Synthesis of Dalanine Cephalexin Condensed Drug Polymer Firyal











- Slides: 19

Synthesis of D-alanine Cephalexin Condensed Drug Polymer Firyal M. A. AL-Salami, * Abbas N. M. AL-Sharify , *. Khudheyer Jawad Kadem and *Eng. Qutada Abood Ahmed AL-Mustansiriyah University College of Science , Dep. of Chemistry * Babylon University *Ministry of water resources

• Abstract: • In this paper a new condensed drug polymer were synthesized from polycondensation of cephalexin acid chloride and D-alanine acid chloride producing polyamide as a bioactive and drug polymer. • The prepared peptide polymer was characterized by using 1 H-NMR , FTIR and UV. Spectroscopy. Intrinsic viscosity was 0. 5 dl/g. The swelling % was calculated for the polymer. The controlled release rates were measured in different p. H values at 370 C.

• Experimental • All chemical materials were purchased from Fluka. All available Chemical reagents were used without further purification. FTIR spectra were taken on (Fourier Transform Infrared spectraphotometer- shimadzu). C. H. N analysis were determined subsequently by C. H. N analyzer, model LECOGO, SC 132 respectively. • Electronic spectra measurements are using cintra-5 -UV. visible spectrophotometer. Intrinsic viscosity was measured by capillary viscometer type Ostwald viscometer at 30 0 C. • Polymer swelling % were determinate using different non solvents according to the following relationship: • S%=(M 1 -M 0)/M 0 x 100 • Where M 0 is the mass of dray polymer at time 0 • M 1 is the mass of swollen polymer at time t

• Conversion of D-alanine or Cephalexin to acidchloride derivatives • In a round bottom flask provided with magnatic bar was placed dissolved D-alanine in dioxane, and the stochiometric amount of thionylchloride was added drop wise at 00 C. The mixture was stirred 20 min. The product was isolated and washed by ether and dried. • The same conditions was used with cephalexin to convert to acidchloride derivative.

• Polycondensation of D-alanine acidchloride and cephalexine acidchloride • A 100 ml round-bottomed flask equipped with a magnetic stirrer, athermometer and reflux condenser was charged with 10 ml of dioxane, ( 0. 1 mole) Dalanine and (0. 1 mole). The mixture was stirred and refluxed for about 3 h. and homogenous solution was achieved. The mixture was cooled at room temperature, and the precipitate condensed polymer was collected. The produced polymer was rapidly washed with alcohol and dried at 50 0 C in vacuum overnight, the yield was 90%. with ηin =0. 5.

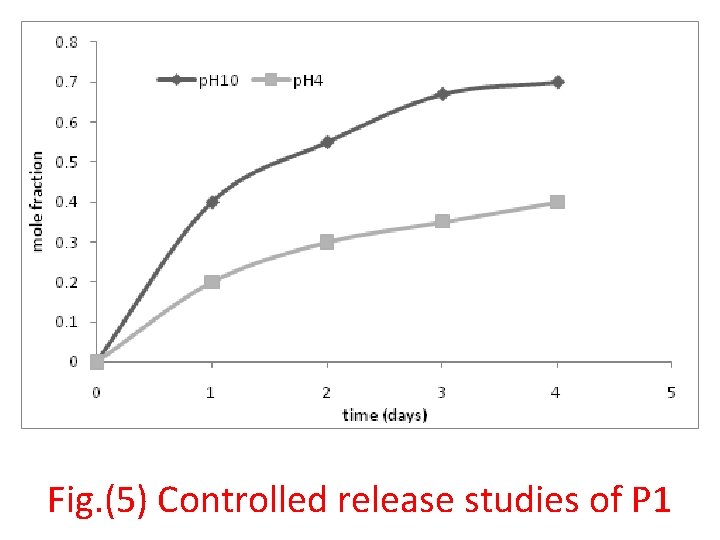
• Controlled drug release studies • 50 mg was placed in 100 ml of buffer solution with p. H 4 and 10 at 37 0 C. At periodic intervals 3 ml of solution containg drug polymer withdrawing and tested at λmax 290 nm using UVVIS spectrophotometer. The release media were changed periodically with fresh KCl-HCl solution. The release studies were continued until the absorbance of the final solution was zero. The amount of released cephalexine was quantified using appropriate calibration curve.

