Systematic Description of Minerals Oxides Hydroxides Carbonates Phosphates

Systematic Description of Minerals Oxides, Hydroxides, Carbonates, Phosphates

Oxides Three main groups based on metal cation/oxygen ratios (impurities not shown) Hematite Group (X 2 O 3) most phases hexagonal Corundum X=Al+3 Hematite X=Fe+3 Ilmenite X= (Fe+2, Ti+4) solid solution) Rutile Group (XO 2) x- +4 cation most phases tetragonal Rutile X=Ti Pyrolusite X=Mn Cassiterite X=Sn Uraninite X=U Spinel Group (XY 2 O 4) X- +2 ; Y- +3 cation most phases isometric or orthorhombic Spinel X=Mg, Y=Al Magnetite X=Fe+2, Y=Fe+3 Chromite X=Fe+2, Y=Cr Chrysoberyl X=Be, Y=Al Ulvospinel X=Ti+4, Y=Fe+2
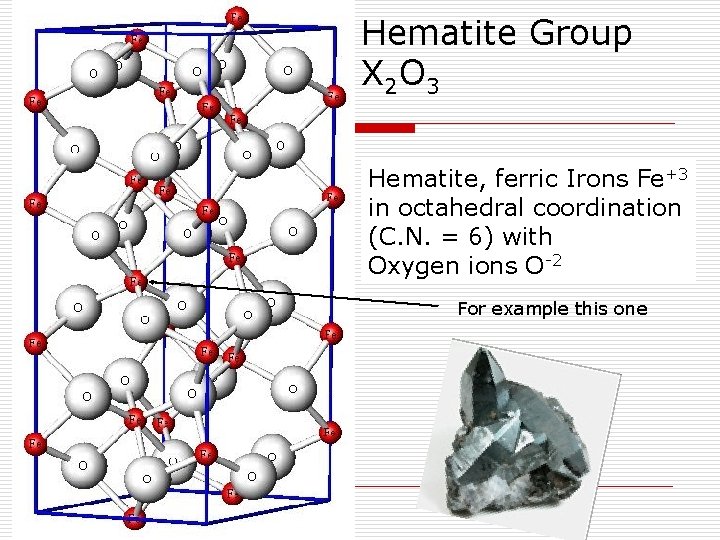
Hematite Group X 2 O 3 Hematite, ferric Irons Fe+3 in octahedral coordination (C. N. = 6) with Oxygen ions O-2 For example this one
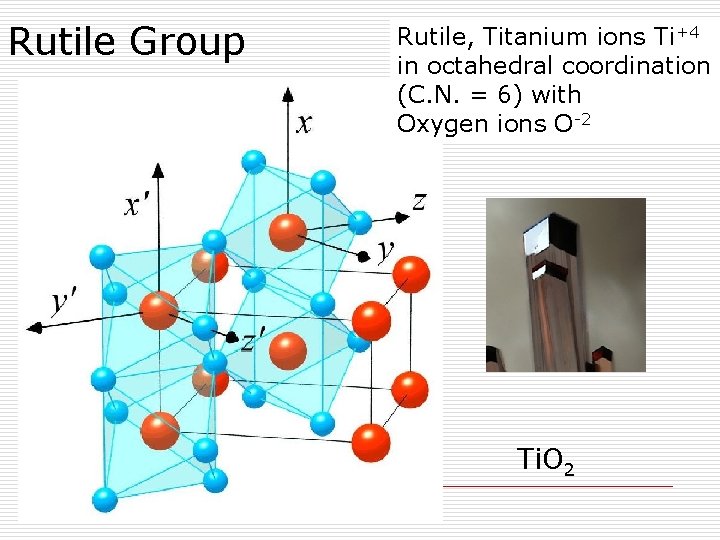
Rutile Group Rutile, Titanium ions Ti+4 in octahedral coordination (C. N. = 6) with Oxygen ions O-2 Ti. O 2

