Synthetic and Biological Polymers Macromolecules formed by the

Synthetic and Biological Polymers: Macromolecules formed by the covalent attachment of a set of small molecules termed monomers. Polymers are classified as: (1) Man-made or synthetic polymers that are synthesized in the laboratory; (2) Biological polymer that are found in nature. Synthetic polymers: nylon, poly-ethylene, poly-styrene Biological polymers: DNA, proteins, carbohydrates 1

Hydrocarbons ex: Alkanes 1 – Meth 2 – Eth 3 – Prop 4 – But 5 – Pent 6 – Hex 7 – Hept 8 – Oct 9 – Non 10 – Dec 11 – Undec 12 – Dodec

Hydrocarbons at Room Temperature Gas Methane Ethane Propane Butane Liquid 5 to 19 Carbons Waxy 20 to 40 Carbons Plastic 40 or more Carbons

Melting Point As the length of hydrocarbons get longer, the Melting Point grows Higher. Why?

What other material properties change? Viscosity Hardness Toughness Flammability
Bonding Covalent Ionic (Na. Cl) Polar (H 2 O) Van der Waals

Methods for making polymers Addition polymerization and condensation polymerization Addition polymerization: monomers react to form a polymer without net loss of atoms. Most common form: free radical chain reaction of ethylenes n monomers one polymer molecule 7
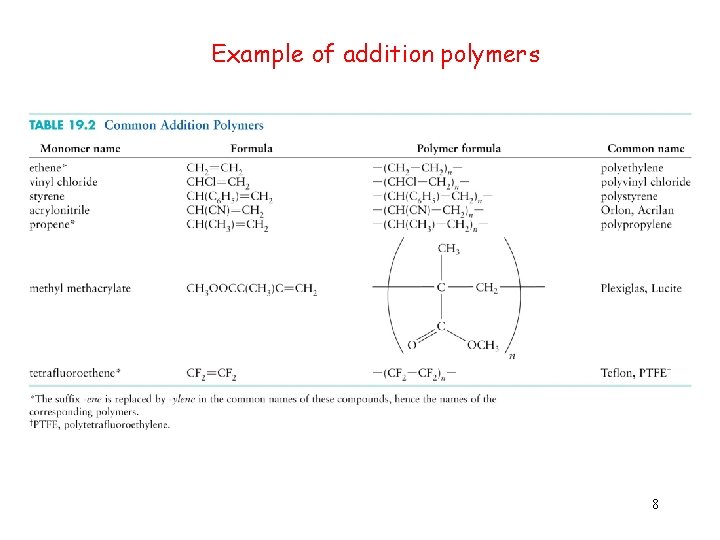
Example of addition polymers 8
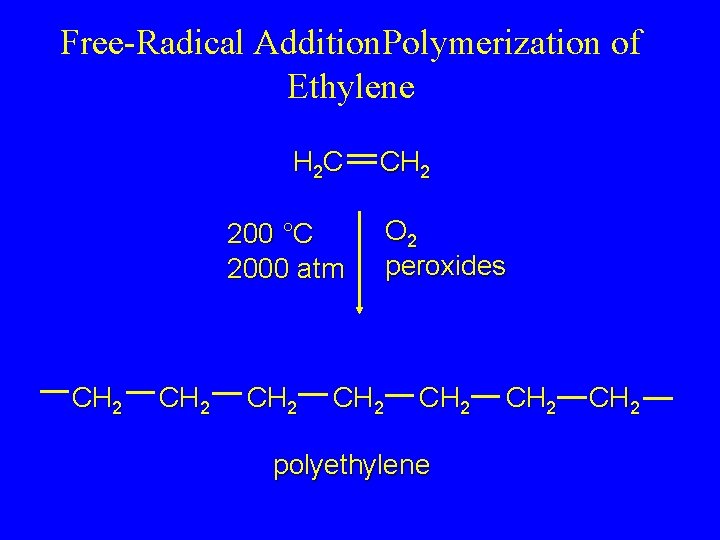
Free-Radical Addition. Polymerization of Ethylene H 2 C CH 2 200 °C 2000 atm CH 2 O 2 peroxides CH 2 polyethylene CH 2

