Syngas Fermentation and Design of Bioreactors Synferon Project

Syngas Fermentation and Design of Bioreactors Synferon Project Meeting #3 WP 2: Microbial fermentation of syngas Ph. D student: Konstantinos Asimakopoulos Supervisors: Ioannis Skiadas, Hariklia Gavala Date: 19/06/2018
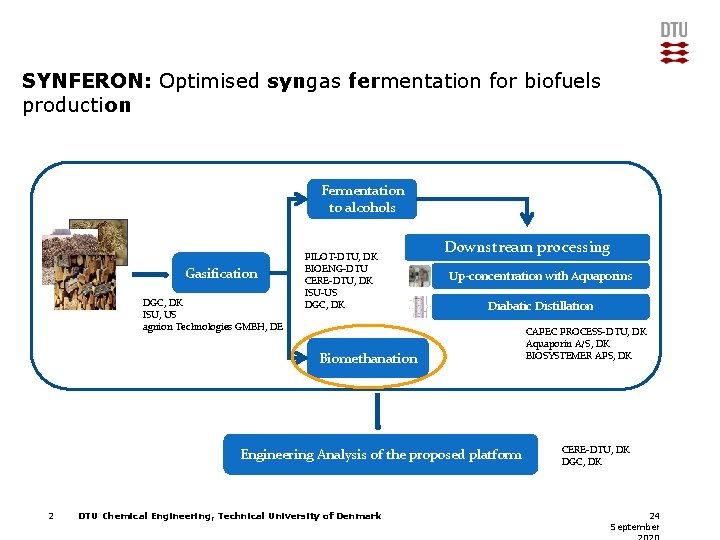
SYNFERON: Optimised syngas fermentation for biofuels production Fermentation to alcohols Gasification DGC, DK ISU, US agnion Technologies GMBH, DE PILOT-DTU, DK BIOENG-DTU CERE-DTU, DK ISU-US DGC, DK Downstream processing Up-concentration with Aquaporins Diabatic Distillation Biomethanation Engineering Analysis of the proposed platform 2 DTU Chemical Engineering, Technical University of Denmark CAPEC PROCESS-DTU, DK Aquaporin A/S, DK BIOSYSTEMER APS, DK CERE-DTU, DK DGC, DK 24 September
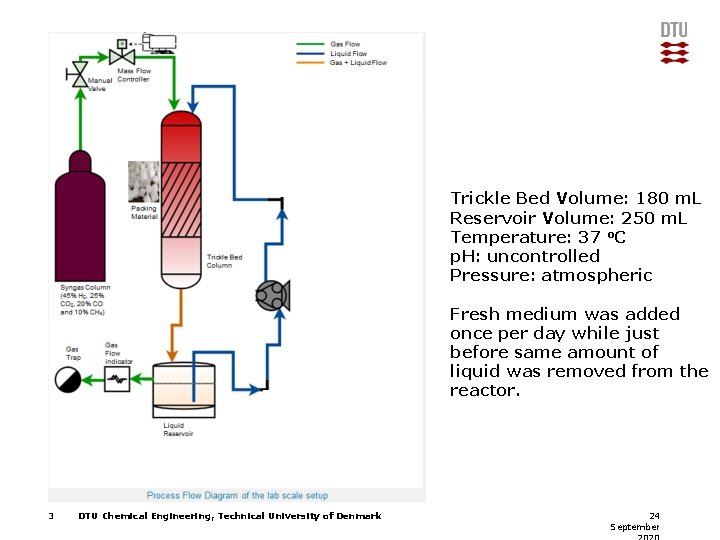
Trickle Bed Volume: 180 m. L Reservoir Volume: 250 m. L Temperature: 37 o. C p. H: uncontrolled Pressure: atmospheric Fresh medium was added once per day while just before same amount of liquid was removed from the reactor. 3 DTU Chemical Engineering, Technical University of Denmark 24 September

Inoculum of the trickcle bed reactors Enrichment/Acclimation Process Transfer 1 Transfer 2 Transfer 3 Transfer 4 Temperature: 37 o. C, p. H = 7 Headspace: Artificial Syngas Synthetic mineral medium Transfer after consumption of the substrate 4 DTU Chemical Engineering, Technical University of Denmark 24 September
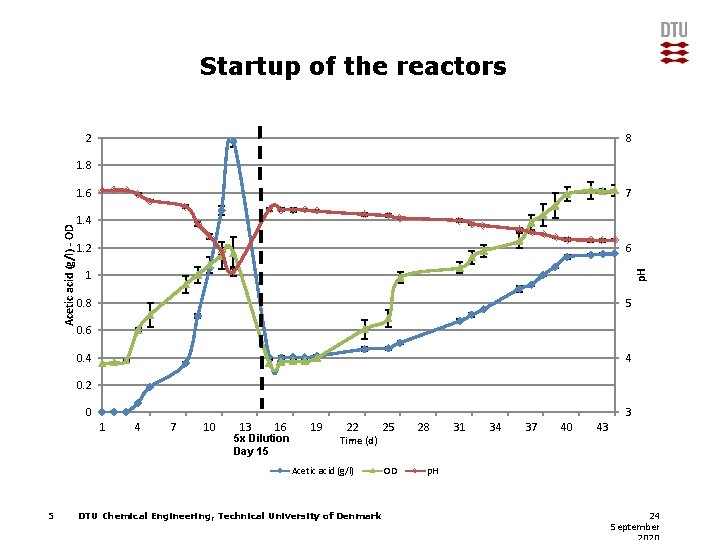
Startup of the reactors 2 8 1. 8 7 1. 4 1. 2 6 1 p. H Acetic acid (g/l) - OD 1. 6 0. 8 5 0. 6 0. 4 4 0. 2 0 3 1 4 7 10 13 16 5 x Dilution Day 15 19 22 Time (d) Acetic acid (g/l) 5 DTU Chemical Engineering, Technical University of Denmark 25 OD 28 31 34 37 40 43 p. H 24 September
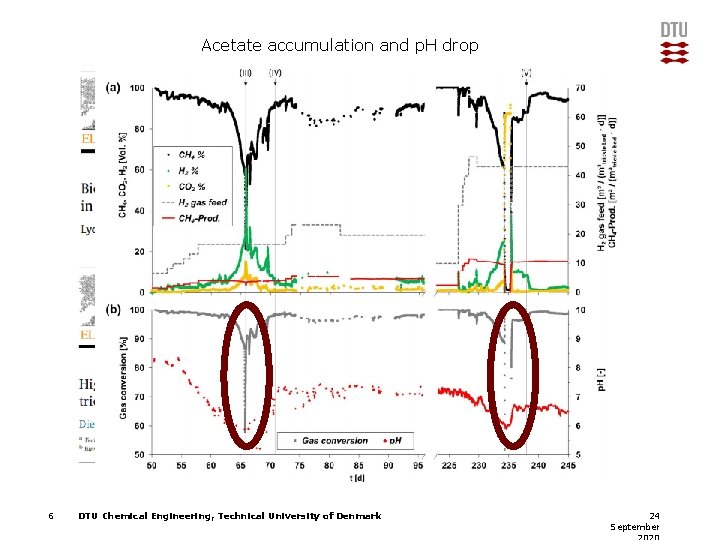
Acetate accumulation and p. H drop 6 DTU Chemical Engineering, Technical University of Denmark 24 September