Fe-Ti oxides Anatase Fe+2 Fe+3
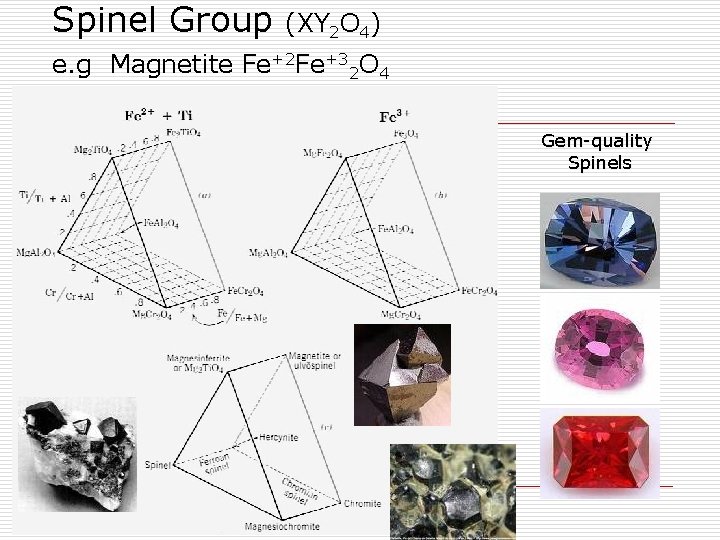
Spinel Group (XY 2 O 4) e. g Magnetite Fe+2 Fe+32 O 4 Gem-quality Spinels
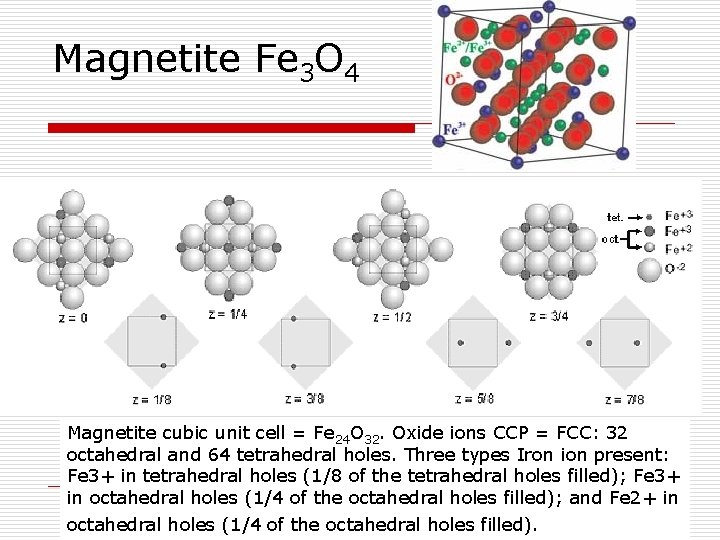
Magnetite Fe 3 O 4 Magnetite cubic unit cell = Fe 24 O 32. Oxide ions CCP = FCC: 32 octahedral and 64 tetrahedral holes. Three types Iron ion present: Fe 3+ in tetrahedral holes (1/8 of the tetrahedral holes filled); Fe 3+ in octahedral holes (1/4 of the octahedral holes filled); and Fe 2+ in octahedral holes (1/4 of the octahedral holes filled).