Free-Radical Polymerization of Propene H 2 C CH CH CH 3 CHCH 3 CH CH CH 3 CH 3 polypropylene
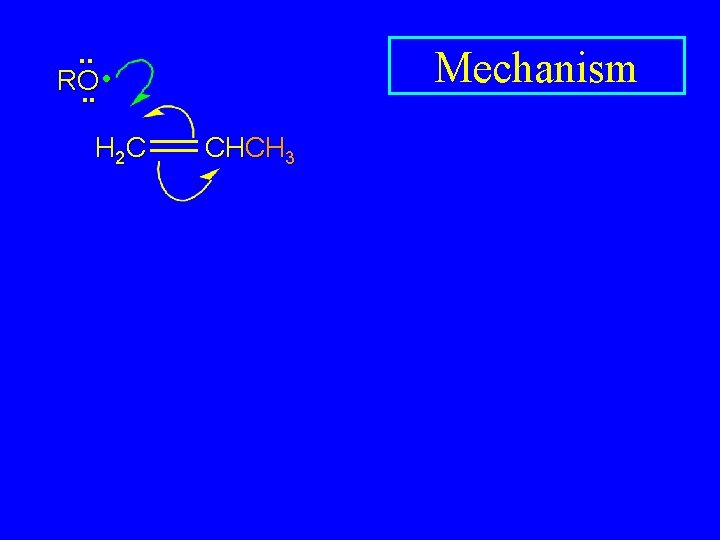
. . • RO. . H 2 C Mechanism CHCH 3
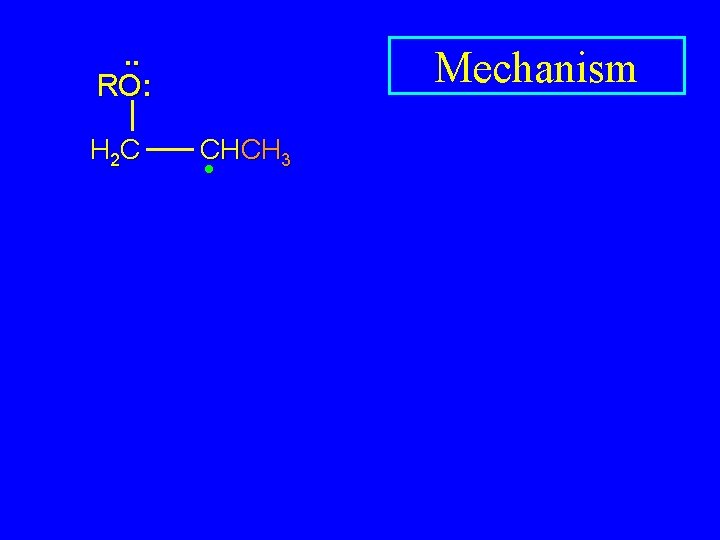
. . RO: H 2 C Mechanism CHCH 3 •
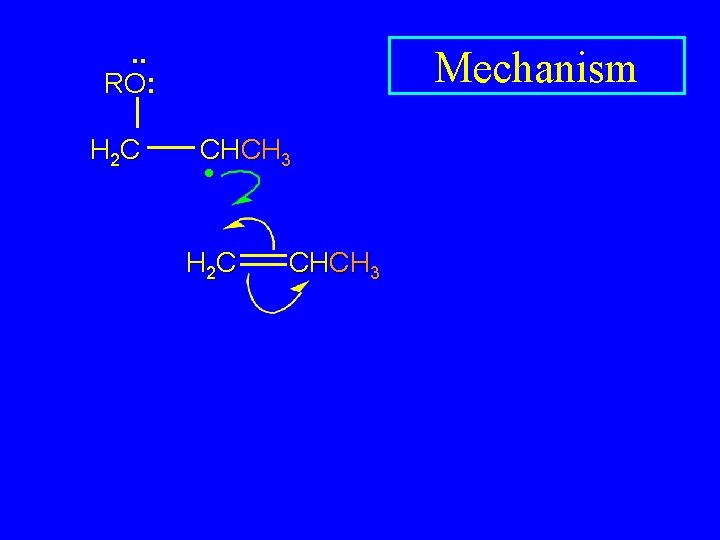
. . RO: H 2 C Mechanism CHCH 3 • H 2 C CHCH 3
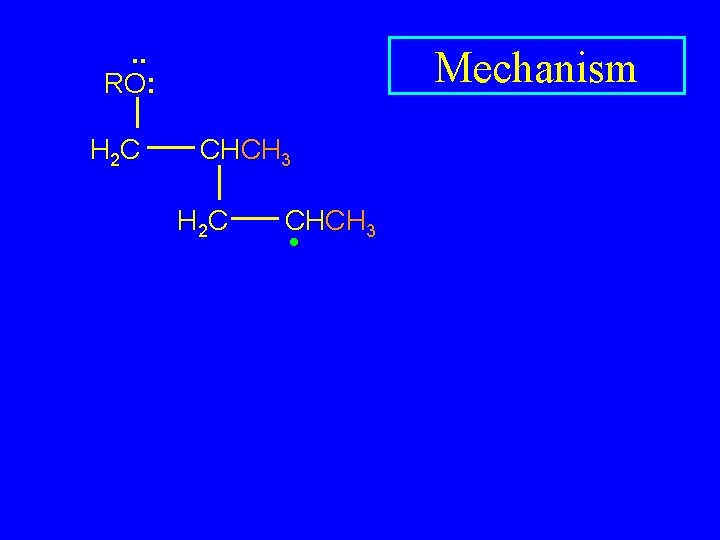
. . RO: H 2 C Mechanism CHCH 3 H 2 C CHCH 3 •
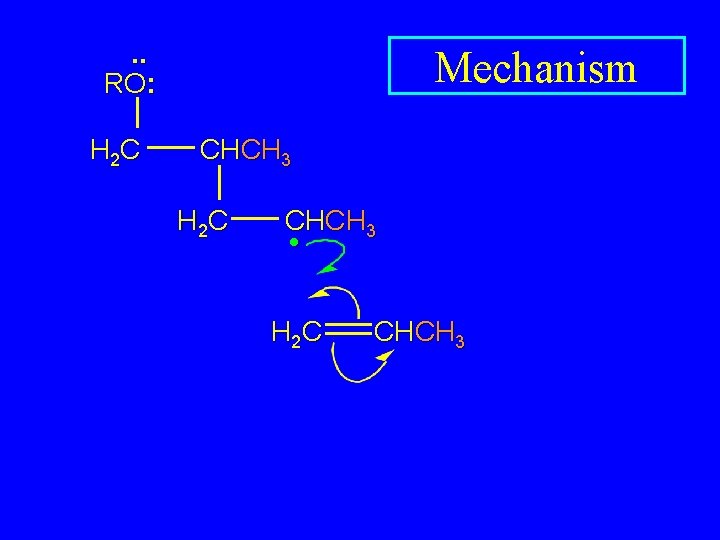
. . RO: H 2 C Mechanism CHCH 3 H 2 C CHCH 3 • H 2 C CHCH 3
