SWITCHING TO DTG3 TC FIXEDDOSE COMBINATION FDC IS

SWITCHING TO DTG/3 TC FIXED-DOSE COMBINATION (FDC) IS NON-INFERIOR TO CONTINUING A TAF-BASED REGIMEN IN MAINTAINING VIROLOGIC SUPPRESSION THROUGH 48 WEEKS (TANGO STUDY) J van Wyk, 1 F Ajana, 2 F Bisshop, 3 S De. Wit, 4 O Osiyemi, 5 J Portilla, 6 JP Routy, 7 C Wyen, 8 M Ait-Khaled, 1 M-C Nascimento, 1 KA Pappa, 9 R Wang, 9 J Wright, 10 AR Tenorio, 9 B Wynne, 9 M Aboud, 1 MJ Gartland, 9 KY Smith 9 1 Vii. V Healthcare, Brentford, UK; 2 Centre Hospitalier de Tourcoing, France; 3 Holdsworth House Medical Brisbane, Queensland, Australia; 4 CHU Saint-Pierre, Brussels, Belgium; 5 Triple O Research Institute PA, West Palm Beach, FL, USA; 6 Hospital General Universitario de Alicante, Spain; 7 Mc. Gill University Health Centre, Montreal, QC, Canada; 8 Praxis am Ebertplatz, Cologne, Germany; 9 Vii. V Healthcare, Research Triangle Park, NC, USA; 10 Glaxo. Smith. Kline, Stockley Park, UK 10 th IAS Conference on HIV Science; July 21– 24, 2019; Mexico City, Mexico

DISCLOSURES • Jean van Wyk is an employee of Vii. V Healthcare van Wyk et al. IAS 2019; Mexico City, Mexico. Slides WEAB 0403 LB. 10 th IAS Conference on HIV Science; July 21– 24, 2019; Mexico City, Mexico 2

BACKGROUND • Two-drug regimens (2 DRs) reduce drug exposure for PLWHIV who need lifelong ART 1 • In the Week 48 primary analysis of the GEMINI studies, DTG + 3 TC was non-inferior to DTG + TDF/FTC in HIV-1–infected treatment-naive adults 2 • The results led to the marketing authorization of DTG/3 TC (DOVATO) as a once-daily, singletablet 2 DR by the US Food and Drug Administration and the European Medicines Agency • TANGO is an ongoing phase III, non-inferiority trial evaluating efficacy and safety of a switch to DTG/3 TC FDC in HIV-1–infected adults with virologic suppression on a 3 - or 4 -drug TAF-based regimen • The Week 48 primary analysis results from TANGO are presented here 1. Kelly et al. Drugs. 