Symbolic model checking of biochemical systems Logic programming

Symbolic model checking of biochemical systems Logic programming steps towards formal biology François Fages, INRIA Rocquencourt http: //contraintes. inria. fr/ Joint work with and Nathalie Chabrier-Rivier Sylvain Soliman In collaboration with ARC CPBIO http: //contraintes. inria. fr/cpbio Alexander Bockmayr, Vincent Danos, Vincent Schächter et al. François Fages ICLP December 2003

Current revolution in Biology • Elucidation of high-level biological processes in terms of their biochemical basis at the molecular level. • Mass production of genomic and post-genomic data: ARN expression, protein synthesis, protein-protein interactions, … • Need for a strong parallel effort on the formal representation of biological processes. • Need formal tools for modeling and reasoning about their global behavior. François Fages ICLP December 2003
![Formalisms for modeling biochemical systems • • Diagrammatic notation Boolean networks [Thomas 73] Milner’s Formalisms for modeling biochemical systems • • Diagrammatic notation Boolean networks [Thomas 73] Milner’s](http://slidetodoc.com/presentation_image/6398b33a54cbba9abb318bbe0ff07290/image-3.jpg)
Formalisms for modeling biochemical systems • • Diagrammatic notation Boolean networks [Thomas 73] Milner’s –calculus [Regev-Silverman-Shapiro 99 -01, Nagasali et al. 00] Concurrent transition systems [Chabrier-Chiaverini-Danos-Fages-Schachter 03] Biochemical abstract machine BIOCHAM [Chabrier-Fages-Soliman 03] Pathway logic [Eker-Knapp-Laderoute-Lincoln-Meseguer-Sonmez 02] • Bio-ambients [Regev-Panina-Silverman-Cardelli-Shapiro 03] • • Differential equations Hybrid Petri nets [Hofestadt-Thelen 98, Matsuno et al. 00] Hybrid automata [Alur et al. 01, Ghosh-Tomlin 01] Hybrid concurrent constraint languages [Bockmayr-Courtois 01] François Fages ICLP December 2003

Our goal Beyond simulation, provide formal tools for querying, validating and completing biological models. Our proposal: • Use of temporal logic CTL as a query language for models of biological processes; • Use of concurrent transition systems for their modeling; • Use of symbolic and constraint-based model checkers for automatically evaluating CTL queries in qualitative and quantitative models. • Use of inductive logic programming for learning models [EU APRIL 2] In course, learn and teach bits of biology with logic programs. François Fages ICLP December 2003

Plan of the talk 1. Introduction 2. A simple algebra of cell molecules 3. Concurrent transition systems of biochemical reactions • Example of the mammalian cell cycle control 4. Temporal logic CTL as a query language • Computational results with BIOCHAM 5. Learning models • An experiment with inductive logic programming 6. Quantitative models • • Simulation with differential equations Constraint-based model checking 7. Conclusion François Fages ICLP December 2003

References A wonderful textbook: Molecular Cell Biology. 5 th Edition, 1100 pages+CD, Freeman Publ. Lodish, Berk, Zipursky, Matsudaira, Baltimore, Darnell. Nov. 2003. Genes and signals. Ptashne, Gann. CSHL Press. 2002. Modeling dynamic phenomena in molecular and cellular biology. Segel. Cambridge Univ. Press. 1987. Modeling and querying bio-molecular interaction networks. Chabrier, Chiaverini, Danos, Fages, Schächter. To appear in TCS. 2003. The biochemical abstract machine BIOCHAM. Chabrier, Fages, Soliman. http: //contraintes. inria. fr/BIOCHAM François Fages ICLP December 2003

2. A Simple Algebra of Cell Molecules Small molecules: covalent bonds (outer electrons shared) 50 -200 kcal/mol • 70% water • 1% ions • 6% amino acids (20), nucleotides (5), fats, sugars, ATP, ADP, … Macromolecules: hydrogen bonds, ionic, hydrophobic, Waals 1 -5 kcal/mol Stability and bindings determined by the number of weak bonds: 3 D shape • 20% proteins (50 -104 amino acids) • RNA (102 -104 nucleotides AGCU) • DNA (102 -106 nucleotides AGCT) François Fages ICLP December 2003
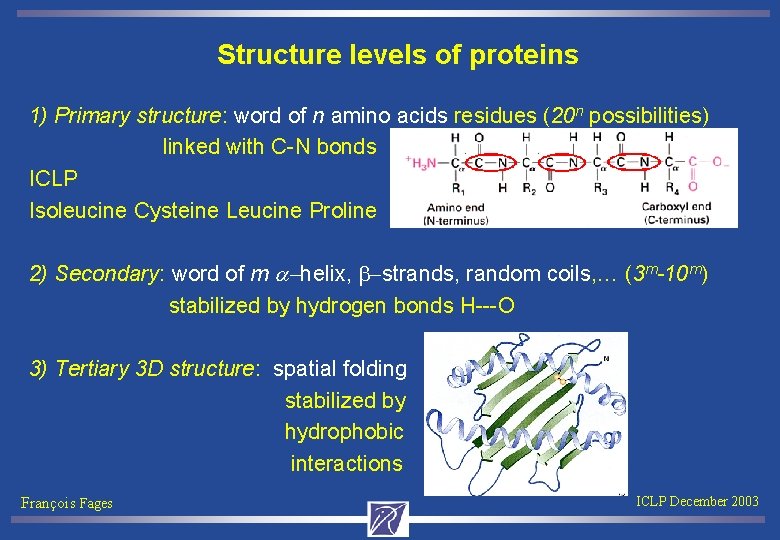
Structure levels of proteins 1) Primary structure: word of n amino acids residues (20 n possibilities) linked with C-N bonds ICLP Isoleucine Cysteine Leucine Proline 2) Secondary: word of m a-helix, b-strands, random coils, … (3 m-10 m) stabilized by hydrogen bonds H---O 3) Tertiary 3 D structure: spatial folding stabilized by hydrophobic interactions François Fages ICLP December 2003
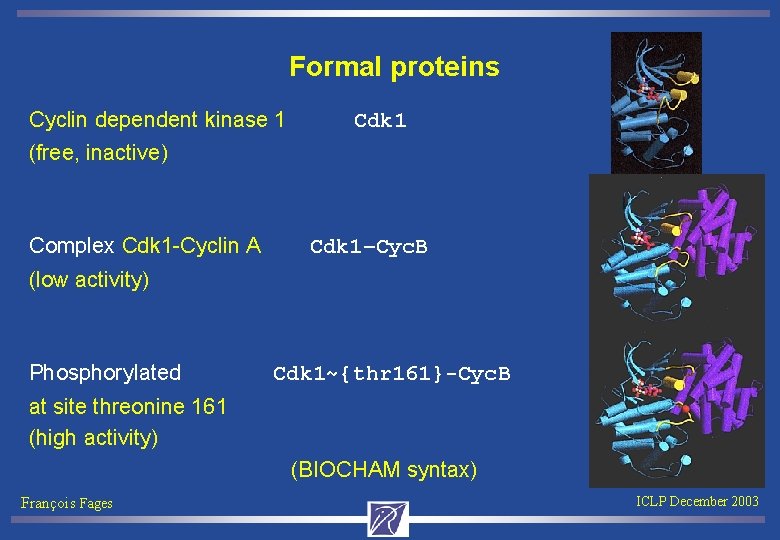
Formal proteins Cyclin dependent kinase 1 Cdk 1 (free, inactive) Complex Cdk 1 -Cyclin A Cdk 1–Cyc. B (low activity) Phosphorylated Cdk 1~{thr 161}-Cyc. B at site threonine 161 (high activity) (BIOCHAM syntax) François Fages ICLP December 2003