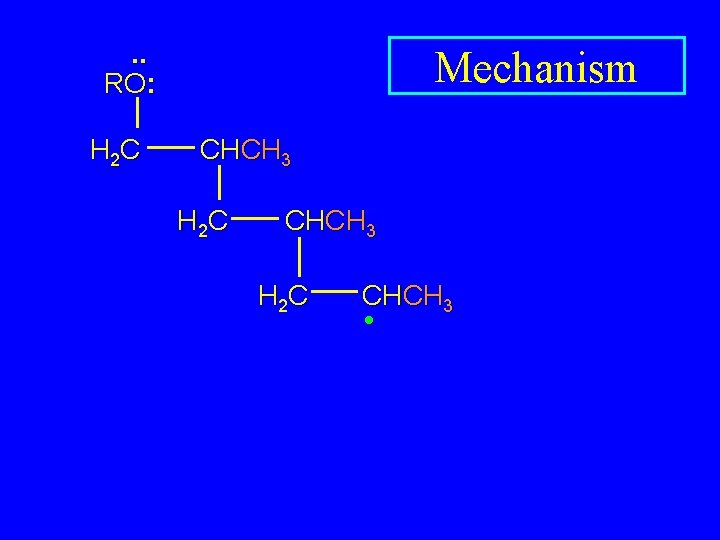
. . RO: H 2 C Mechanism CHCH 3 H 2 C CHCH 3 •
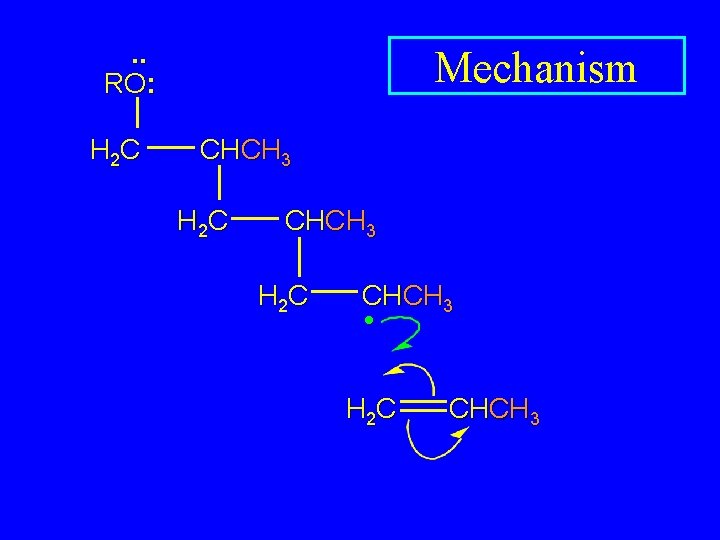
. . RO: H 2 C Mechanism CHCH 3 H 2 C CHCH 3 • H 2 C CHCH 3
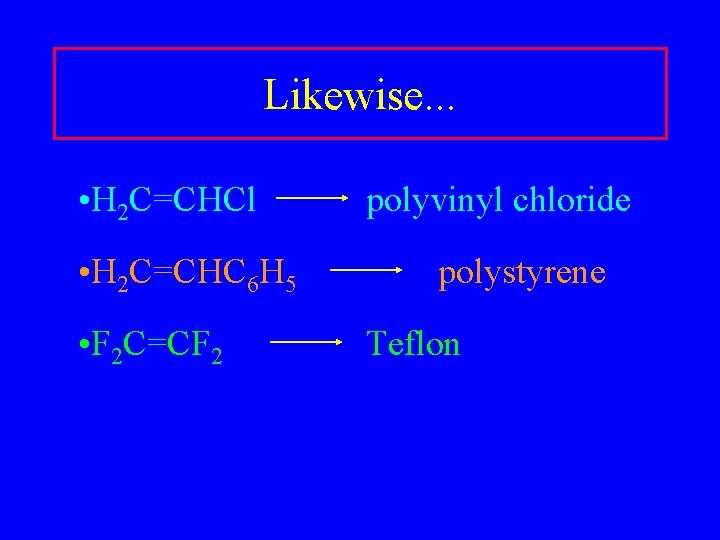
Likewise. . . • H 2 C=CHCl • H 2 C=CHC 6 H 5 • F 2 C=CF 2 polyvinyl chloride polystyrene Teflon
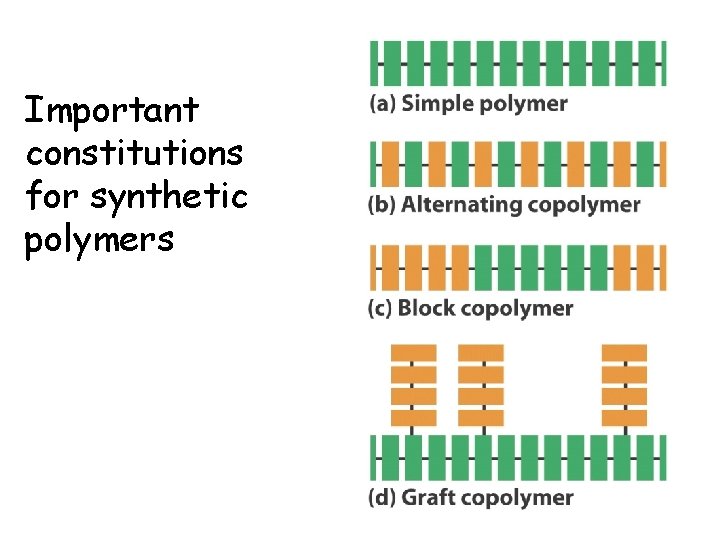
Important constitutions for synthetic polymers 19
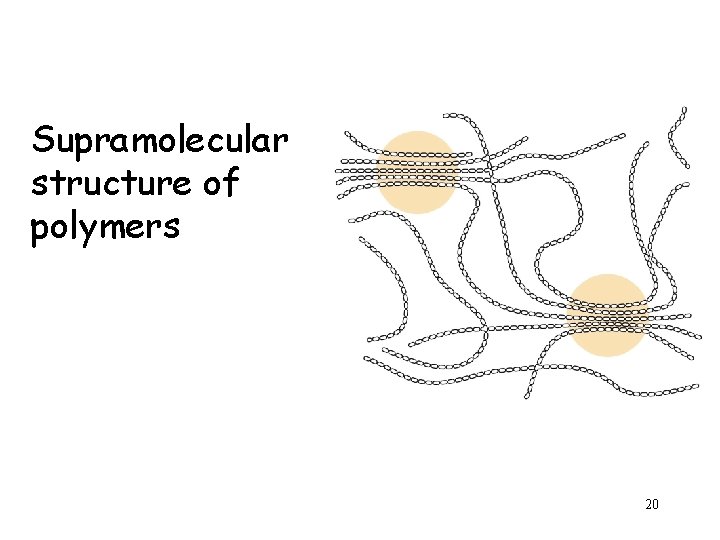
Supramolecular structure of polymers 20