2016; 76: 523 -531. 2. Cahn et al. Lancet. 2019; 393: 143 -155. van Wyk et al. IAS 2019; Mexico City, Mexico. Slides WEAB 0403 LB. 10 th IAS Conference on HIV Science; July 21– 24, 2019; Mexico City, Mexico 3
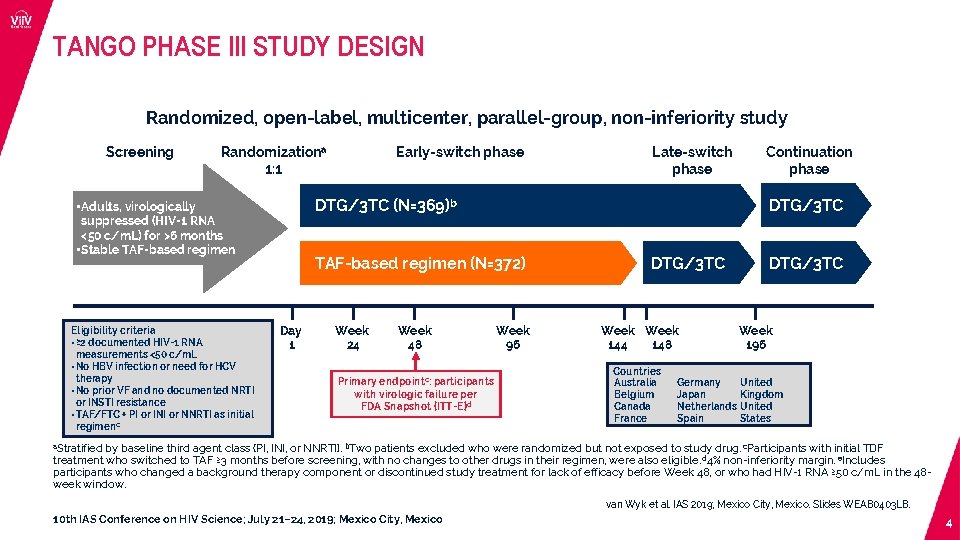
TANGO PHASE III STUDY DESIGN Randomized, open-label, multicenter, parallel-group, non-inferiority study Screening Randomizationa 1: 1 Late-switch phase TAF-based regimen (N=372) Day 1 Week 24 Week 48 Primary endpointc: participants with virologic failure per FDA Snapshot (ITT-E)d Continuation phase DTG/3 TC (N=369)b • Adults, virologically suppressed (HIV-1 RNA <50 c/m. L) for >6 months • Stable TAF-based regimen Eligibility criteria • ≥ 2 documented HIV-1 RNA measurements <50 c/m. L • No HBV infection or need for HCV therapy • No prior VF and no documented NRTI or INSTI resistance • TAF/FTC + PI or INI or NNRTI as initial regimenc Early-switch phase Week 96 DTG/3 TC Week 144 148 Countries Australia Belgium Canada France Germany Japan Netherlands Spain DTG/3 TC Week 196 United Kingdom United States a. Stratified by baseline third agent class (PI, INI, or NNRTI). b. Two patients excluded who were randomized but not exposed to study drug. c. Participants with initial TDF treatment who switched to TAF ≥ 3 months before screening, with no changes to other drugs in their regimen, were also eligible. d 4% non-inferiority margin. e. Includes participants who changed a background therapy component or discontinued study treatment for lack of efficacy before Week 48, or who had HIV-1 RNA ≥ 50 c/m. L in the 48 week window. van Wyk et al. IAS 2019; Mexico City, Mexico. Slides WEAB 0403 LB. 10 th IAS Conference on HIV Science; July 21– 24, 2019; Mexico City, Mexico 4