Adjustment of the buffering capacity of the medium with phosphate buffer Initial medium Media Component Currently Used NH 4 Cl Na. Cl Mg. Cl 2, 6 H 2 O Ca. Cl 2 K 2 HPO 4 KH 2 PO 4 Na. HCO 3 Na 2 S, 9 H 2 O Nitrilotriacetic acid Trace Metal Solution Vitamin Solution 7 Concentration per 1. 0 L of Media 1 g or 18. 7 mmol 0. 1 g or 1. 71 mmol 0. 1 g or 0. 5 mmol 0. 05 g or 0. 34 mmol 0. 304 g or 1. 8 mmol 0 2. 6 g or 31 mmol 24 g or 100 mmol 0. 015 g or 0. 078 mmol 1 ml 10 ml Adjusted medium Media Component Currently Used NH 4 Cl Na. Cl Mg. Cl 2, 6 H 2 O Ca. Cl 2 K 2 HPO 4 KH 2 PO 4 Na. HCO 3 Na 2 S, 9 H 2 O Nitrilotriacetic acid Trace Metal Solution Vitamin Solution DTU Chemical Engineering, Technical University of Denmark Concentration per 1. 0 L of Media 1 g or 18. 7 mmol 0. 1 g or 1. 71 mmol 0. 1 g or 0. 5 mmol 0. 05 g or 0. 34 mmol 15 g or 87 mmol 1. 77 or 13 mmol 2. 6 g or 31 mmol 24 g or 100 mmol 0. 015 g or 0. 078 mmol 1 ml 10 ml 24 September
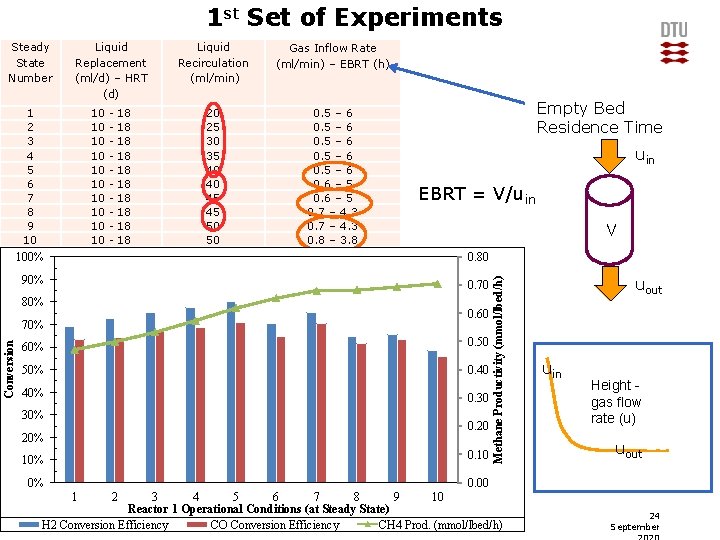
1 st Set of Experiments Liquid Replacement (ml/d) – HRT (d) 1 2 3 4 5 6 7 8 9 10 10 10 - 18 18 18 Gas Inflow Rate (ml/min) – EBRT (h) 20 25 30 35 40 40 45 45 50 50 0. 5 – 6 0. 6 – 5 0. 7 – 4. 3 0. 8 – 3. 8 Empty Bed Residence Time uin EBRT = V/uin V 100% 0. 90 0. 80 90% 0. 80 0. 70 80% 0. 70 0. 60 70% Conversion Liquid Recirculation (ml/min) 0. 60 0. 50 0. 40 0. 30 60% 50% 40% 30% 20% 0. 20 10% 0. 10 0% 0. 00 1 Methane Productivity (mmol/lbed/h) Steady State Number 3 4 5 6 7 8 9 10 Reactor Conditions (at Steady State) Reactor 2 1 Operational Conditions (at Steady State) 8 DTU Chemical Engineering, Technical University of Denmark H 2 Conversion Efficiency CO Conversion Efficiency CH 4 Prod. (mmol/lbed/h) uout uin Height gas flow rate (u) uout 2 24 September
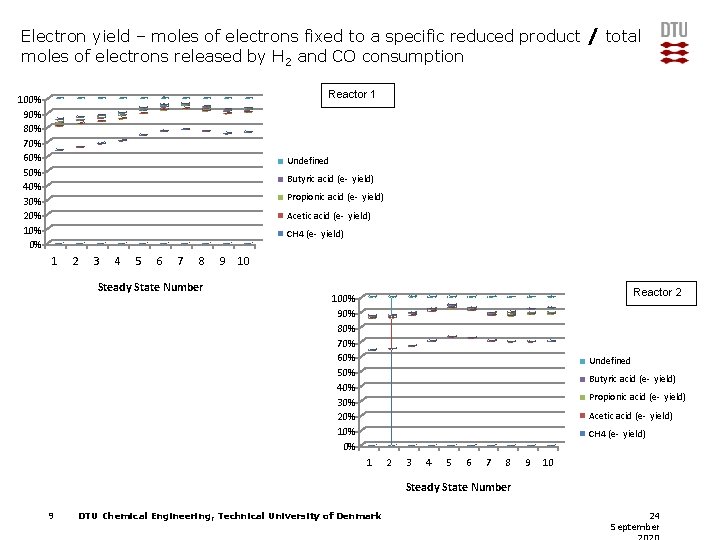