Structural properties of linear polymers: conformational flexibility and strength 21
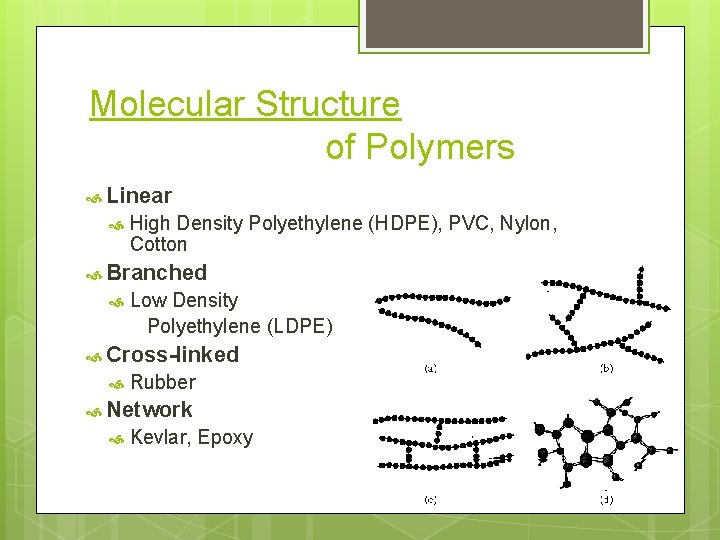
Molecular Structure of Polymers Linear High Density Polyethylene (HDPE), PVC, Nylon, Cotton Branched Low Density Polyethylene (LDPE) Cross-linked Rubber Network Kevlar, Epoxy