• Results and Discussion • The objective of this research work was to incorporating cephalexine and D-alanine through backbone of condensed polymer containing amide groups. • In basic idea behind the development of such a system is to maintain a constant level of drug in the blood plasma in spite of the fact that the drug does not undergo disintegration [15].

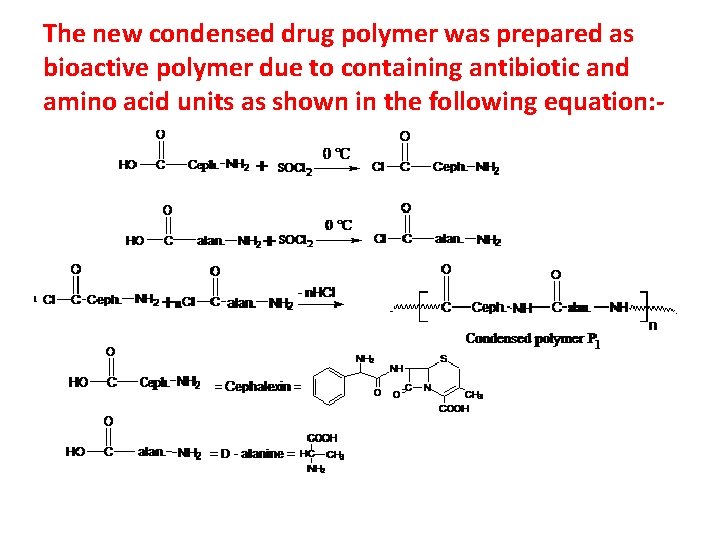
The new condensed drug polymer was prepared as bioactive polymer due to containing antibiotic and amino acid units as shown in the following equation: -