Electron yield – moles of electrons fixed to a specific reduced product / total moles of electrons released by H 2 and CO consumption Reactor 1 100% 90% 80% 70% 60% 50% 40% 30% 20% 10% 0% Undefined Butyric acid (e- yield) Propionic acid (e- yield) Acetic acid (e- yield) CH 4 (e- yield) 1 2 3 4 5 6 7 8 Steady State Number 9 10 Reactor 2 100% 90% 80% 70% 60% 50% 40% 30% 20% 10% 0% Undefined Butyric acid (e- yield) Propionic acid (e- yield) Acetic acid (e- yield) CH 4 (e- yield) 1 2 3 4 5 6 7 8 9 10 Steady State Number 9 DTU Chemical Engineering, Technical University of Denmark 24 September
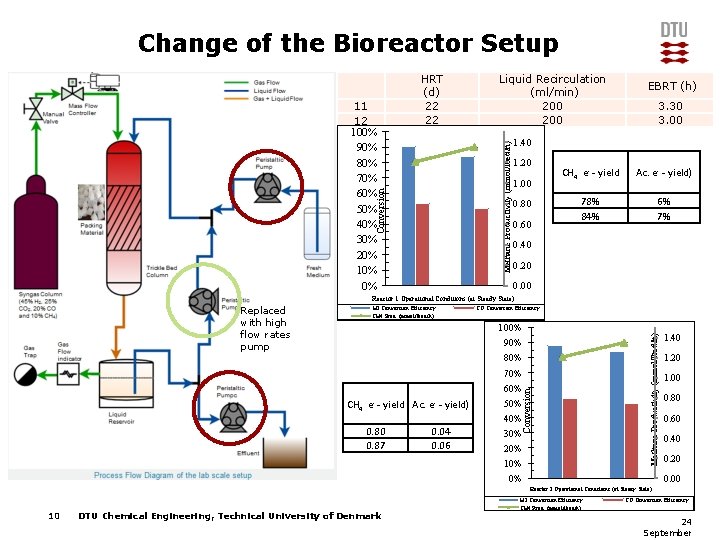
Change of the Bioreactor Setup Liquid Recirculation (ml/min) 200 Methane Productivity (mmol/lbed/h) Conversion 11 12 100% 90% 80% 70% 60% 50% 40% 30% 20% 10% 0% HRT (d) 22 22 EBRT (h) 3. 30 3. 00 1. 40 1. 20 1. 00 0. 80 0. 60 CH 4 e- - yield Ac. e- - yield) 78% 84% 6% 7% 0. 40 0. 20 0. 00 Reactor 1 Operational Conditions (at Steady State) CH 4 e- - yield Ac. e- - yield) 0. 80 0. 87 0. 04 0. 06 CO Conversion Efficiency 100% 90% 80% 70% 60% 50% 40% 30% 20% 10% 0% Methane Productivity (mmol/lbed/h) H 2 Conversion Efficiency CH 4 Prod. (mmol/lbed/h) Conversion Replaced with high flow rates pump 1. 40 1. 20 1. 00 0. 80 0. 60 0. 40 0. 20 0. 00 Reactor 2 Operational Conditions (at Steady State) 10 DTU Chemical Engineering, Technical University of Denmark H 2 Conversion Efficiency CH 4 Prod. (mmol/lbed/h) CO Conversion Efficiency 24 September
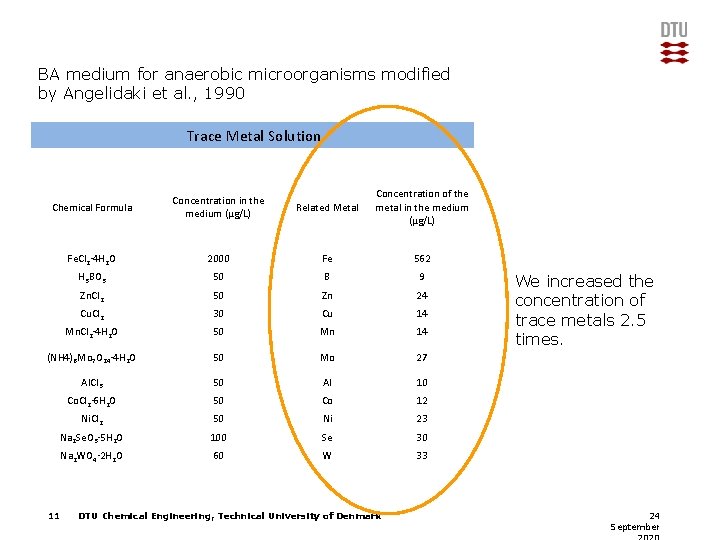
BA medium for anaerobic microorganisms modified by Angelidaki et al. , 1990 Trace Metal Solution Chemical Formula Concentration in the medium (μg/L) Related Metal Concentration of the metal in the medium (μg/L) Fe. Cl 2· 4 H 2 O 2000 Fe 562 H 3 BO 3 50 B 9 Zn. Cl 2 50 Zn 24 Cu. Cl 2 30 Cu 14 Mn. Cl 2· 4 H 2 O 50 Mn 14 (NH 4)6 Mo 7 O 24· 4 H 2 O 50 Mo 27 Al. Cl 3 50 Al 10 Co. Cl 2· 6 H 2 O 50 Co 12 Ni. Cl 2 50 Ni 23 Na 2 Se. O 3· 5 H 2 O 100 Se 30 Na 2 WO 4· 2 H 2 O 60 W 33 11 DTU Chemical Engineering, Technical University of Denmark We increased the concentration of trace metals 2. 5 times. 24 September