Other Common Oxides in color Corundum (Al 2 O 3) Rutile (Ti. O 2) Hardness=9 Rutile needles in Quartz Pyrolusite (Mn. O 2) Blue = Sapphire Red = Ruby Chrysoberyl (Be. Al 2 O 4) Mass of botryoidal Pyrolusite dendrites on surface Cyclic twinning in Chrysoberyl
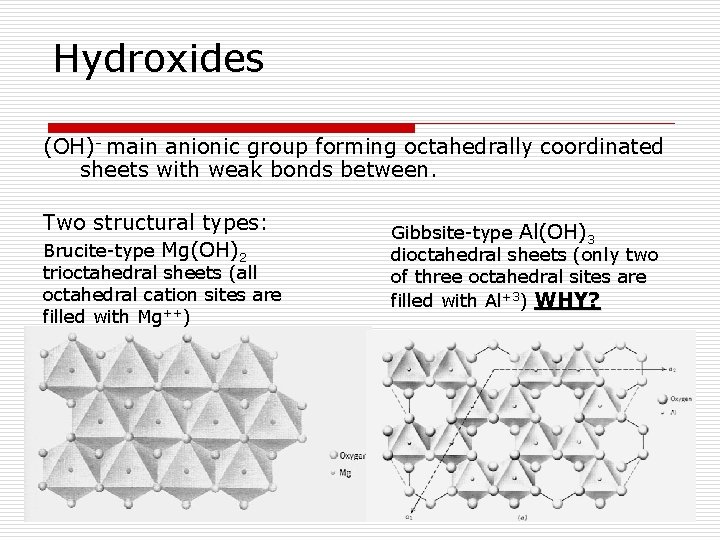
Hydroxides (OH)- main anionic group forming octahedrally coordinated sheets with weak bonds between. Two structural types: Brucite-type Mg(OH)2 trioctahedral sheets (all octahedral cation sites are filled with Mg++) Gibbsite-type Al(OH)3 dioctahedral sheets (only two of three octahedral sites are filled with Al+3) WHY?
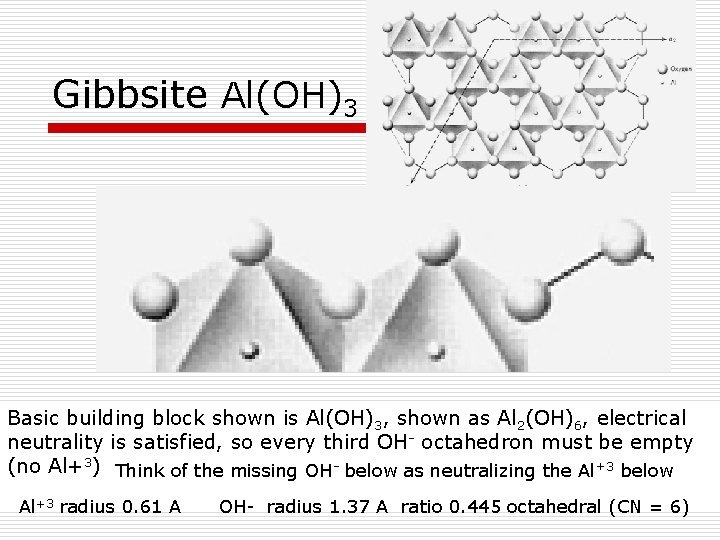
Gibbsite Al(OH)3 Basic building block shown is Al(OH)3, shown as Al 2(OH)6, electrical neutrality is satisfied, so every third OH- octahedron must be empty (no Al+3) Think of the missing OH- below as neutralizing the Al+3 below Al+3 radius 0. 61 A OH- radius 1. 37 A ratio 0. 445 octahedral (CN = 6)

Common Types of Hydroxides Brucite Mg(OH)2 Gibbsite Al(OH)3 Manganite Mn. O(OH) Diaspore Al. O(OH) Goethite Fe. O(OH) Bauxite Al-hydroxide* Pronunciations: Gear-tight Go-eth-thite Gurrr-tite Seem to vary with region *mixture of diaspore, gibbsite, and boehmite ( Al. O(OH))

Carbonates Reason for electrostatic valency calculations: Amount of residual charge indicates relative strength of bonds with cations, which are reflected in the hardness of the mineral