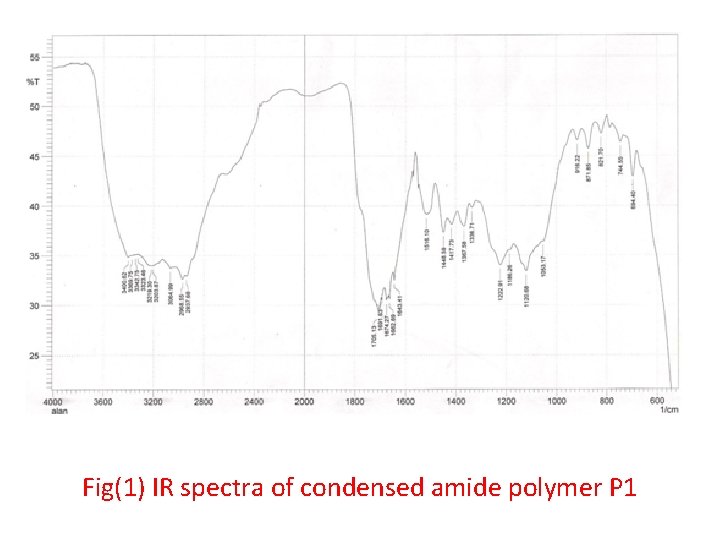
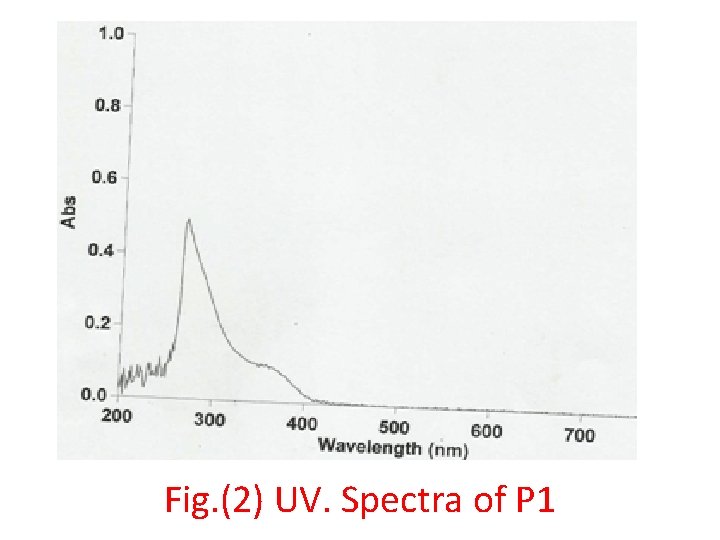
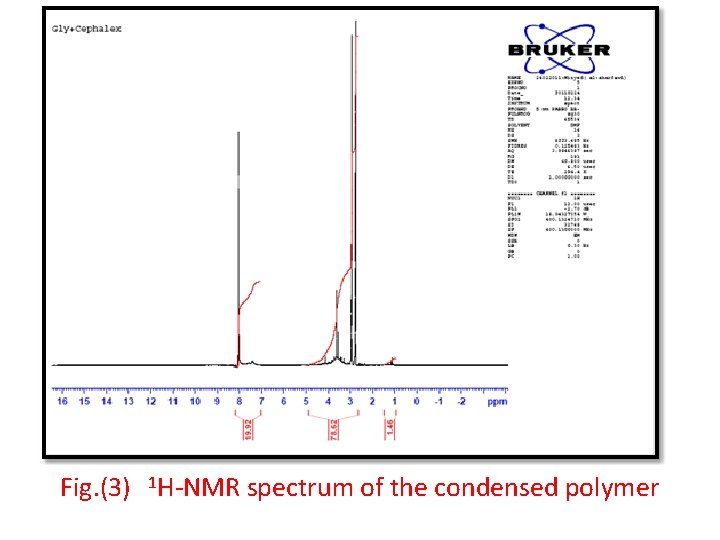
• This amide polymer P 1 was characterized by FTIR spectroscopy, Fig. (1) shows the absorption at 3200 cm-1 for ν-NH group and 1656 cm-1 due to ν-C=O amide and around 2950 -2860 cm-1 due to νC-H aliphatic and the other at 3100 cm-1 which attributed to ν- C-H aromatic and ν-C=O acid at 1724 cm-1. • Fig. (2) of UV. Spectra shows the two peaks at λmax(360, 260)nm due to (n-π*) and (π-π*) transitions. • Fig. (3) of the 1 H-NMR spectra of the prepared polymer shows the chemical shifts as shown follow: • δ( 1. 2 ) ppm (CH 2, CH 3) • δ( 8. 3 ) ppm ( for different environment aromatic protons) • δ( 3. 5 ) ppm ( for HN-C=O amide) • δ( 2. 7 ) ppm (CH neighboring amide and carbonyl )