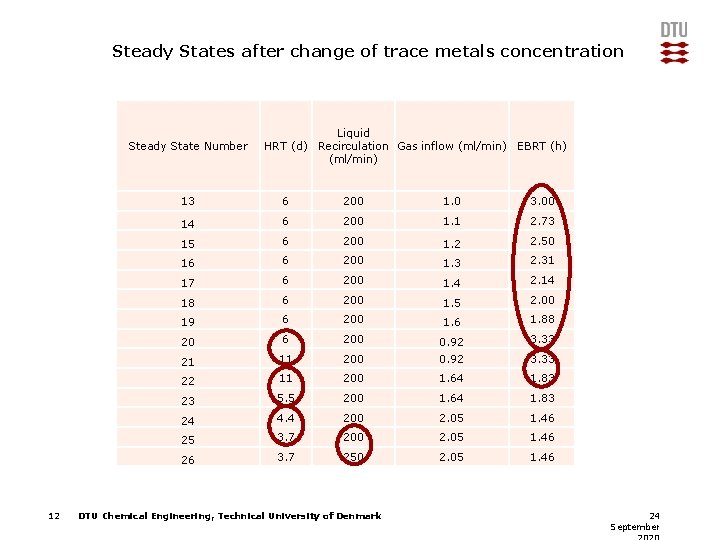
Steady States after change of trace metals concentration Steady State Number 12 Liquid HRT (d) Recirculation Gas inflow (ml/min) EBRT (h) (ml/min) 13 6 200 1. 0 3. 00 14 6 200 1. 1 2. 73 15 6 200 1. 2 2. 50 16 6 200 1. 3 2. 31 17 6 200 1. 4 2. 14 18 6 200 1. 5 2. 00 19 6 200 1. 6 1. 88 20 6 200 0. 92 3. 33 21 11 200 0. 92 3. 33 22 11 200 1. 64 1. 83 23 5. 5 200 1. 64 1. 83 24 4. 4 200 2. 05 1. 46 25 3. 7 200 2. 05 1. 46 26 3. 7 250 2. 05 1. 46 DTU Chemical Engineering, Technical University of Denmark 24 September
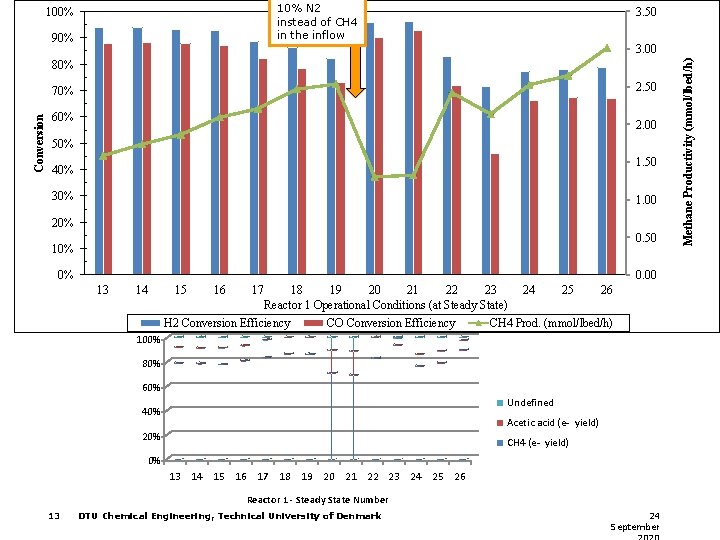
10% N 2 instead of CH 4 in the inflow 100% 90% 3. 50 80% 2. 50 Conversion 70% 60% 2. 00 50% 1. 50 40% 30% 1. 00 20% 0. 50 10% 0% 0. 00 13 14 15 16 17 18 19 20 21 22 23 24 25 26 Reactor 1 Operational Conditions (at Steady State) H 2 Conversion Efficiency CO Conversion Efficiency CH 4 Prod. (mmol/lbed/h) 100% 80% 60% Undefined 40% Acetic acid (e- yield) 20% CH 4 (e- yield) 0% 13 14 15 16 17 18 19 20 21 22 23 24 25 26 Reactor 1 - Steady State Number 13 DTU Chemical Engineering, Technical University of Denmark 24 September Methane Productivity (mmol/lbed/h) 3. 00
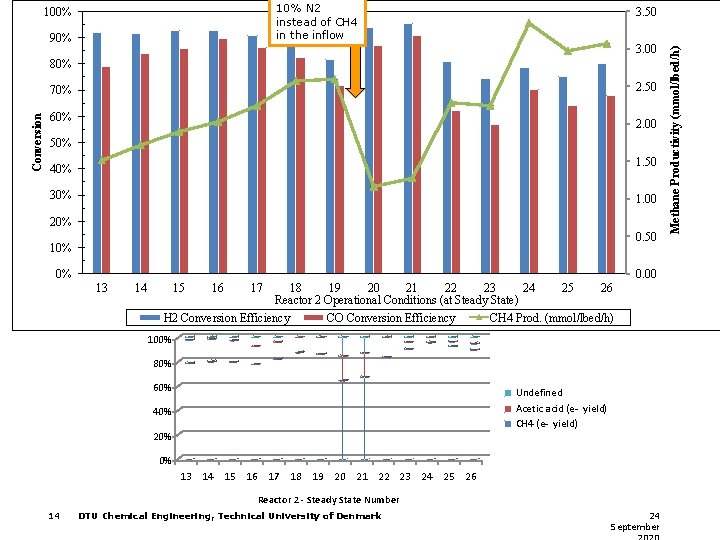
10% N 2 instead of CH 4 in the inflow 90% 3. 50 3. 00 80% 2. 50 Conversion 70% 60% 2. 00 50% 1. 50 40% 30% 1. 00 20% 0. 50 10% 0% 0. 00 13 14 15 16 17 18 19 20 21 22 23 24 25 26 Reactor 2 Operational Conditions (at Steady State) H 2 Conversion Efficiency CO Conversion Efficiency CH 4 Prod. (mmol/lbed/h) 100% 80% 60% Undefined Acetic acid (e- yield) CH 4 (e- yield) 40% 20% 0% 13 14 15 16 17 18 19 20 21 22 23 24 25 26 Reactor 2 - Steady State Number 14 DTU Chemical Engineering, Technical University of Denmark 24 September Methane Productivity (mmol/lbed/h) 100%
