Systems and Scale How systems depend on CARBON

Systems and Scale How systems depend on CARBON and CHEMICAL ENERGY

Driving question Why do some things burn and other things do not burn? Why does ethanol behave more like wood than water?

Why does ethanol behave more like wood than water?
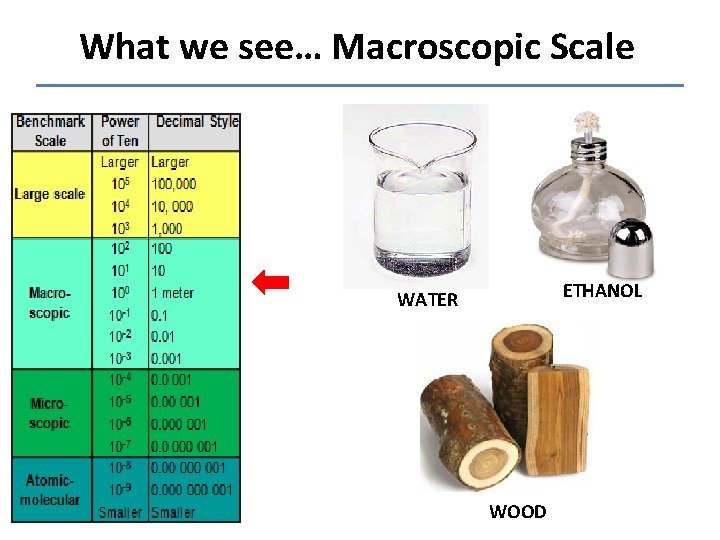
What we see… Macroscopic Scale ETHANOL WATER WOOD
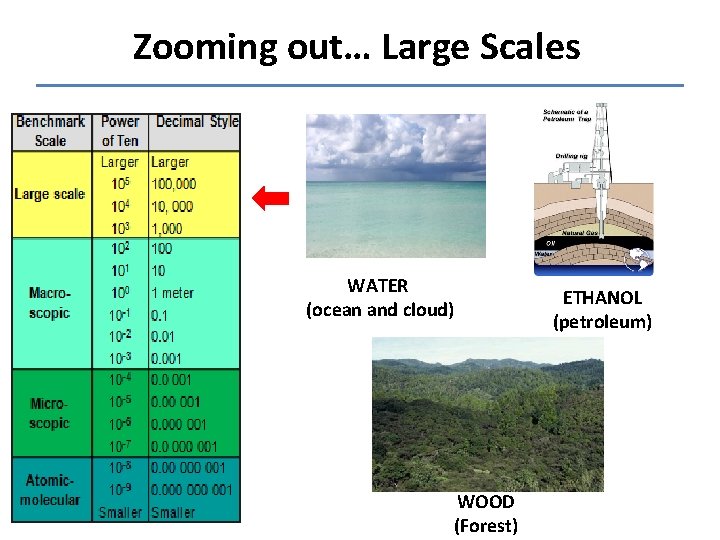
Zooming out… Large Scales WATER (ocean and cloud) WOOD (Forest) ETHANOL (petroleum)

Zooming in… Microscopic Scale WATER (a single droplet) WOOD CELLS ETHANOL (a single droplet)
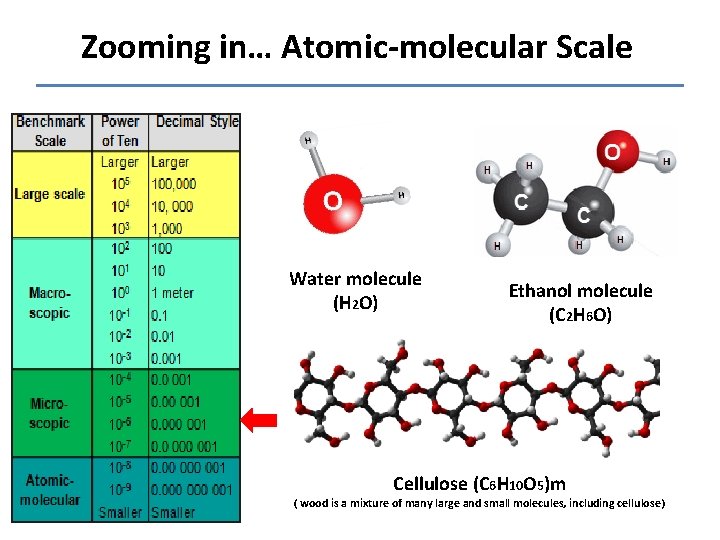