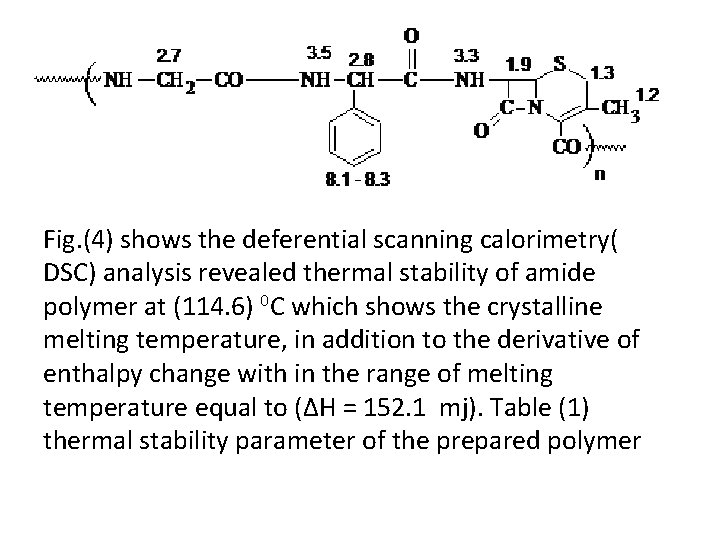
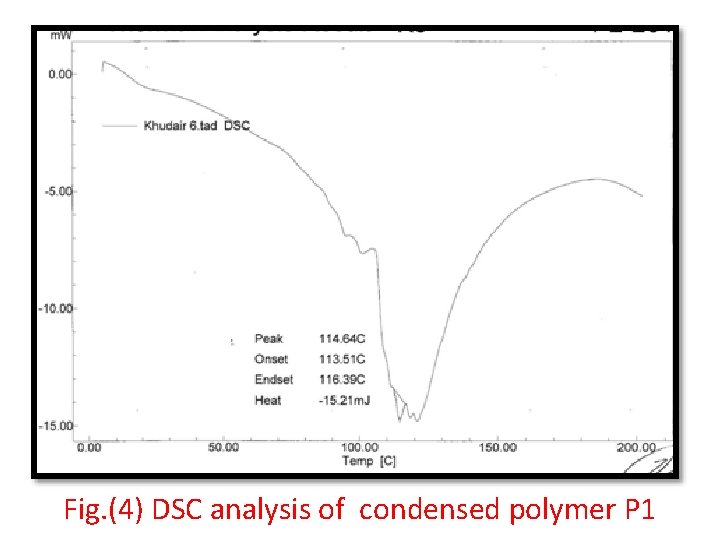
Fig. (4) shows the deferential scanning calorimetry( DSC) analysis revealed thermal stability of amide polymer at (114. 6) 0 C which shows the crystalline melting temperature, in addition to the derivative of enthalpy change with in the range of melting temperature equal to (ΔH = 152. 1 mj). Table (1) thermal stability parameter of the prepared polymer


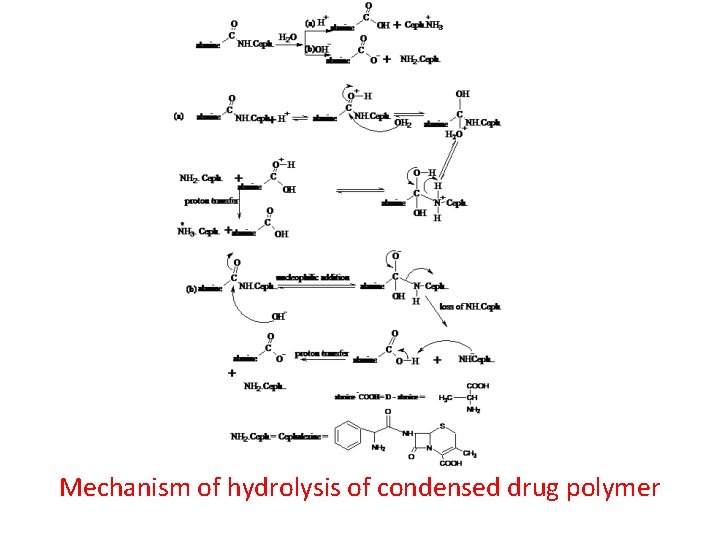
Mechanism of hydrolysis of condensed drug polymer

• Conclusions • The synthesized condensed polymer containing medicinal compounds such as cephalexin, and aminoacid such as D-alanine as bioactive materials were incorporating through the backbone of amide polymer which could hydrolysis in different p. H values at 370 C to be desirable for long term drug delivery, and release over a prolong time, and to minimize the drug side effects.

Fig(1) IR spectra of condensed amide polymer P 1

Fig. (2) UV. Spectra of P 1

Fig. (3) 1 H-NMR spectrum of the condensed polymer

Fig. (4) DSC analysis of condensed polymer P 1

Fig. (5) Controlled release studies of P 1

Thank you for listening