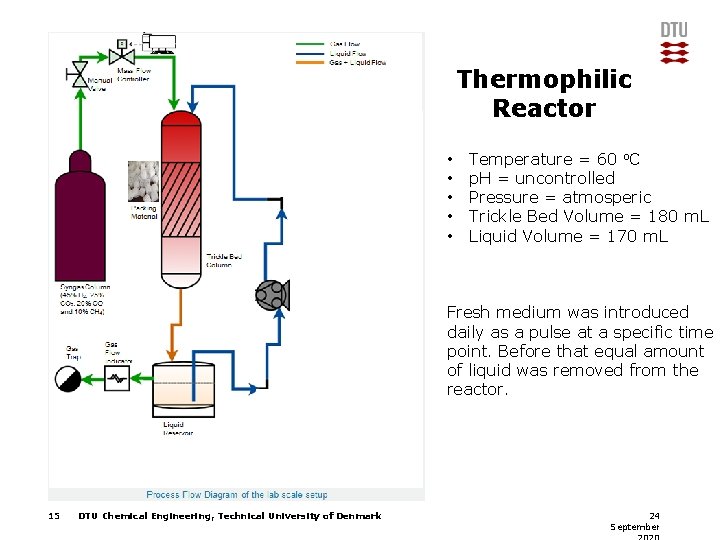
Thermophilic Reactor • • • Temperature = 60 o. C p. H = uncontrolled Pressure = atmosperic Trickle Bed Volume = 180 m. L Liquid Volume = 170 m. L Fresh medium was introduced daily as a pulse at a specific time point. Before that equal amount of liquid was removed from the reactor. 15 DTU Chemical Engineering, Technical University of Denmark 24 September
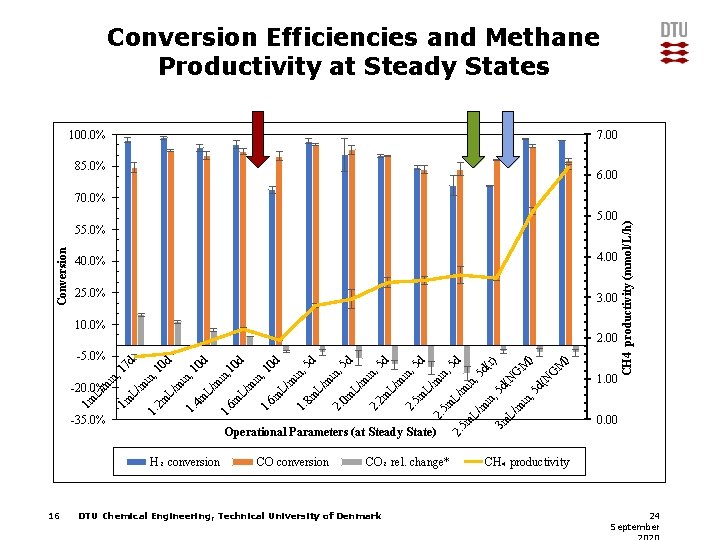
Conversion Efficiencies and Methane Productivity at Steady States 100. 0% 7. 00 85. 0% 6. 00 5. 00 Conversion 55. 0% 40. 0% 4. 00 25. 0% 3. 00 10. 0% CO conversion CO₂ rel. change* DTU Chemical Engineering, Technical University of Denmark G M ) ) M G d( N N , 5 1. 00 in L/ m 3 m L/ m in , 5 d( in , 5 d (r) 5 d 2. 5 m L/ m in , 5 d in , 5 m 2. L/ m in , 2 m 0 m 2. L/ m m L/ 8 m 1. 5 d 5 d in , d 6 m L/ m in , 1. m L/ 6 m 1. Operational Parameters (at Steady State) H₂ conversion 16 10 d 10 in , m L/ 4 m 1. L/ m in , 10 d 0 d , 1 2 m L/ m 1. -35. 0% 1 m 1 m L/ m -20. 0% in in , 1 7 d -5. 0% 5 d 2. 00 CH 4 productivity (mmol/L/h) 70. 0% 0. 00 CH₄ productivity 24 September
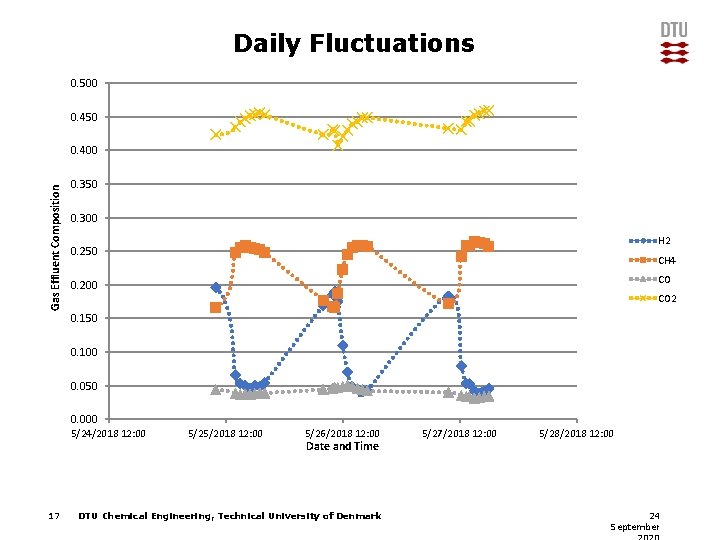
Daily Fluctuations 0. 500 0. 450 Gas Effluent Composition 0. 400 0. 350 0. 300 Η 2 0. 250 CH 4 CO 0. 200 CO 2 0. 150 0. 100 0. 050 0. 000 5/24/2018 12: 00 17 5/25/2018 12: 00 5/26/2018 12: 00 Date and Time DTU Chemical Engineering, Technical University of Denmark 5/27/2018 12: 00 5/28/2018 12: 00 24 September
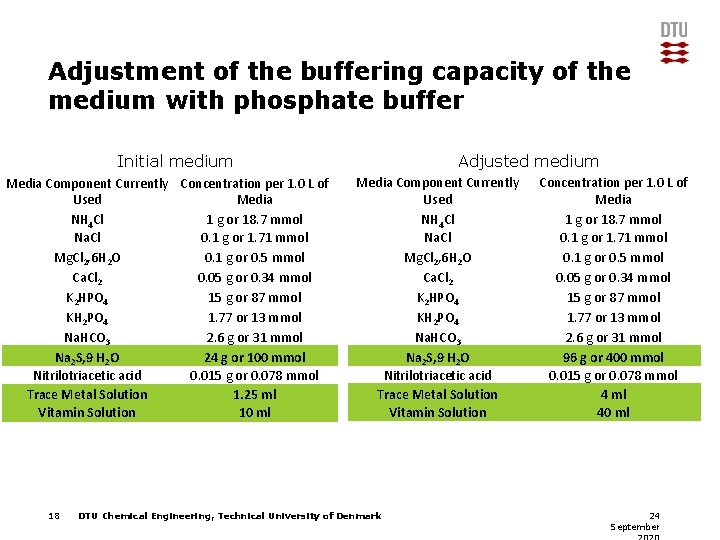