Chain Length: 1000 - 2000 Low-Density Polyethylene (LDPE)

Chain Length: 4, 000 – 5, 000 PVC – (polyvinyl chloride) More Polar Stronger Bonding

Chain Length: 10, 000 – 100, 000 High-Density Polyethylene (HDPE)

Chain Length: 2 -6 million Ultra-high-molecular-weight polyethylene (UHMWPE) Joint Replacement Helmet Gears

Rubber Tree Sap: Sticky Viscous Gooey Goodyear Experiment Luck Profit ($0)

Vulcanization
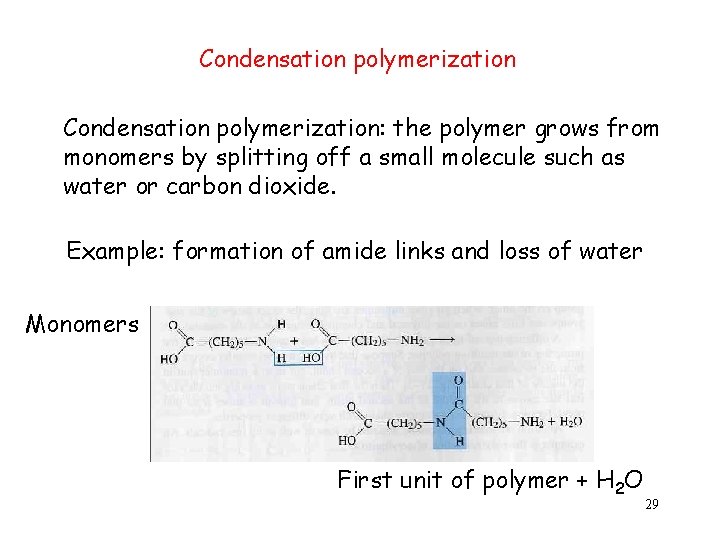
Condensation polymerization: the polymer grows from monomers by splitting off a small molecule such as water or carbon dioxide. Example: formation of amide links and loss of water Monomers First unit of polymer + H 2 O 29
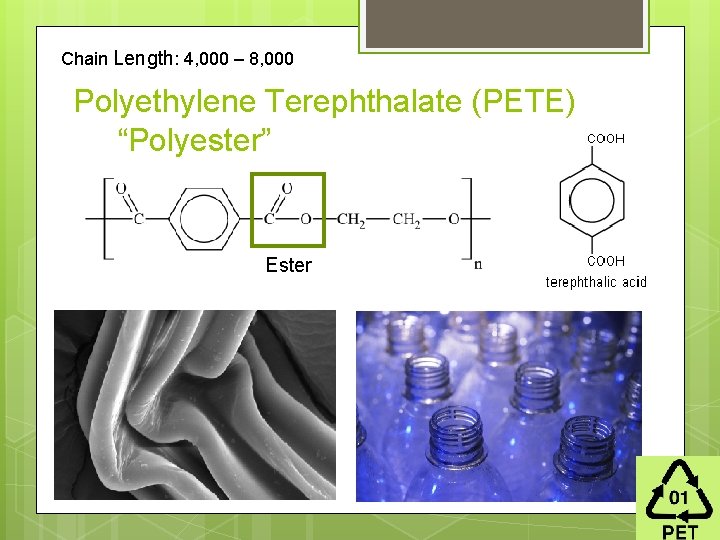
Chain Length: 4, 000 – 8, 000 Polyethylene Terephthalate (PETE) “Polyester” Ester

