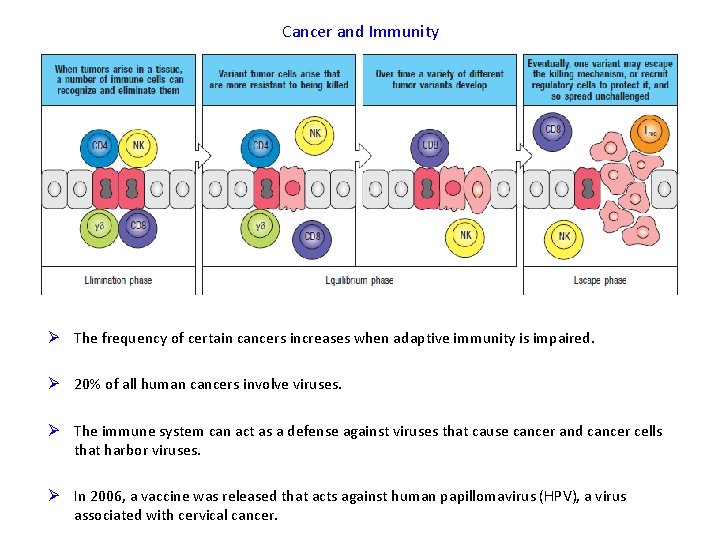
Cancer and Immunity Ø The frequency of certain cancers increases when adaptive immunity is impaired. Ø 20% of all human cancers involve viruses. Ø The immune system can act as a defense against viruses that cause cancer and cancer cells that harbor viruses. Ø In 2006, a vaccine was released that acts against human papillomavirus (HPV), a virus associated with cervical cancer.
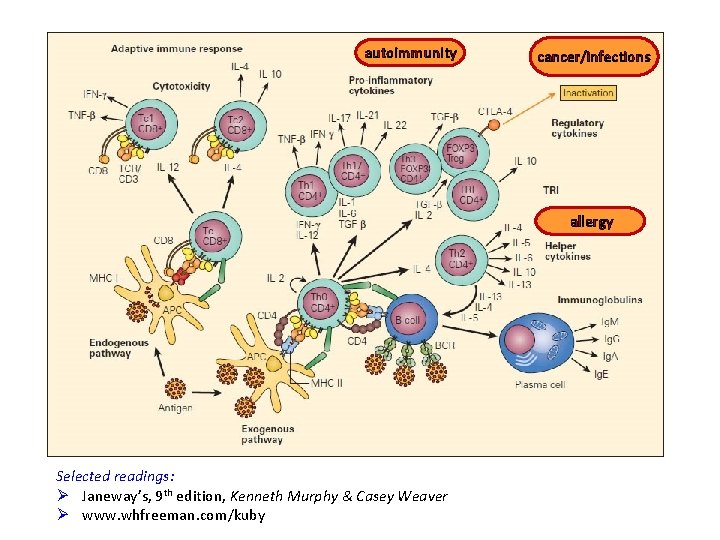
autoimmunity cancer/infections allergy Selected readings: Ø Janeway’s, 9 th edition, Kenneth Murphy & Casey Weaver Ø www. whfreeman. com/kuby
- Slides: 52