Adjustment of the buffering capacity of the medium with phosphate buffer Initial medium Media Component Currently Concentration per 1. 0 L of Used Media NH 4 Cl 1 g or 18. 7 mmol Na. Cl 0. 1 g or 1. 71 mmol Mg. Cl 2, 6 H 2 O 0. 1 g or 0. 5 mmol Ca. Cl 2 0. 05 g or 0. 34 mmol K 2 HPO 4 15 g or 87 mmol KH 2 PO 4 1. 77 or 13 mmol Na. HCO 3 2. 6 g or 31 mmol Na 2 S, 9 H 2 O 24 g or 100 mmol Nitrilotriacetic acid 0. 015 g or 0. 078 mmol Trace Metal Solution 1. 25 ml Vitamin Solution 10 ml 18 Adjusted medium Media Component Currently Used NH 4 Cl Na. Cl Mg. Cl 2, 6 H 2 O Ca. Cl 2 K 2 HPO 4 KH 2 PO 4 Na. HCO 3 Na 2 S, 9 H 2 O Nitrilotriacetic acid Trace Metal Solution Vitamin Solution DTU Chemical Engineering, Technical University of Denmark Concentration per 1. 0 L of Media 1 g or 18. 7 mmol 0. 1 g or 1. 71 mmol 0. 1 g or 0. 5 mmol 0. 05 g or 0. 34 mmol 15 g or 87 mmol 1. 77 or 13 mmol 2. 6 g or 31 mmol 96 g or 400 mmol 0. 015 g or 0. 078 mmol 4 ml 40 ml 24 September
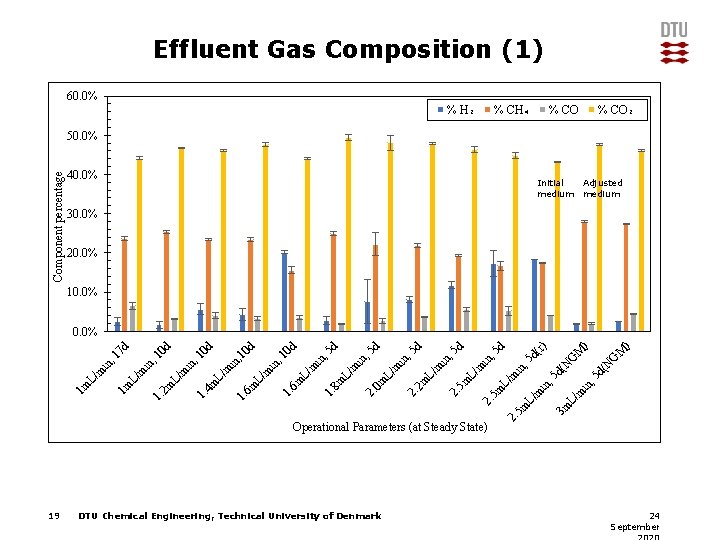
Effluent Gas Composition (1) 60. 0% % H₂ % CH₄ % CO₂ Component percentage 50. 0% 40. 0% Initial Adjusted medium 30. 0% 20. 0% 10. 0% DTU Chemical Engineering, Technical University of Denmark G d( N N d( , 5 3 m L/ m in in m L/ M ) ) M G 5 d in , m 5 m 2. Operational Parameters (at Steady State) 19 (r) 5 d L/ 2. 5 m 5 m L/ m in , 5 d in , 2. 2 m L/ m in , 2. 0 m L/ m m L/ 5 d 5 d in , 5 d 8 m 1. L/ 6 m 1. m m in , 10 d 0 d L/ 6 m 1. L/ 4 m 1. m L/ 2 m 1. m in , 1 10 d 0 d in L/ m 1 m 1 m L/ m in , 1 7 d 0. 0% 24 September
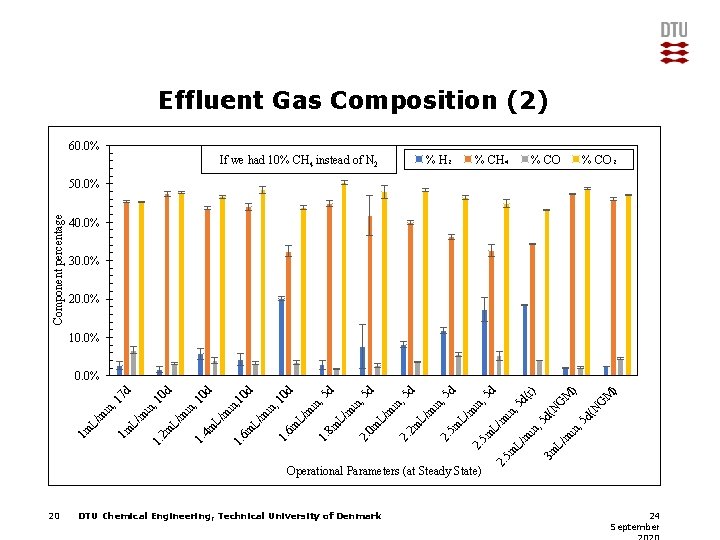
Effluent Gas Composition (2) 60. 0% % H₂ If we had 10% CH 4 instead of N 2 % CH₄ % CO₂ Component percentage 50. 0% 40. 0% 30. 0% 20. 0% 10. 0% M ) d( N G M ) G in , 5 N d( L/ m 3 m 2. 5 m L/ m in , 5 L/ m 5 m 2. DTU Chemical Engineering, Technical University of Denmark , 5 in m L/ Operational Parameters (at Steady State) 20 d( r) 5 d in , 5 d 5 m 2. L/ m in , 5 d 2 m 2. 0 m L/ m in , 5 d in , 2. L/ 8 m 1. m L/ 6 m m in , 10 in , 1. m L/ 6 m 5 d d d in , 1. 4 m L/ m in , 1. m L/ 10 d 10 0 d , 1 in 2 m 1. L/ m 1 m 1 m L/ m in , 1 7 d 0. 0% 24 September