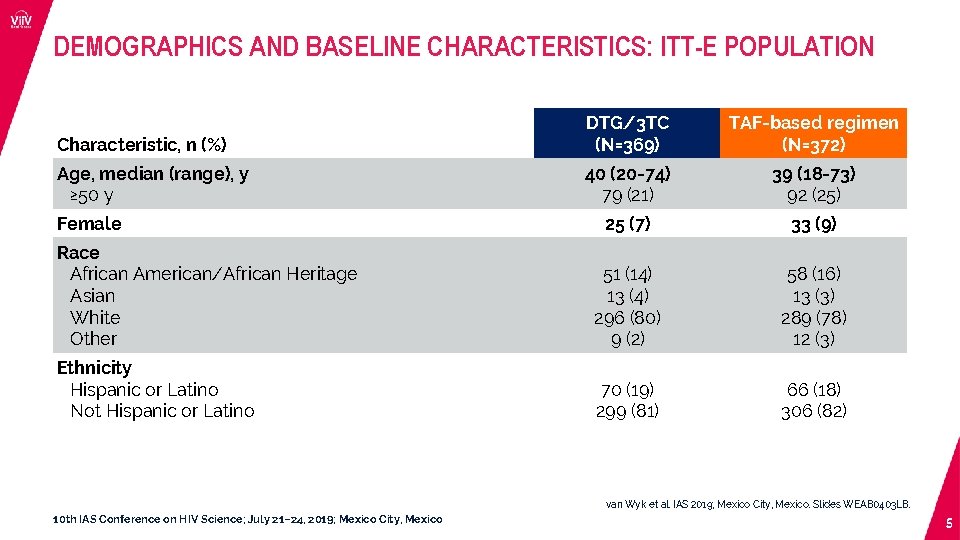
DEMOGRAPHICS AND BASELINE CHARACTERISTICS: ITT-E POPULATION Characteristic, n (%) DTG/3 TC (N=369) TAF-based regimen (N=372) Age, median (range), y ≥ 50 y 40 (20 -74) 79 (21) 39 (18 -73) 92 (25) 25 (7) 33 (9) Race African American/African Heritage Asian White Other 51 (14) 13 (4) 296 (80) 9 (2) 58 (16) 13 (3) 289 (78) 12 (3) Ethnicity Hispanic or Latino Not Hispanic or Latino 70 (19) 299 (81) 66 (18) 306 (82) Female van Wyk et al. IAS 2019; Mexico City, Mexico. Slides WEAB 0403 LB. 10 th IAS Conference on HIV Science; July 21– 24, 2019; Mexico City, Mexico 5

DEMOGRAPHICS AND BASELINE CHARACTERISTICS: ITT-E POPULATION (cont) DTG/3 TC (N=369) TAF-based regimen (N=372) 682 (133 -1904) 720 (119 -1810) CD 4+ cell count, cells/mm 3 <350 ≥ 350 35 (9) 334 (91) 30 (8) 342 (92) Baseline third agent class INI EVG/c 289 (78) 243 (66) 296 (80) 249 (67) NNRTI RPV 51 (14) 43 (12) 48 (13) 45 (12) PI 29 (8) 25 (7) 28 (8) 27 (7) 33. 8 (7. 1 -201. 2) 35. 1 (7. 0 -160. 8) Characteristic, n (%) Median CD 4+ cell count (range), cells/mm 3 b. DRV Duration of ART before Day 1, median (range), mo van Wyk et al. IAS 2019; Mexico City, Mexico. Slides WEAB 0403 LB. 10 th IAS Conference on HIV Science; July 21– 24, 2019; Mexico City, Mexico 6
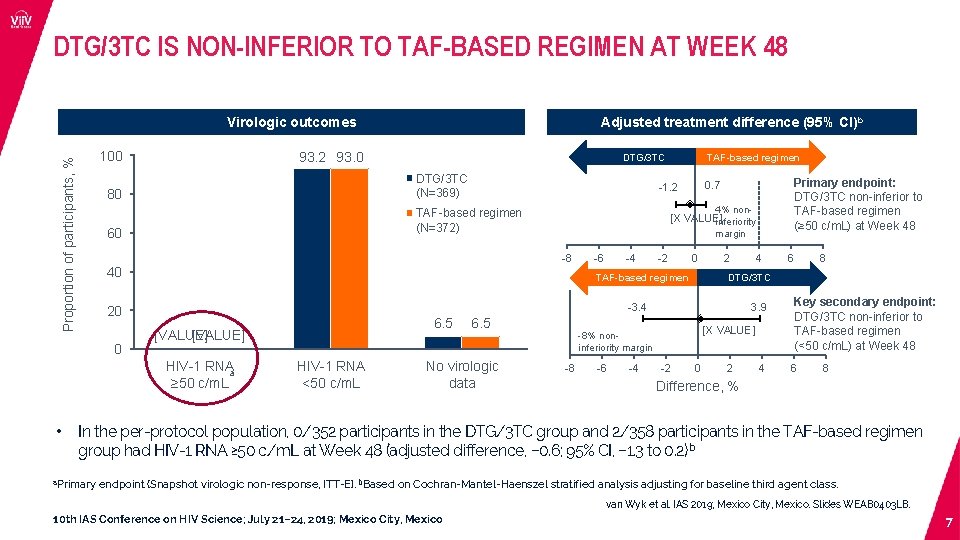
DTG/3 TC IS NON-INFERIOR TO TAF-BASED REGIMEN AT WEEK 48 Virologic outcomes Proportion of participants, % 100 93. 2 93. 0 DTG/3 TC (N=369) 80 TAF-based regimen 4% non- [X VALUE] inferiority margin -8 40 -6 -4 -2 0 TAF-based regimen 2 6. 5 [VALUE] HIV-1 RNA a ≥ 50 c/m. L HIV-1 RNA <50 c/m. L 6. 