Gene expression: DNA RNA protein DNA: word over 4 nucleotides Adenine, Guanine, Cytosine, Thymine double helix of pairs A--T and C---G Replication: DNA synthesis Genes: parts of DNA Transcription: RNA copying from a gene ERCC 1 -(PRB-JUN-CFOS) François Fages ICLP December 2003

Genome Size Species Genome size Chromosomes Coding DNA E. Coli (bacteria) 5 Mb 1 circular 100 % S. Cerevisae (yeast) 12 Mb 16 70 % Mouse, Human 3 Gb 20, 23 15 % … 15 Gb … 140 Gb 3, 200, 000 pairs of nucleotides single nucleotide polymorphism 1 / 2 kb François Fages ICLP December 2003
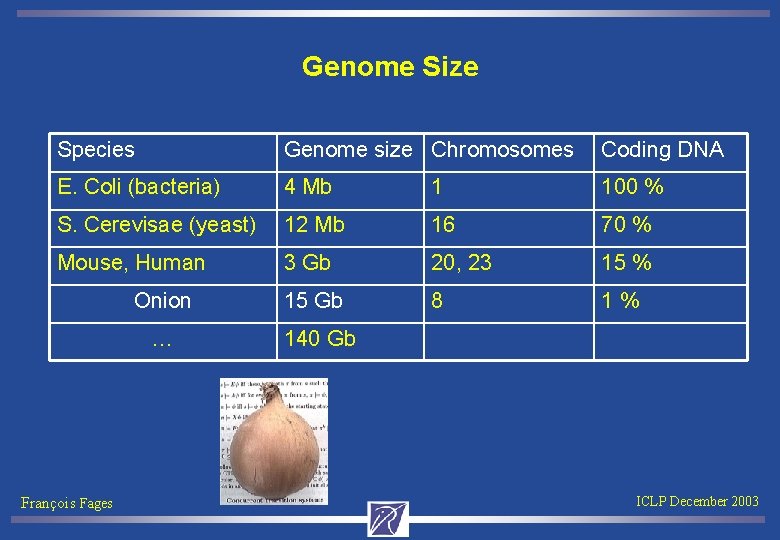
Genome Size Species Genome size Chromosomes Coding DNA E. Coli (bacteria) 4 Mb 1 100 % S. Cerevisae (yeast) 12 Mb 16 70 % Mouse, Human 3 Gb 20, 23 15 % Onion 15 Gb 8 1 % … 140 Gb François Fages ICLP December 2003
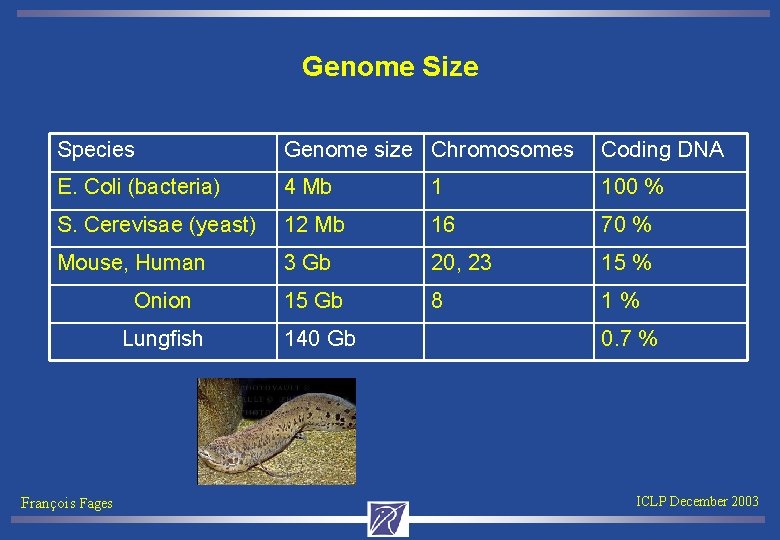
Genome Size Species Genome size Chromosomes Coding DNA E. Coli (bacteria) 4 Mb 1 100 % S. Cerevisae (yeast) 12 Mb 16 70 % Mouse, Human 3 Gb 20, 23 15 % Onion 15 Gb 8 1 % Lungfish 140 Gb François Fages 0. 7 % ICLP December 2003