Kevlar Strong Network of Covalent Bonds And Polar Hydrogen Bonds
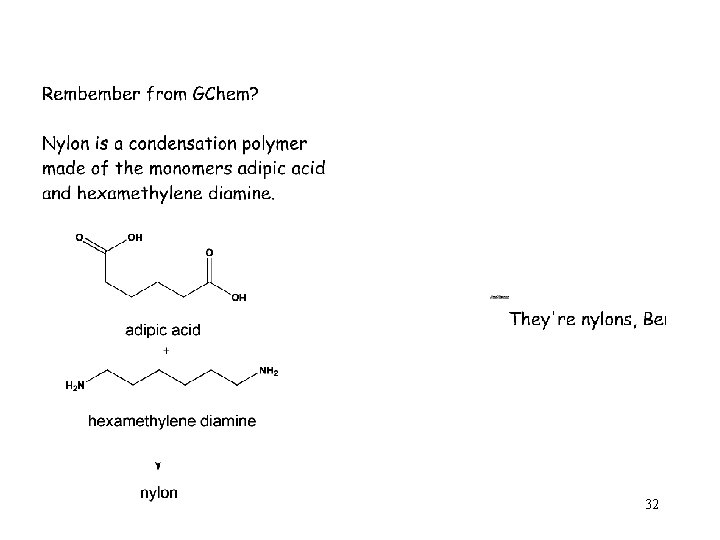
32

Nylon
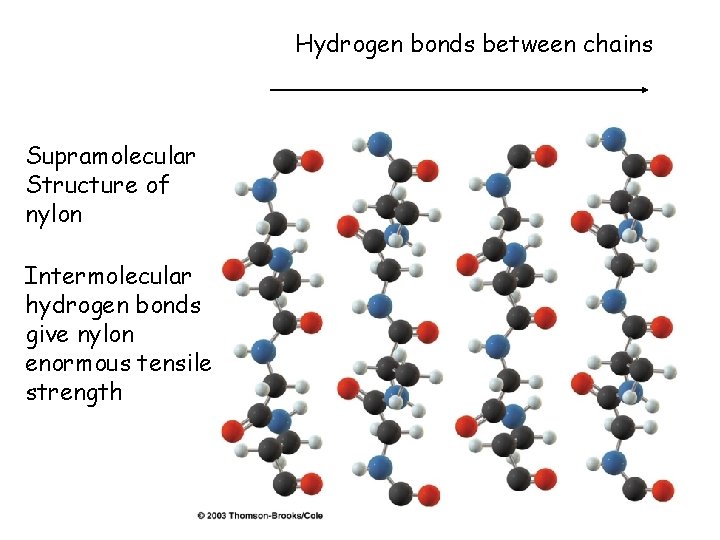
Hydrogen bonds between chains Supramolecular Structure of nylon Intermolecular hydrogen bonds give nylon enormous tensile strength 34

Biopolymers Nucleic acid polymers (DNA, RNA) Amino acids polymers (Proteins) Sugar polymers (Carbohydrates) Genetic information for the cell: DNA Structural strength and catalysis: Proteins Energy source: Carbohydrates 35
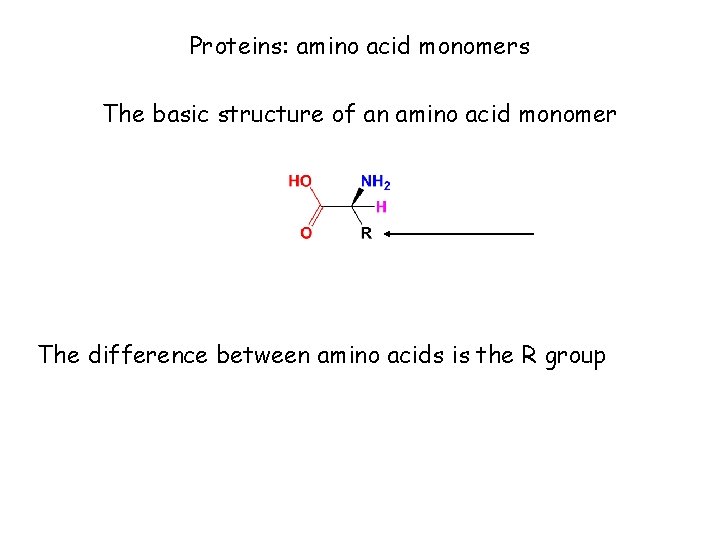
Proteins: amino acid monomers The basic structure of an amino acid monomer The difference between amino acids is the R group

Cotton Long Strands of Cellulose + Hydrogen Bonds Cellulose is the most common organic material on earth! It is also a primary constituent of wood and paper.