Zooming in… Atomic-molecular Scale Water molecule (H 2 O) Ethanol molecule (C 2 H 6 O) Cellulose (C 6 H 10 O 5)m ( wood is a mixture of many large and small molecules, including cellulose)
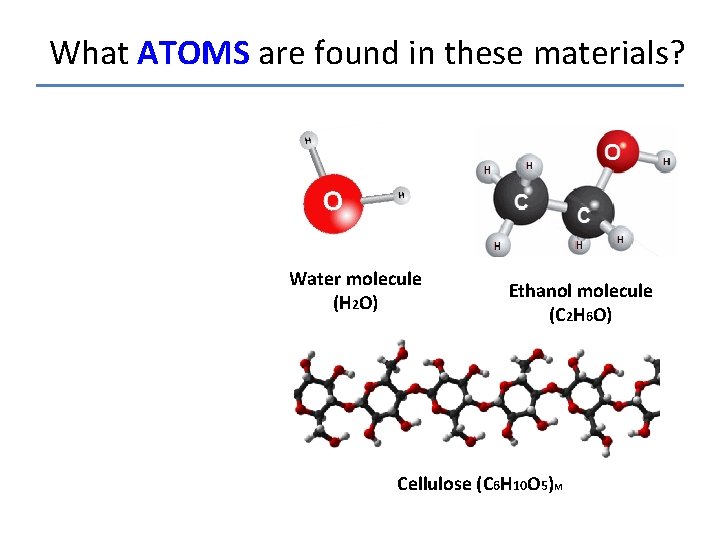
What ATOMS are found in these materials? Water molecule (H 2 O) Ethanol molecule (C 2 H 6 O) Cellulose (C 6 H 10 O 5)M
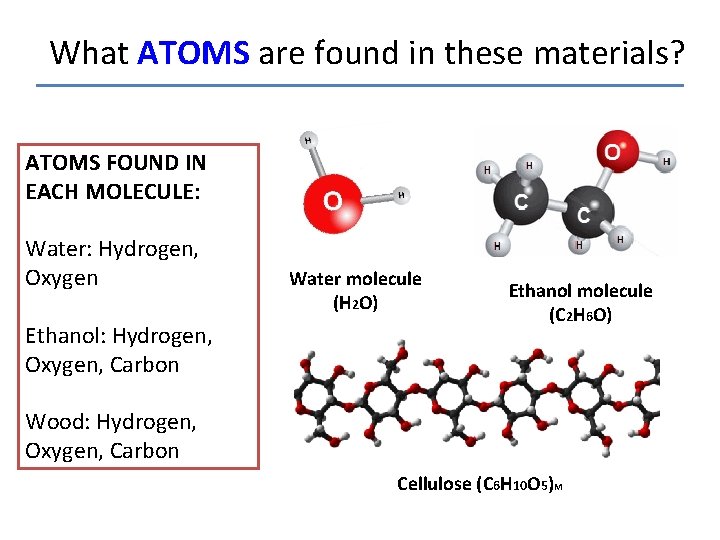
What ATOMS are found in these materials? ATOMS FOUND IN EACH MOLECULE: Water: Hydrogen, Oxygen Ethanol: Hydrogen, Oxygen, Carbon Water molecule (H 2 O) Ethanol molecule (C 2 H 6 O) Wood: Hydrogen, Oxygen, Carbon Cellulose (C 6 H 10 O 5)M
What BONDS are found in these materials? Water molecule (H 2 O) Ethanol molecule (C 2 H 6 O) Cellulose (C 6 H 10 O 5)M