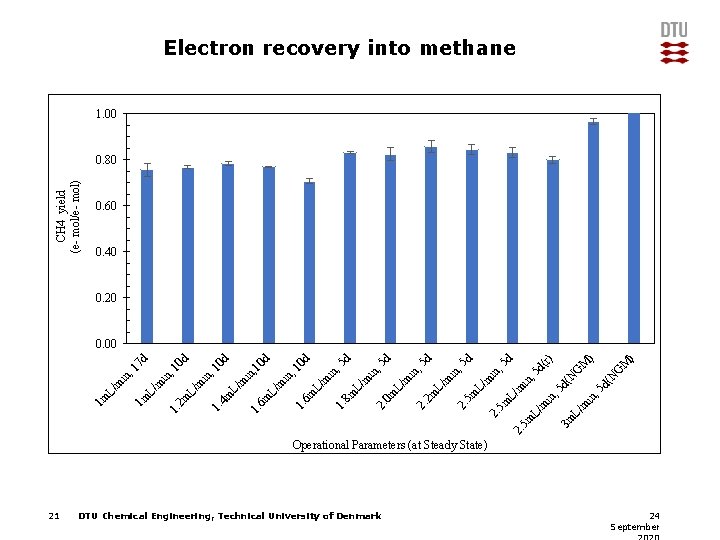
21 DTU Chemical Engineering, Technical University of Denmark N G d( M ) G 5 d 5 d (r) 5 d N d( , 5 in L/ m 3 m , 5 in , m L/ in m L/ 5 m 2. in , m L/ 2 m 2. in , m L/ 0 m 2. 5 d d 10 in , m L/ 8 m 1. m L/ 6 m 1. in , d d 10 10 in , m L/ 6 m 1. L/ 4 m 1. 0 d , 1 7 d , 1 in m L/ 2 m 1. L/ m 1 m in L/ m 1 m CH 4 yield (e- mol/e- mol) Electron recovery into methane 1. 00 0. 80 0. 60 0. 40 0. 20 0. 00 Operational Parameters (at Steady State) 24 September
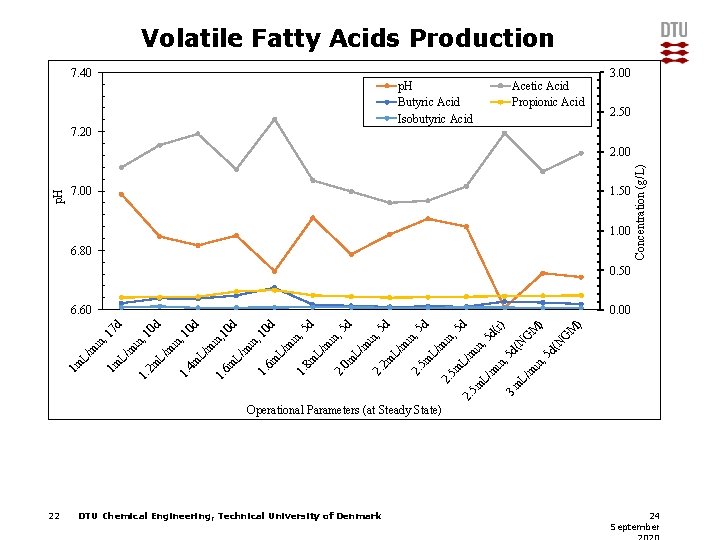
Volatile Fatty Acids Production 7. 40 3. 00 p. H Butyric Acid Isobutyric Acid Acetic Acid Propionic Acid 2. 50 7. 20 7. 00 1. 50 1. 00 6. 80 Concentration (g/L) p. H 2. 00 0. 50 M ) ) (N G M G m L/ m 3 5 m L/ m in in , , 5 m L/ 2. 5 d in , m 5 m 2. d( N 5 d (r) 5 d in , 5 d L/ 5 m 2. m in , 5 d L/ 2 m 2. L/ m in , 5 d in , 0 m 2. m L/ 8 m 1. L/ 6 m 1. m in , 10 in , m L/ 6 m 5 d d d in , m 1. L/ 4 m 1. m L/ 10 d 10 in , , 1 2 m 1. L/ m in , 1 1 m in m L/ 1 m 0 d 0. 00 7 d 6. 60 Operational Parameters (at Steady State) 22 DTU Chemical Engineering, Technical University of Denmark 24 September
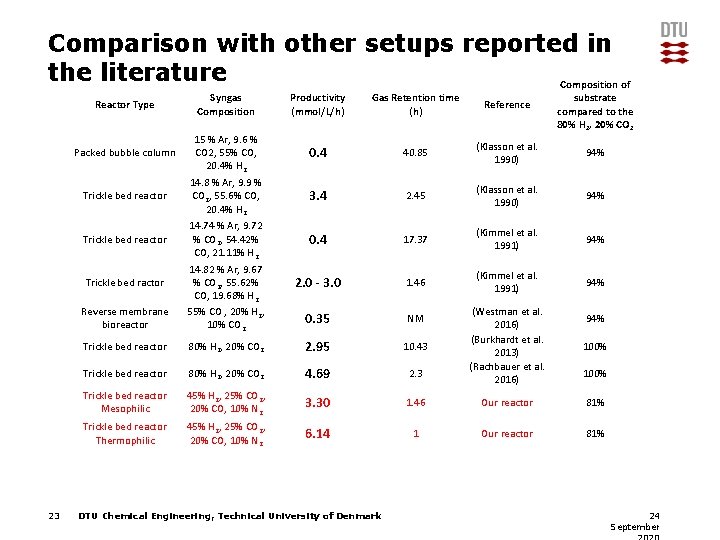
Comparison with other setups reported in the literature Composition of Reactor Type Syngas Composition Productivity (mmol/L/h) Gas Retention time (h) Reference Packed bubble column 15 % Ar, 9. 6 % CO 2, 55% CO, 20. 4% H 2 0. 4 40. 85 (Klasson et al. 1990) 94% Trickle bed reactor 14. 8 % Ar, 9. 9 % CO 2, 55. 6% CO, 20. 4% H 2 3. 4 2. 45 (Klasson et al. 1990) 94% Trickle bed reactor 14. 74 % Ar, 9. 72 % CO 2, 54. 42% CO, 21. 11% H 2 0. 4 17. 37 (Kimmel et al. 1991) 94% 2. 0 - 3. 0 1. 46 (Kimmel et al. 1991) 94% 0. 35 NM Reverse membrane bioreactor 14. 82 % Ar, 9. 67 % CO 2, 55. 62% CO, 19. 68% H 2 55% CO , 20% H 2, 10% CO 2 Trickle bed reactor 80% H 2, 20% CO 2 2. 95 10. 43 Trickle bed reactor 80% H 2, 20% CO 2 4. 69 2. 