Polymers in Biology Starch DNA Sugar Proteins
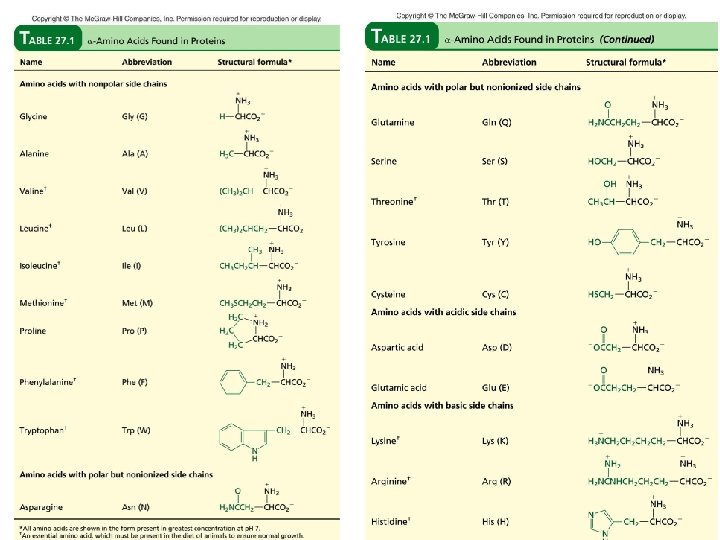
39

Proteins: condensation polymers Formed by condensation polymerization of amino acids Monomers: 20 essential amino acids General structure of an amino acid R is the only variable group Glycine (R = H) + Glycine First step toward poly(glycine) 40
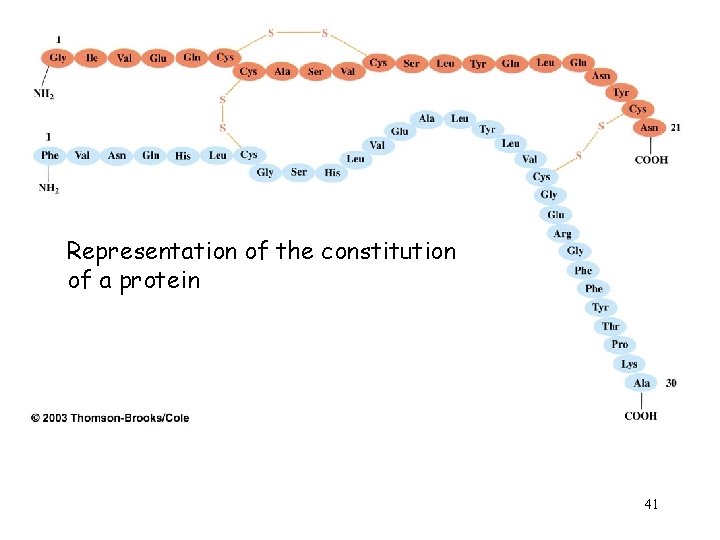
Representation of the constitution of a protein 41
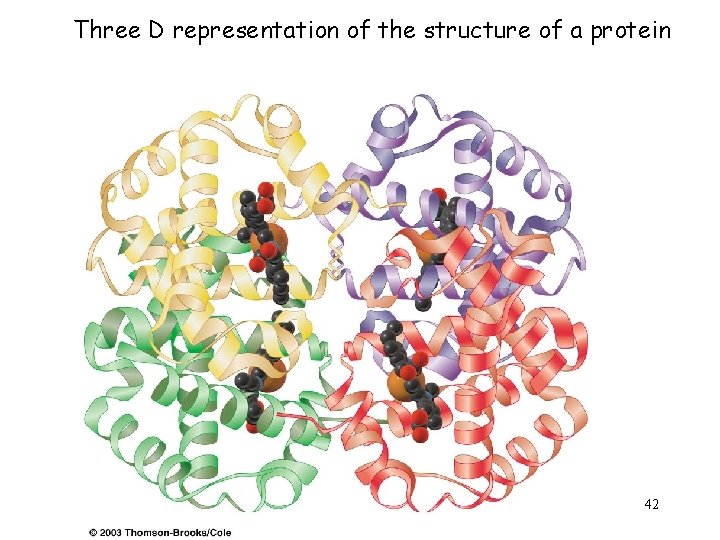
Three D representation of the structure of a protein 42