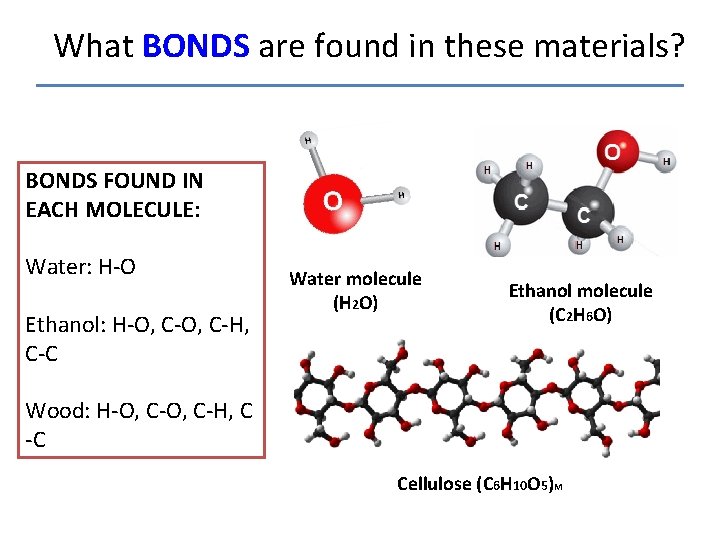
What BONDS are found in these materials? BONDS FOUND IN EACH MOLECULE: Water: H-O Ethanol: H-O, C-H, C-C Water molecule (H 2 O) Ethanol molecule (C 2 H 6 O) Wood: H-O, C-H, C -C Cellulose (C 6 H 10 O 5)M
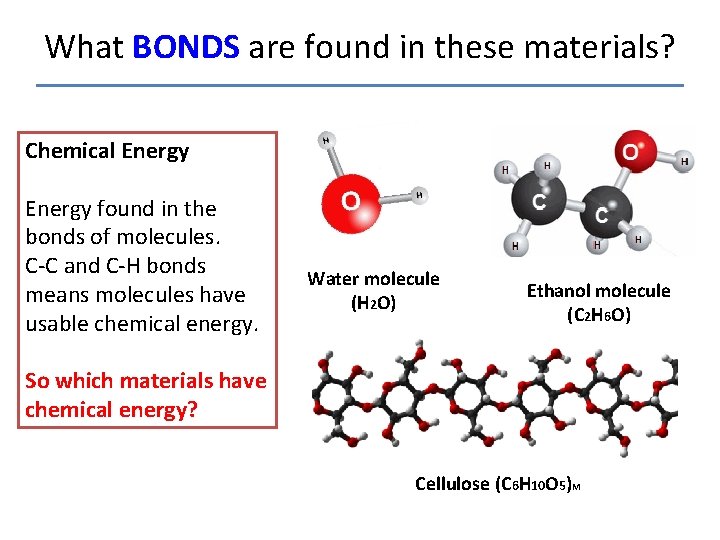
What BONDS are found in these materials? Chemical Energy found in the bonds of molecules. C-C and C-H bonds means molecules have usable chemical energy. Water molecule (H 2 O) Ethanol molecule (C 2 H 6 O) So which materials have chemical energy? Cellulose (C 6 H 10 O 5)M
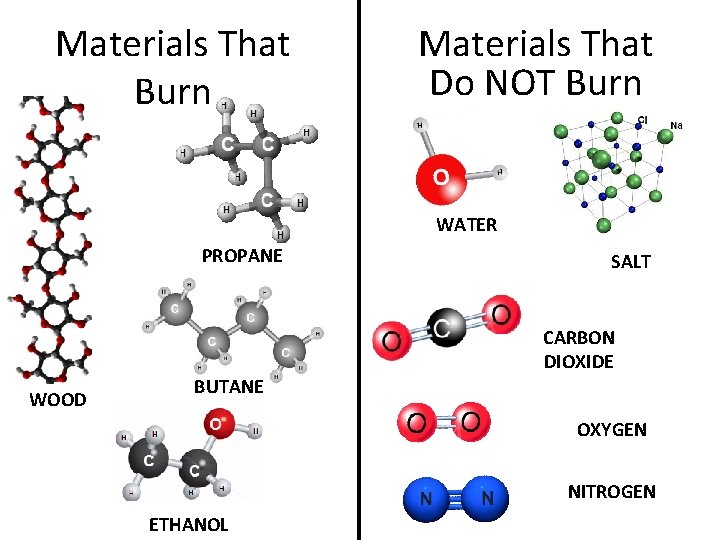