3 Trickle bed reactor Mesophilic 45% H 2, 25% CO 2, 20% CO, 10% N 2 3. 30 Trickle bed reactor Thermophilic 45% H 2, 25% CO 2, 20% CO, 10% N 2 6. 14 Trickle bed ractor 23 DTU Chemical Engineering, Technical University of Denmark substrate compared to the 80% H 2, 20% CO 2 (Westman et al. 2016) (Burkhardt et al. 2013) (Rachbauer et al. 2016) 100% 1. 46 Our reactor 81% 1 Our reactor 81% 94% 100% 24 September
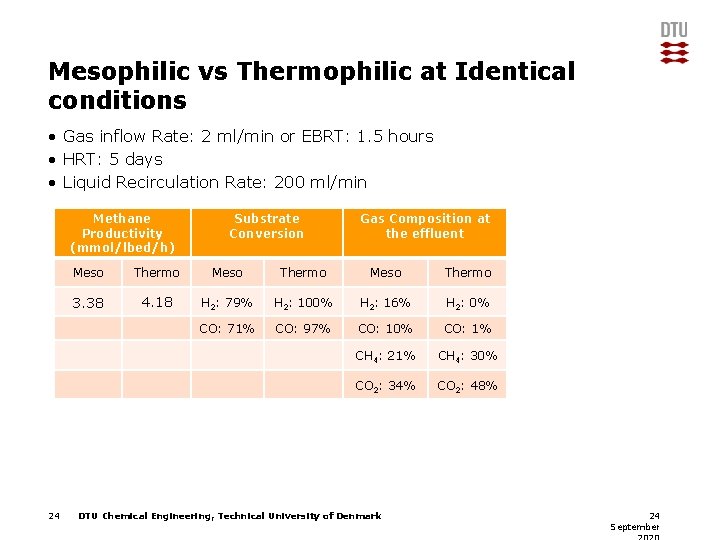
Mesophilic vs Thermophilic at Identical conditions • Gas inflow Rate: 2 ml/min or EBRT: 1. 5 hours • HRT: 5 days • Liquid Recirculation Rate: 200 ml/min Methane Productivity (mmol/lbed/h) 24 Substrate Conversion Gas Composition at the effluent Meso Thermo 3. 38 4. 18 H 2: 79% H 2: 100% H 2: 16% H 2: 0% CO: 71% CO: 97% CO: 10% CO: 1% CH 4: 21% CH 4: 30% CO 2: 34% CO 2: 48% DTU Chemical Engineering, Technical University of Denmark 24 September

Future Work on Syngas Biomethanation • External H 2 supply for the conversion of the remaining CO 2 in the gas phase • Construction and experimentation with a 5 L trickle bed reactor. The design has been completed and the construction has initiated from the workshop. • Pilot plant experiment with on line biomass gasifier at DGC. 25 DTU Chemical Engineering, Technical University of Denmark 24 September

Dissemination Work International Conferences • C 1 Net Conference (Nottigham, UK, 11/2017) : Oral Presentation • EUBCE Conference (Copenhagen, DK, 05/2018): Poster Presentation • Waste. Eng Conference (Prague, CZ, 07/2018): Oral Presentation Local Conferences • Research Day 2016 • Research Day 2017 • ATV Sustain Conference 2017 Publications • Special Issue on Waste and Biomass Valorization (09/2018) 26 DTU Chemical Engineering, Technical University of Denmark 24 September

Ackknowledgements 27 DTU Chemical Engineering, Technical University of Denmark 24 September
- Slides: 27