Algebra of Cell Molecules E : : = Name|E-E|E~{E, …, E}|(E) S : : = _|E|S+S Names: proteins, gene binding sites, molecules, abstract processes… - : binding operator for protein complexes, gene binding sites, … Non associative, non commutative (could be in most cases) ~{…}: modification operator for phosphorylated sites, … Associative, Commutative, Idempotent. + : solution operator, “soup aspect”, Assoc. Comm. Idempotent, Neutral _ No membranes, no transport formalized. Bitonal calculi [Cardelli 03]. François Fages ICLP December 2003

Plan of the talk 1. Introduction 2. A simple algebra of cell molecules 3. Concurrent transition systems of biochemical reactions • Example of the mammalian cell cycle control 4. Temporal logic CTL as a query language • Computational results with BIOCHAM 5. Learning models • An experiment with inductive logic programming 6. Quantitative models • • Simulation with differential equations Constraint-based model checking 7. Conclusion François Fages ICLP December 2003

3. Concurrent Transition. Syst. of Biochemical Reactions Enzymatic reactions: R : : = S=>S | S=[E]=>S | S=[R]=>S | S<=[E]=>S (where A<=>B stands for A=>B B=>A and A=[C]=>B for A+C=>B+C, etc. ) define a concurrent transition system over integers denoting the multiplicity of the molecules (multiset rewriting). One can associate a finite abstract CTS over boolean state variables denoting the presence/absence of molecules which correctly over-approximates the set of all possible behaviors If we translate a reaction A+B=>C+D by 4 rules for possible consumption: A+B+C+D A+B +C+D A+B A+ B+C+D François Fages ICLP December 2003

Four Rule Schemas Complexation: A + B => A-B Cdk 1+Cyc. B => Cdk 1–Cyc. B Phosphorylation: A =[C]=> A~{p} Cdk 1–Cyc. B =[Myt 1]=> Cdk 1~{thr 161}-Cyc. B Cdk 1~{thr 14, tyr 15}-Cyc. B =[Cdc 25~{Nterm}]=> Cdk 1 -Cyc. B Synthesis: _ =[C]=> A. _ =[Ge 2 -E 2 f 13 -Dp 12]=> Cyc. A Degradation: A =[C]=> _. Cyc. E =[Ubi. Pro]=> _ (not for Cyc. E-Cdk 2 which is stable) François Fages ICLP December 2003
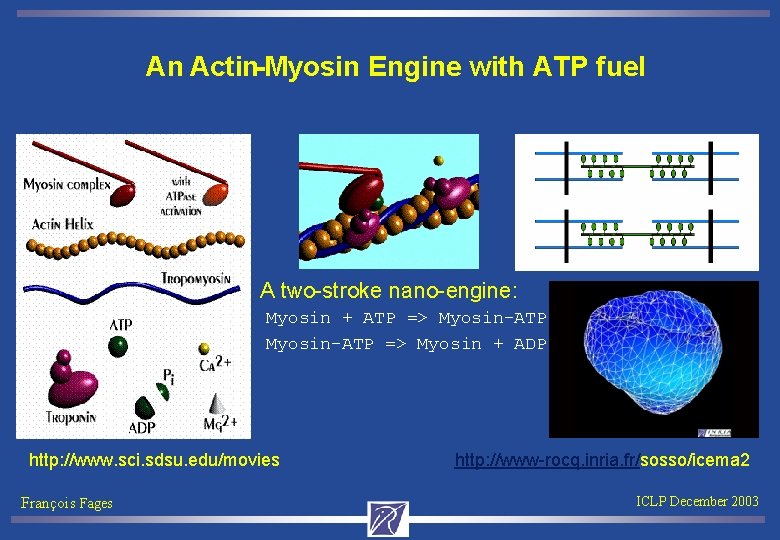
An Actin-Myosin Engine with ATP fuel A two-stroke nano-engine: Myosin + ATP => Myosin-ATP Myosin-ATP => Myosin + ADP http: //www. sci. sdsu. edu/movies http: //www-rocq. inria. fr/sosso/icema 2 François Fages ICLP December 2003
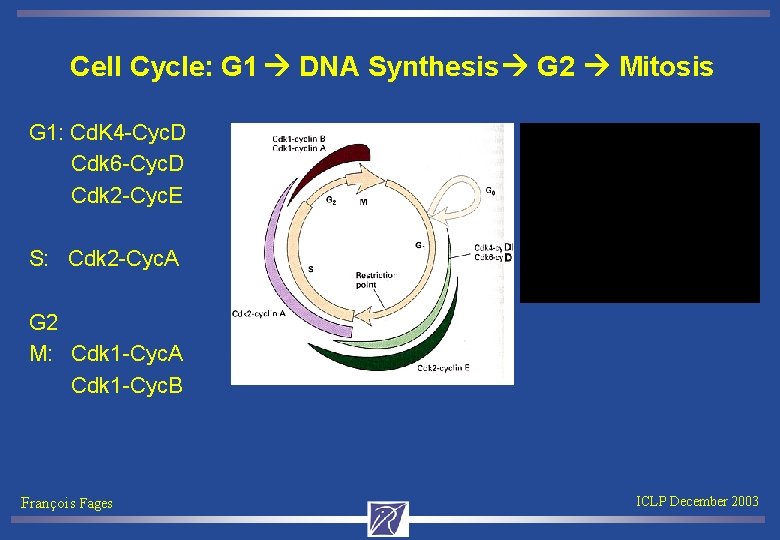
Cell Cycle: G 1 DNA Synthesis G 2 Mitosis G 1: Cd. K 4 -Cyc. D Cdk 6 -Cyc. D Cdk 2 -Cyc. E S: Cdk 2 -Cyc. A G 2 M: Cdk 1 -Cyc. A Cdk 1 -Cyc. B François Fages ICLP December 2003
![Mammalian Cell Cycle Control. Map [Kohn 99] François Fages ICLP December 2003 Mammalian Cell Cycle Control. Map [Kohn 99] François Fages ICLP December 2003](http://slidetodoc.com/presentation_image/6398b33a54cbba9abb318bbe0ff07290/image-20.jpg)
Mammalian Cell Cycle Control. Map [Kohn 99] François Fages ICLP December 2003
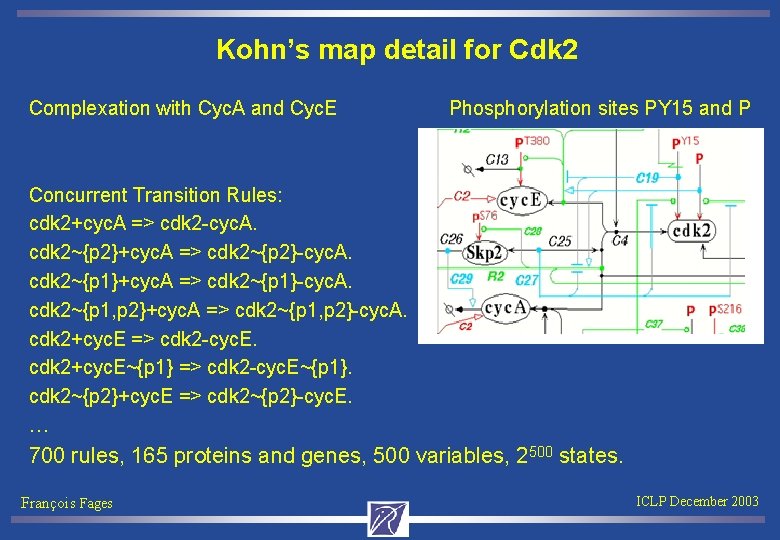
Kohn’s map detail for Cdk 2 Complexation with Cyc. A and Cyc. E Phosphorylation sites PY 15 and P Concurrent Transition Rules: cdk 2+cyc. A => cdk 2 -cyc. A. cdk 2~{p 2}+cyc. A => cdk 2~{p 2}-cyc. A. cdk 2~{p 1}+cyc. A => cdk 2~{p 1}-cyc. A. cdk 2~{p 1, p 2}+cyc. A => cdk 2~{p 1, p 2}-cyc. A. cdk 2+cyc. E => cdk 2 -cyc. E. cdk 2+cyc. E~{p 1} => cdk 2 -cyc. E~{p 1}. cdk 2~{p 2}+cyc. E => cdk 2~{p 2}-cyc. E. … 700 rules, 165 proteins and genes, 500 variables, 2500 states. François Fages ICLP December 2003