DNA
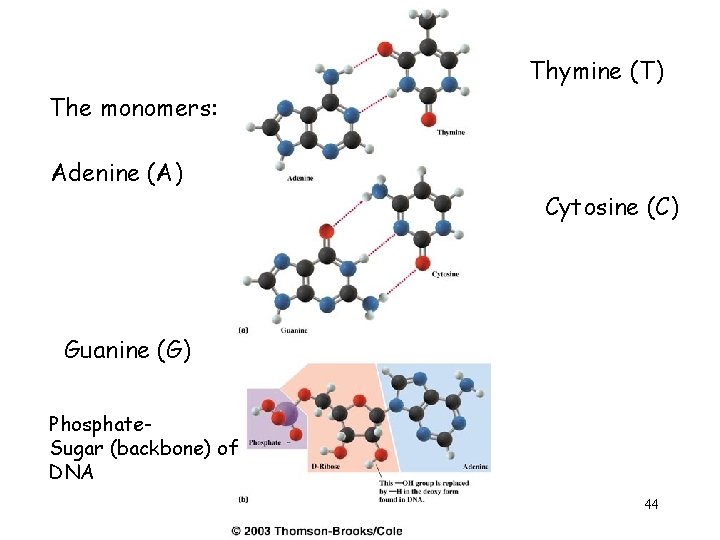
Thymine (T) The monomers: Adenine (A) Cytosine (C) Guanine (G) Phosphate. Sugar (backbone) of DNA 44
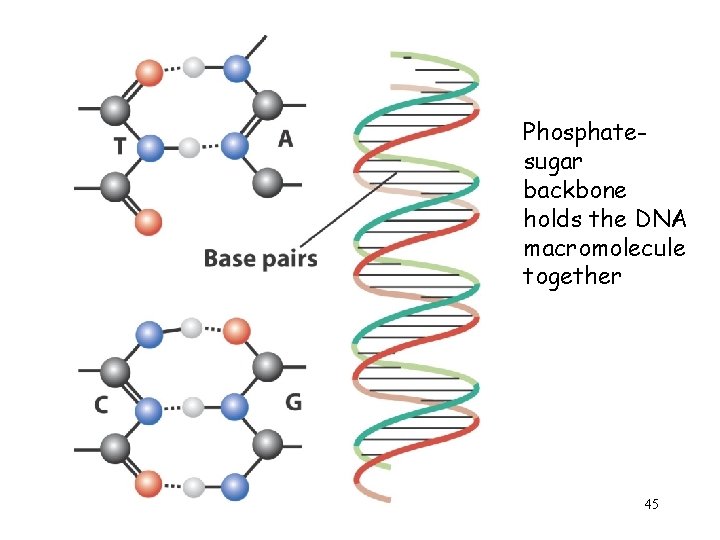
Phosphatesugar backbone holds the DNA macromolecule together 45
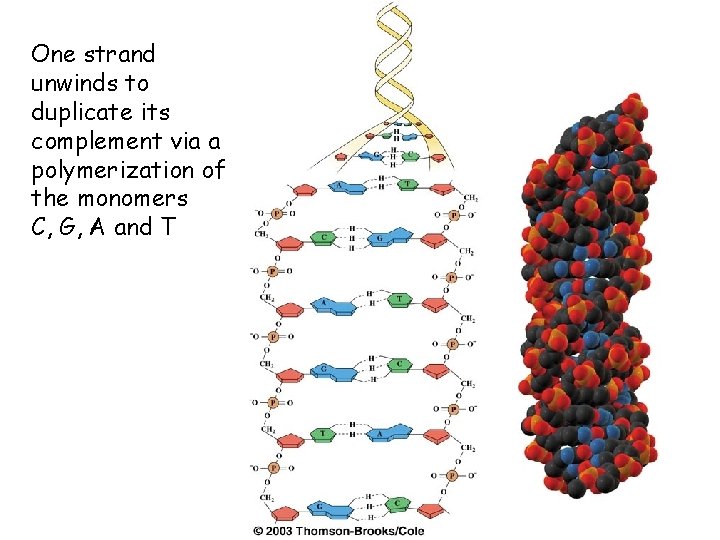
One strand unwinds to duplicate its complement via a polymerization of the monomers C, G, A and T 46

Carbohydrates
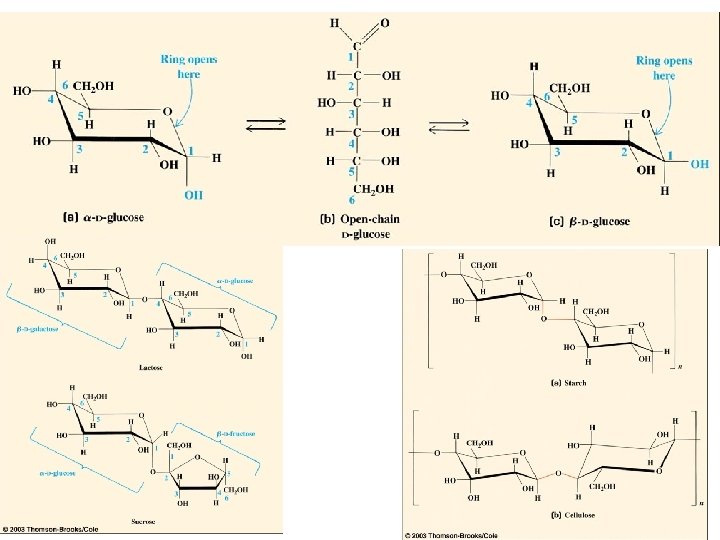
48
Endless Possibilities New Functional Groups Different Polymer Backbones

Conclusions: Polymers make up all sorts of materials that are all around us! They can have a huge range or material properties based on their: Functional Groups Structure Backbone Keep thinking about how chemical interactions on the nano-scale correspond to material properties on the macro-scale
- Slides: 50