Materials That Burn Materials That Do NOT Burn WATER PROPANE SALT CARBON DIOXIDE WOOD BUTANE OXYGEN NITROGEN ETHANOL
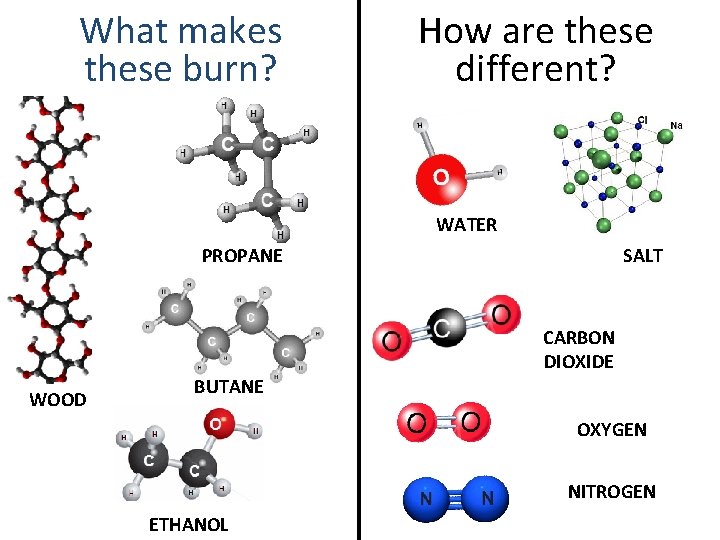
What makes these burn? How are these different? WATER PROPANE SALT CARBON DIOXIDE WOOD BUTANE OXYGEN NITROGEN ETHANOL

What makes something organic? ORGANIC AND INORGANIC MATERIALS
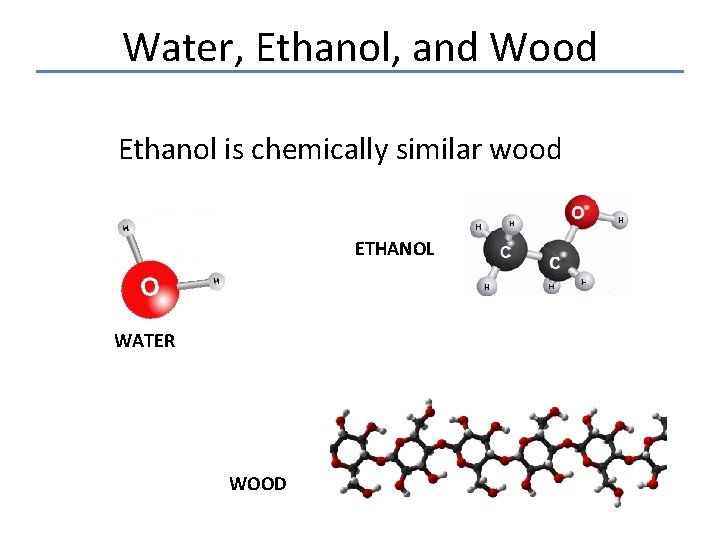
Water, Ethanol, and Wood Ethanol is chemically similar wood ETHANOL WATER WOOD
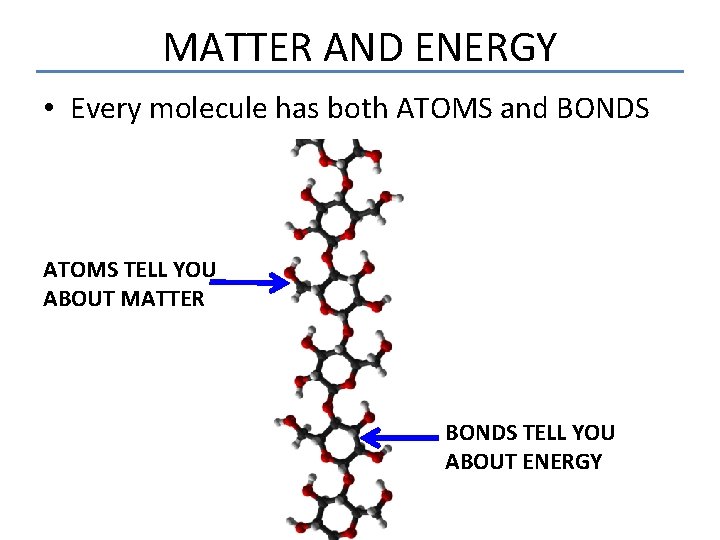
MATTER AND ENERGY • Every molecule has both ATOMS and BONDS ATOMS TELL YOU ABOUT MATTER BONDS TELL YOU ABOUT ENERGY
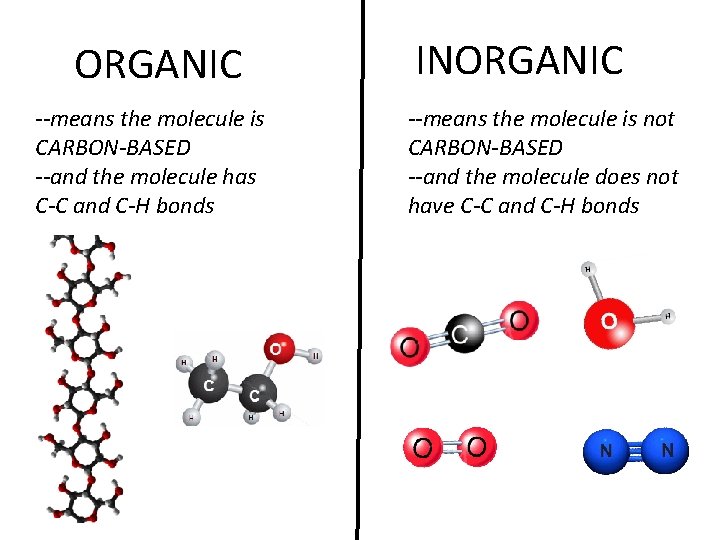