Translation in Prolog Encode states with a single predicate p(A, B, C, D, E) A+B C+D. p(1, 1, _, _, E): -p(_, _, 1, 1, E). C A. p(_, B, 1, D, E): - p(1, B, _, D, E). Thm. [Delzanno-Podelski 99] Predecessor(S) = TP(S) Backward analysis by computing lfp(TP {p(x): -s}). CLP-based Deductive Model Checker DMC [Delzanno-Podelski 99] More efficient implementation using state-of-the-art symbolic modelchecker Nu. SMV [Cimatti Clarke Giunchiglia Pistore 02]. François Fages ICLP December 2003

Plan of the talk 1. Introduction 2. A simple algebra of cell molecules 3. Concurrent transition systems of biochemical reactions • Example of the mammalian cell cycle control 4. Temporal logic CTL as a query language • Computational results with BIOCHAM 5. Learning models • An experiment with inductive logic programming 6. Quantitative models • Simulation with differential equations • Constraint-based model checking 7. Conclusion François Fages ICLP December 2003
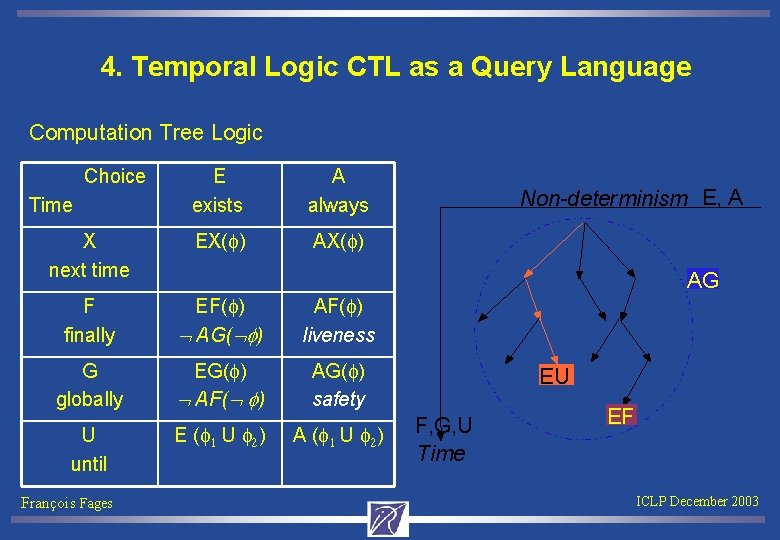
4. Temporal Logic CTL as a Query Language Computation Tree Logic Choice Time E exists A always X next time EX(f) AX(f) F finally EF(f) AG( f) AF(f) liveness G globally EG(f) AF( f) AG(f) safety U until François Fages Non-determinism E, A AG E (f 1 U f 2) A (f 1 U f 2) EU F, G, U Time EF ICLP December 2003