Carbonates Aragonite (High-P) Orthorhombic Calcite (Low-P - Hexagonal) Most are Hexagonal
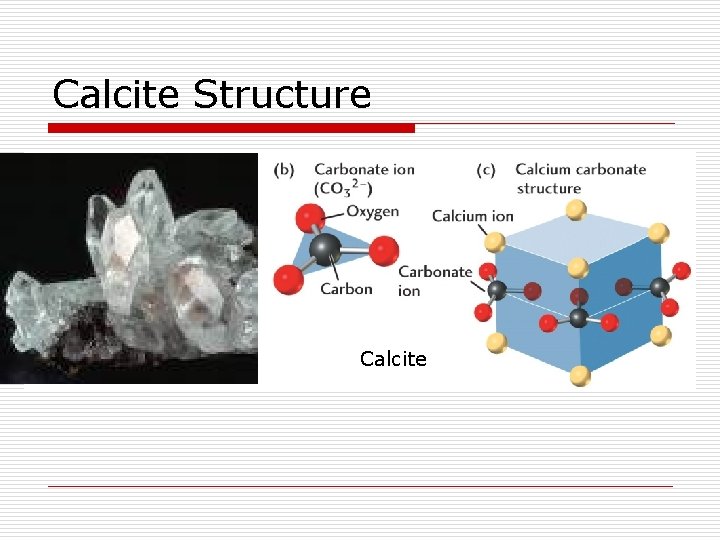
Calcite Structure Calcite
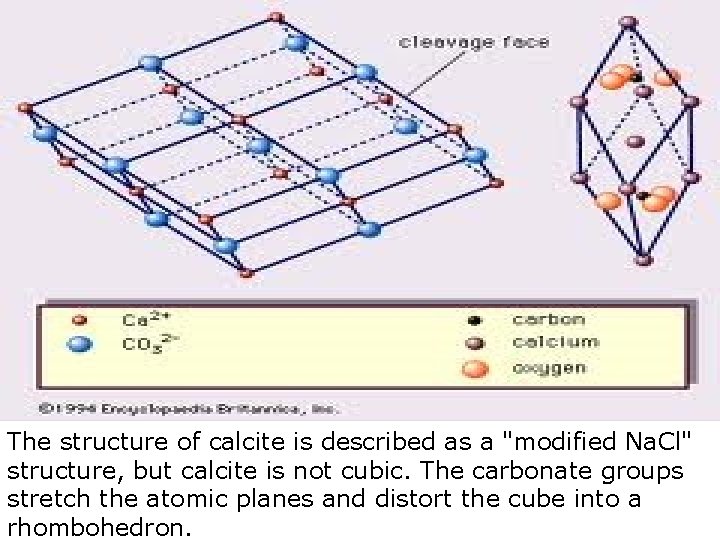
The structure of calcite is described as a "modified Na. Cl" structure, but calcite is not cubic. The carbonate groups stretch the atomic planes and distort the cube into a rhombohedron.

Aragonite Group Carbonate minerals with a single divalent cation of radius > 1. 00 Å. With increasing radius the species are Aragonite (Ca. CO 3), Strontianite (Sr. CO 3), Cerussite (Pb. CO 3), and Witherite (Ba. CO 3). Aragonite is denser than calcite, and is the high P polymorph. It crystallizes at ambient conditions and persists metastably for millions of years. The orthorhombic structure is nearly hexagonal with c as the unique axis.

Dolomite Ca. Mg(CO 3)2 Forms during seasonal high tides that flood limestone (calcite) islands with seawater. Mg++ in the seawater replaces some of the Calcium ions. Has very distinctive crystals

Other Carbonates Azurite - Cu 3(CO 3)2(OH)2 (Blue) Malachite – Cu 2 CO 3(OH)2 (Green) Rhodochrosite – Mn. CO 3

Borates Kernite – Na 2 B 4 O 6(OH)2· 3 H 2 O H – 3; SG – 1. 95 Borax - Na 2 B 4 O 5(OH)4· 8 H 2 O H – 2 -2. 5; SG – 1. 7 Ulexite – Na. Ca. B 5 O 6(OH)6· 5 H 2 O H – 1 -2. 5; SG – 1. 96 “Television Rock”

Tungstates & Molybdates Wolframite – (Fe, Mn)WO 4 SG: 7 -7. 5 Scheelite – Ca. WO 4 SG: ~6 Wulfenite – Pb. Mo. O 4 SG: 6. 8

Phosphates o Apatite – Ca 5(PO 4)3(F, Cl, OH) n prismatic hexagonal crystals n common in igneous rocks and hydrothermal deposits - variable colors “the deceiver”

Other Common Phosphates Monazite – (Ce, La, Y, Th)PO 4 Ore mineral for Rare Earth Elements Useful mineral in U-Pb and Th age dating Wavellite – Al 3(PO 4)2(OH)3· 5 H 2 O Radiating globular aggregates Turquoise – Cu. Al 6(PO 4)4(OH)8· 5 H 2 O

A rare Chromate: Crocoite Pb. Cr. O 4 Monoclinic 2/m. Commonly in prismatic crystals, vertically striated b=102 o 33’ Cleavage {110} perfect H 2. 5 – 3 G 5. 9 -6. 1 Luster Adamantine Color bright red to orange- red Streak orange-yellow
- Slides: 23