ORGANIC --means the molecule is CARBON-BASED --and the molecule has C-C and C-H bonds INORGANIC --means the molecule is not CARBON-BASED --and the molecule does not have C-C and C-H bonds
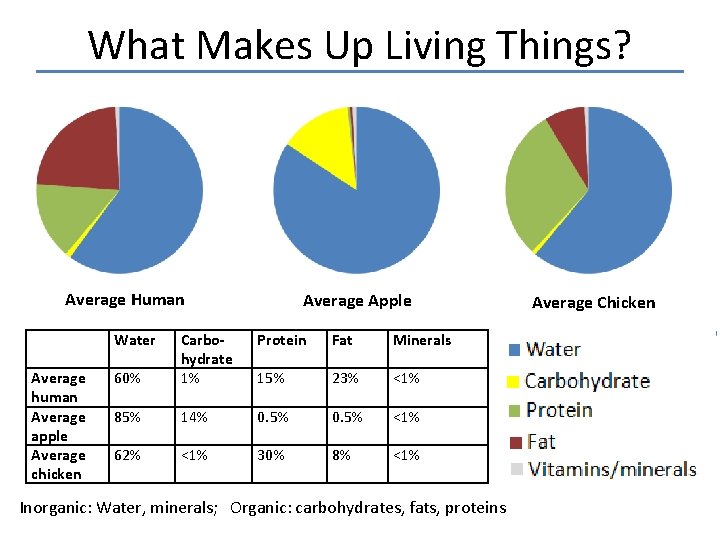
What Makes Up Living Things? Average Human Water Average human Average apple Average chicken Average Apple Protein Fat Minerals 60% Carbohydrate 1% 15% 23% <1% 85% 14% 0. 5% <1% 62% <1% 30% 8% <1% Inorganic: Water, minerals; Organic: carbohydrates, fats, proteins Average Chicken

BIOMASS • All the organic matter found in living and recently living organisms (this does not include water and minerals that are not incorporated into organic materials).
- Slides: 20