Kripke Structures A Kripke structure K is a triple (S; R; L) where S is a set of states, and R Sx. S is a total relation. s |= f if f is true in s, s |= E f if there is a path from s such that |= f, s |= A f if for every path from s, |= f, |= f if s |= f where s is the starting state of , |= X f if 1 |= f, |= F f if there exists k >0 such that k |= f, |= G f if for every k >0, k |= f, |= f 1 U f 2 iff there exists k>0 such that k |= f for all j < k j |= f. Following [Emerson 90] we identify a formula f to the set of states which satisfy it f ~ {s S : s |= f }. François Fages ICLP December 2003

Symbolic Model Checking is an algorithm for computing, in a given finite Kripke structure the set of states satisfying a CTL formula: {s S : s |= f }. Basic algorithm: represent K as a graph and iteratively label the nodes with the subformulas of f which are true in that node. Add f to the states satisfying f Add EF f (EX f) to the (immediate) predecessors of states labeled by f Add E(f 1 U f 2 ) to the predecessor states of f 2 while they satisfy f 1 Add EG f to the states for which there exists a path leading to a non trivial strongly connected component of the subgraph of states satisfying f Symbolic model checking: use OBDDs to represent states and transitions as boolean formulas (S is finite). François Fages ICLP December 2003

Biological Queries (1/3) About reachability: • Given an initial state init, can the cell produce some protein P? init EF(P) • Which are the states from which a set of products P 1, . . . , Pn can be produced simultaneously? EF(P 1^…^Pn) About pathways: • Can the cell reach a state s while passing by another state s 2? init EF(s 2^EFs) • Is state s 2 a necessary checkpoint for reaching state s? EF( s 2 U s) • Is it possible to produce P without using nor creating Q? EF( Q U s) • Can the cell reach a state s without violating some constraints c? init EF(c. Us) François Fages ICLP December 2003

Biological Queries (2/3) About stability: • Is a certain (partially described) state s a stable state? s AG(s) (s denotes both the state and the formula describing it). • Is s a steady state (with possibility of escaping) ? s EG(s) • Can the cell reach a stable state? init EF(AG(s))not a LTL formula. • Must the cell reach a stable state? init AF(AG(s)) • What are the stable states? Not expressible in CTL [Chan 00]. • Can the system exhibit a cyclic behavior w. r. t. the presence of P ? init EG((P EF P) ^ ( P EF P)) François Fages ICLP December 2003

Biological Queries (3/3) About the correctness of the model: • Can one see the inaccuracies of the model and correct them? Exhibit a counterexample pathway or a witness. Suggest refinements of the model or biological experiments to validate/invalidate the property of the model. About durations: • How long does it take for a molecule to become activated? • In a given time, how many Cyclins A can be accumulated? • What is the duration of a given cell cycle’s phase? CTL operators abstract from durations. Time intervals can be modeled in FO by adding numerical arguments for start times and durations. François Fages ICLP December 2003
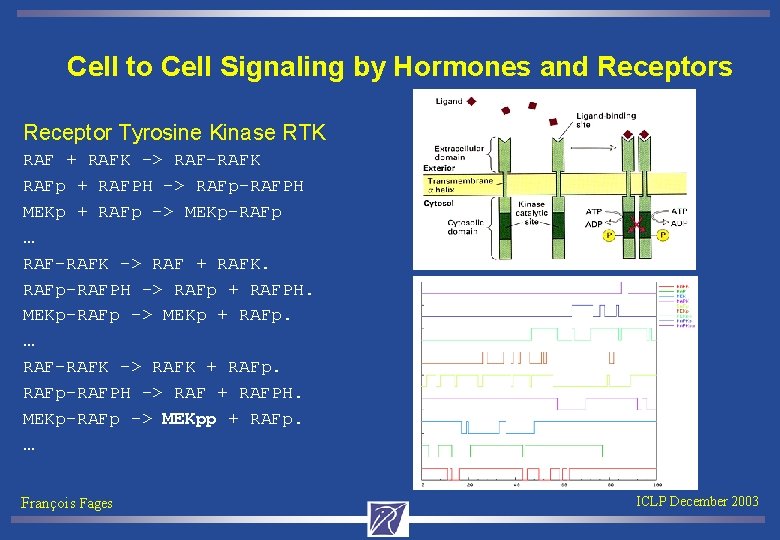
Cell to Cell Signaling by Hormones and Receptors Receptor Tyrosine Kinase RTK RAF + RAFK -> RAF-RAFK RAFp + RAFPH -> RAFp-RAFPH MEKp + RAFp -> MEKp-RAFp … RAF-RAFK -> RAF + RAFK. RAFp-RAFPH -> RAFp + RAFPH. MEKp-RAFp -> MEKp + RAFp. … RAF-RAFK -> RAFK + RAFp-RAFPH -> RAF + RAFPH. MEKp-RAFp -> MEKpp + RAFp. … François Fages ICLP December 2003
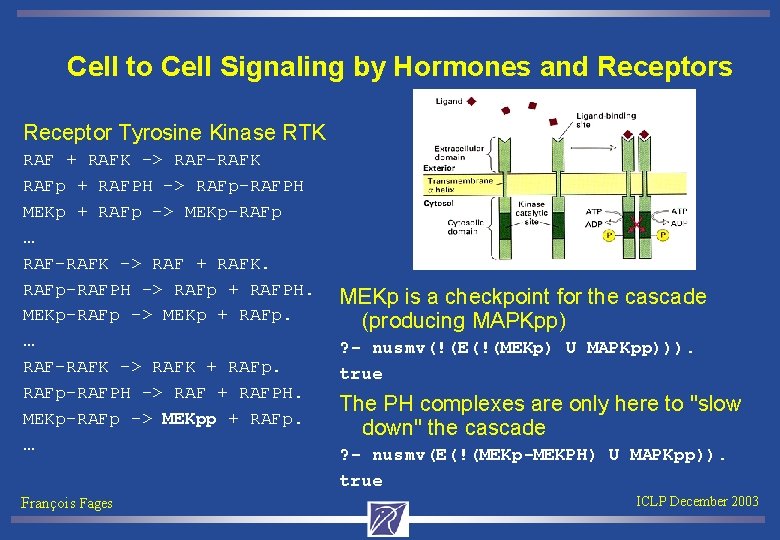
Cell to Cell Signaling by Hormones and Receptors Receptor Tyrosine Kinase RTK RAF + RAFK -> RAF-RAFK RAFp + RAFPH -> RAFp-RAFPH MEKp + RAFp -> MEKp-RAFp … RAF-RAFK -> RAF + RAFK. RAFp-RAFPH -> RAFp + RAFPH. MEKp-RAFp -> MEKp + RAFp. … RAF-RAFK -> RAFK + RAFp-RAFPH -> RAF + RAFPH. MEKp-RAFp -> MEKpp + RAFp. … François Fages MEKp is a checkpoint for the cascade (producing MAPKpp) ? - nusmv(!(E(!(MEKp) U MAPKpp))). true The PH complexes are only here to "slow down" the cascade ? - nusmv(E(!(MEKp-MEKPH) U MAPKpp)). true ICLP December 2003
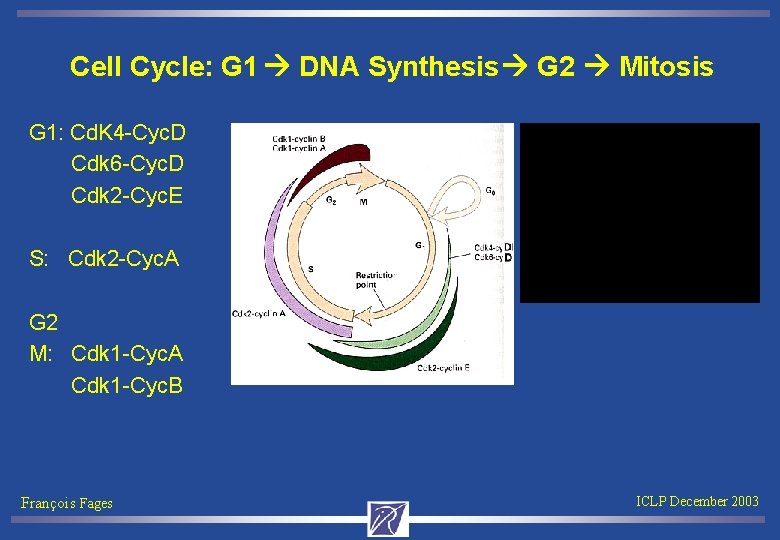
Cell Cycle: G 1 DNA Synthesis G 2 Mitosis G 1: Cd. K 4 -Cyc. D Cdk 6 -Cyc. D Cdk 2 -Cyc. E S: Cdk 2 -Cyc. A G 2 M: Cdk 1 -Cyc. A Cdk 1 -Cyc. B François Fages ICLP December 2003
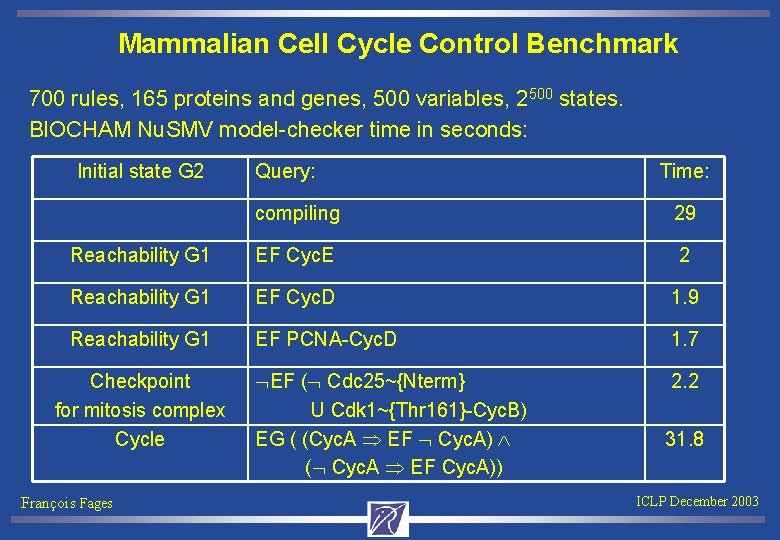
Mammalian Cell Cycle Control Benchmark 700 rules, 165 proteins and genes, 500 variables, 2500 states. BIOCHAM Nu. SMV model-checker time in seconds: Initial state G 2 Query: Time: compiling 29 Reachability G 1 EF Cyc. E 2 Reachability G 1 EF Cyc. D 1. 9 Reachability G 1 EF PCNA-Cyc. D 1. 7 EF ( Cdc 25~{Nterm} U Cdk 1~{Thr 161}-Cyc. B) EG ( (Cyc. A EF Cyc. A) ( Cyc. A EF Cyc. A)) 2. 2 Checkpoint for mitosis complex Cycle François Fages 31. 8 ICLP December 2003

Plan of the talk 1. Introduction 2. A simple algebra of cell molecules 3. Concurrent transition systems of biochemical reactions • Example of the mammalian cell cycle control 4. Temporal logic CTL as a query language • Computational results with BIOCHAM 5. Learning models • An experiment with inductive logic programming 6. Quantitative models • Simulation with differential equations • Constraint-based model checking 7. Conclusion François Fages ICLP December 2003

5. Learning Models Basic idea: learn reaction rules from temporal properties of the system. Learning of yeast cell cycle rules from reachability properties and counterexamples with Progol [Muggleton 00]. reaction([m_CP, m_Y], [m_p. M]). reaction([m_CP], [m_C 2]). % reaction([m_p. M], [m_M]). reaction([m_M], [m_C 2, m_YP]). reaction([m_C 2], [m_CP]). reaction([m_YP], []). reaction([], [m_Y]). pathway(S 1, S 2) : - same(S 1, S 2). pathway(S 1, S 2) : - reaction(L 1, L 2), transition(S 1, L 1, S 3, L 2), pathway(S 3, S 2). François Fages ICLP December 2003
![Inductive Logic Programming pathway([m_CP, m_Y], [m_M]). pathway([m_CP, m_Y], [m_M, m_p. M ]). pathway([m_CP, m_Y], Inductive Logic Programming pathway([m_CP, m_Y], [m_M]). pathway([m_CP, m_Y], [m_M, m_p. M ]). pathway([m_CP, m_Y],](http://slidetodoc.com/presentation_image/6398b33a54cbba9abb318bbe0ff07290/image-36.jpg)
Inductive Logic Programming pathway([m_CP, m_Y], [m_M]). pathway([m_CP, m_Y], [m_M, m_p. M ]). pathway([m_CP, m_Y], [m_M, m_Y, m_p. M ]). pathway([m_CP, m_Y], [m_M, m_CP, m_Y ]). pathway([m_CP, m_Y], [m_M, m_CP, m_p. M ]). pathway([m_CP, m_Y], [m_M, m_CP, m_Y, m_p. M]). pathway([m_p. M], [m_C 2, m_YP]). pathway([m_p. M], [m_M, m_C 2, m_YP]). pathway([m_p. M], [m_p. M, m_C 2, m_YP]). pathway([m_p. M], [m_M, m_p. M, m_C 2, m_Y P]). : -pathway([], [m_C 2]). : -pathway([], [m_CP]). : -pathway([], [m_C 2, m_CP]). : -pathway([], [m_M]). : -pathway([], [m_YP, m_Y]). : -pathway([], [m_Y, m_p. M]). : -pathway([], [m_CP, m_p. M]). : -pathway([], [m_Y, m_M]). : -pathway([m_CP, m_C 2], [m_YP]). : -pathway([m_CP], [m_YP]). : -pathway([m_C 2], [m_YP]). : -pathway([m_Y], []). reaction([m_p. M], [m_M]) learned… 6 th PCRD APRIL 2 “Applications of Probabilistic Inductive Logic Progr. ” Luc de Raedt, Univ. Freiburg, Stephen Muggleton, Univ. London. François Fages ICLP December 2003

Plan of the talk 1. Introduction 2. A simple algebra of cell molecules 3. Concurrent transition systems of biochemical reactions • Example of the mammalian cell cycle control 4. Temporal logic CTL as a query language • Computational results with BIOCHAM 5. Learning models • An experiment with inductive logic programming 6. Quantitative models • Simulation with differential equations • Constraint-based model checking 7. Conclusion François Fages ICLP December 2003

6. Quantitative Models Enzymatic reactions with rates k 1 k 2 k 3 E+S k 1 C k 2 E+P E+S k 3 C can be compiled by the law of mass action into a system of Ordinary Differential Equations d. E/dt = -k 1 ES+(k 2+k 3)C d. S/dt = -k 1 ES+k 3 C d. C/dt = k 1 ES-(k 2+k 3)C d. P/dt = k 2 C François Fages ICLP December 2003
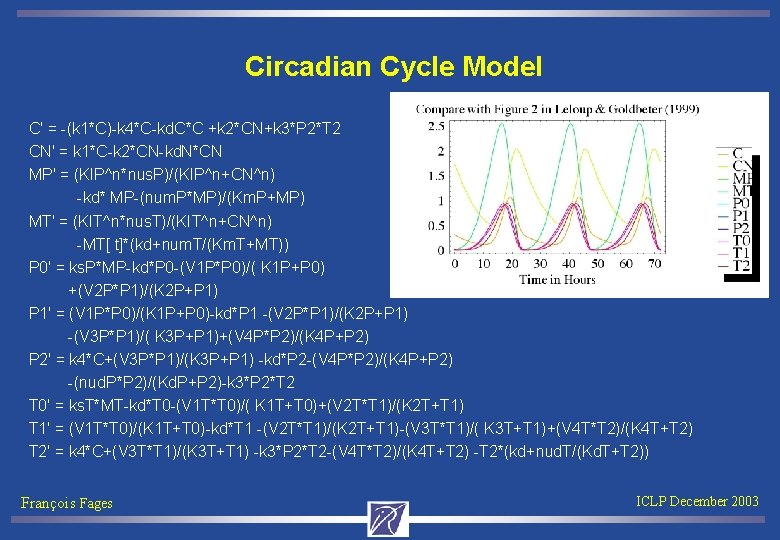
Circadian Cycle Model C' = -(k 1*C)-k 4*C-kd. C*C +k 2*CN+k 3*P 2*T 2 CN' = k 1*C-k 2*CN-kd. N*CN MP' = (KIP^n*nus. P)/(KIP^n+CN^n) -kd* MP-(num. P*MP)/(Km. P+MP) MT' = (KIT^n*nus. T)/(KIT^n+CN^n) -MT[ t]*(kd+num. T/(Km. T+MT)) P 0' = ks. P*MP-kd*P 0 -(V 1 P*P 0)/( K 1 P+P 0) +(V 2 P*P 1)/(K 2 P+P 1) P 1' = (V 1 P*P 0)/(K 1 P+P 0)-kd*P 1 -(V 2 P*P 1)/(K 2 P+P 1) -(V 3 P*P 1)/( K 3 P+P 1)+(V 4 P*P 2)/(K 4 P+P 2) P 2' = k 4*C+(V 3 P*P 1)/(K 3 P+P 1) -kd*P 2 -(V 4 P*P 2)/(K 4 P+P 2) -(nud. P*P 2)/(Kd. P+P 2)-k 3*P 2*T 2 T 0' = ks. T*MT-kd*T 0 -(V 1 T*T 0)/( K 1 T+T 0)+(V 2 T*T 1)/(K 2 T+T 1) T 1' = (V 1 T*T 0)/(K 1 T+T 0)-kd*T 1 -(V 2 T*T 1)/(K 2 T+T 1)-(V 3 T*T 1)/( K 3 T+T 1)+(V 4 T*T 2)/(K 4 T+T 2) T 2' = k 4*C+(V 3 T*T 1)/(K 3 T+T 1) -k 3*P 2*T 2 -(V 4 T*T 2)/(K 4 T+T 2) -T 2*(kd+nud. T/(Kd. T+T 2)) François Fages ICLP December 2003
![Gene Interaction Networks Gene interaction example [Bockmayr-Courtois 01] Hybrid Concurrent Constraint Programming HCC [Saraswat Gene Interaction Networks Gene interaction example [Bockmayr-Courtois 01] Hybrid Concurrent Constraint Programming HCC [Saraswat](http://slidetodoc.com/presentation_image/6398b33a54cbba9abb318bbe0ff07290/image-40.jpg)
Gene Interaction Networks Gene interaction example [Bockmayr-Courtois 01] Hybrid Concurrent Constraint Programming HCC [Saraswat et al. ] 2 genes x and y. dx/dt = 0. 01 – 0. 02*x if y < 0. 8 dx/dt = – 0. 02*x if y ≥ 0. 8 dy/dt = 0. 01*x François Fages ICLP December 2003

Concurrent Transition System Time discretized using Euler’s method (Runge-Kutta method in HCC): y < 0. 8 x’ = x + dt*(0. 01 -0. 02*x) , y’ = y + dt*0. 01*x y ≥ 0. 8 x’ = x + dt*(0. 01 -0. 02*x) , y’ = y + dt*0. 01*x Initial condition: x=0, y=0. CLP(R) program Init : - X=0, Y=0, p(X, Y): -X>=0, Y<0. 8, X 1=X-0. 02*X+0. 01, Y 1=Y+0. 01*X, p(X 1, Y 1). p(X, Y): -X>=0, Y>=0. 8, X 1=X-0. 02*X, Y 1=Y+0. 01*X, p(X 1, Y 1). François Fages ICLP December 2003
![Proving CTL properties by computing fixpointsof CLP programs Theorem [Delzanno Podelski 99] EF(f)=lfp(TP {p(x): Proving CTL properties by computing fixpointsof CLP programs Theorem [Delzanno Podelski 99] EF(f)=lfp(TP {p(x):](http://slidetodoc.com/presentation_image/6398b33a54cbba9abb318bbe0ff07290/image-42.jpg)
Proving CTL properties by computing fixpointsof CLP programs Theorem [Delzanno Podelski 99] EF(f)=lfp(TP {p(x): -f}), EG(f)=gfp(TP f ). Safety property AG( f) iff EF(f) iff init lfp(TP {f}) Liveness property AG(f 1 AF(f 2)) iff init lfp(TP f 1 gfp(TP {f 2}) ) Prolog-based implementation in CLP(R, B) [Delzanno 00] Applications to life in silico: Proof of protocols, cache consistency, etc. [Delzanno 01] François Fages ICLP December 2003

Deductive Model Checker DMC: Gene Interaction r(init, p(s_s, A, B), {A=0, B=0}). r(p(s_s, A, B), p(s_s, C, D), {A>=0, B>=0. 8, C=A-0. 02*A, D=B+0. 01*A}). r(p(s_s, A, B), p(s_s, C, D), {A>=0, B<0. 8, C=A-0. 02*A+0. 01, D=B+0. 01*A}). | ? - prop(P, S). P = unsafe, S = p: s*(x>=0. 6) | ? - ti. Property satisfied. Execution time 0. 0 | ? - ls. s(0, p(s_s, A, _), {A>=0. 6}, 1, (0, 0)). François Fages ICLP December 2003

Demonstration DMC (continued) | ? - prop(P, S). P = unsafe, S = p: s*(x>=0. 2) ? | ? - ti. Property NOT satisfied. Execution time 1. 5 | ? - ls. s(0, p(s_s, A, _), {A>=0. 2}, 1, (0, 0)). s(1, p(s_s, A, B), {B<0. 8, B>=-0. 0, A>=0. 19387755102040816}, 2, (2, 1)). … s(26, p(s_s, A, B), {B>=0. 0, A>=0. 0, B+0. 1982676351105516*A<0. 7741338175552753}, 27, (2, 26)). s(27, init, {}, 28, (1, 27)). François Fages ICLP December 2003

7. Conclusion The great ambition of logic programming is to make of programming a modeling task in the first place, with equations, constraints and logical formulae. In this respect, computational molecular biology offers numerous challenges to the logic programming community at large. Besides combinatorial search and optimization problems coming from molecular biology (DNA and protein sequence comparison, protein structure prediction, …) there is a need to model globally the system at hand automate reasoning on all its possible behaviors. François Fages ICLP December 2003

Conclusion The biochemical abstract machine BIOCHAM project aims at developing: Qualitative models of complex biochemical processes: • Intracellular and extracellular signaling, cell-cycle control, … [http: //contraintes. inria. fr/CMBSlib] • Prolog-based implementation + BDD symbolic model-checking • ILP-based learning of models from temporal properties [6 th. PCRD APRIL 2] Membranes and transportation not modeled Bitonal algebras [Cardelli et al. 03] Bio. Ambients, Brane calculi [Cardelli et al. 03] François Fages ICLP December 2003

Perspectives for LP Quantitative models: • Differential equations • Hybrid discrete-continuous time models • Hybrid concurrent constraint programming [Bockmayr-Courtois-Eveillard 03] • CLP-based model-checking [Delzanno-Podelski 99] [Chabrier-Fages 03] Multi-scale molecular-electro-physiological models [Sorine et al. 03] http: //www-rocq. inria. fr/sosso/icema 2 http: //www. sci. sdsu. edu/movies François Fages ICLP December 2003
- Slides: 47