5 No virologic data 3. 9 [X VALUE] -8% noninferiority margin -8 -6 -4 4 -2 6 8 DTG/3 TC -3. 4 20 Primary endpoint: DTG/3 TC non-inferior to TAF-based regimen (≥ 50 c/m. L) at Week 48 0. 7 -1. 2 TAF-based regimen (N=372) 60 0 • Adjusted treatment difference (95% CI)b 0 2 4 Key secondary endpoint: DTG/3 TC non-inferior to TAF-based regimen (<50 c/m. L) at Week 48 6 8 Difference, % In the per-protocol population, 0/352 participants in the DTG/3 TC group and 2/358 participants in the TAF-based regimen group had HIV-1 RNA ≥ 50 c/m. L at Week 48 (adjusted difference, − 0. 6; 95% CI, − 1. 3 to 0. 2) b a. Primary endpoint (Snapshot virologic non-response, ITT-E). b. Based on Cochran-Mantel-Haenszel stratified analysis adjusting for baseline third agent class. van Wyk et al. IAS 2019; Mexico City, Mexico. Slides WEAB 0403 LB. 10 th IAS Conference on HIV Science; July 21– 24, 2019; Mexico City, Mexico 7
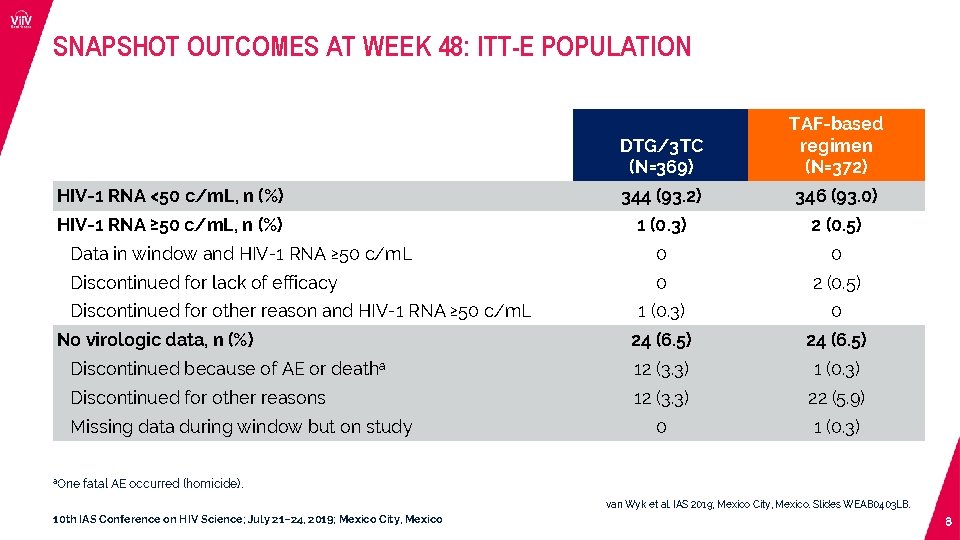
SNAPSHOT OUTCOMES AT WEEK 48: ITT-E POPULATION DTG/3 TC (N=369) TAF-based regimen (N=372) HIV-1 RNA <50 c/m. L, n (%) 344 (93. 2) 346 (93. 0) HIV-1 RNA ≥ 50 c/m. L, n (%) 1 (0. 3) 2 (0. 5) Data in window and HIV-1 RNA ≥ 50 c/m. L 0 0 Discontinued for lack of efficacy 0 2 (0. 5) 1 (0. 3) 0 24 (6. 5) Discontinued because of AE or deatha 12 (3. 3) 1 (0. 3) Discontinued for other reasons 12 (3. 3) 22 (5. 9) 0 1 (0. 3) Discontinued for other reason and HIV-1 RNA ≥ 50 c/m. L No virologic data, n (%) Missing data during window but on study a. One fatal AE occurred (homicide). van Wyk et al. IAS 2019; Mexico City, Mexico. Slides WEAB 0403 LB. 10 th IAS Conference on HIV Science; July 21– 24, 2019; Mexico City, Mexico 8
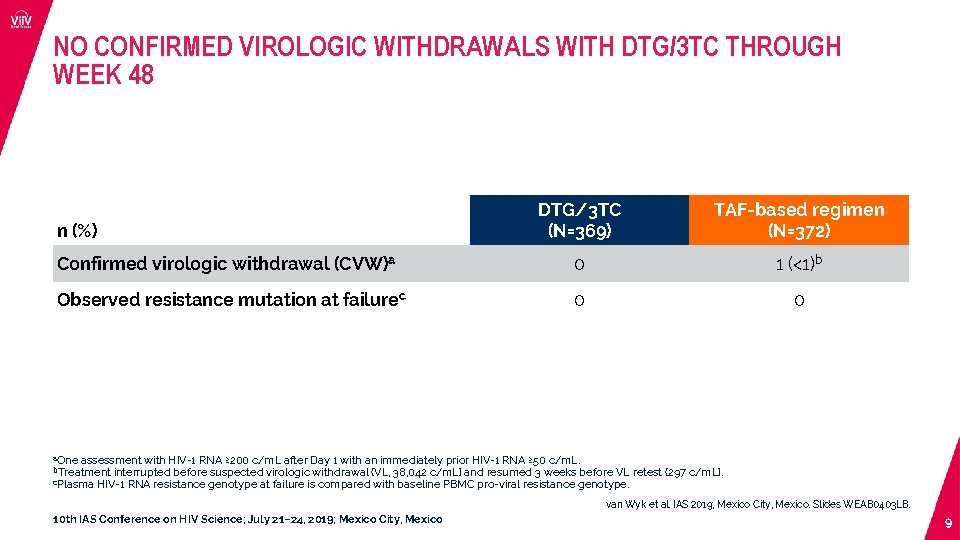
NO CONFIRMED VIROLOGIC WITHDRAWALS WITH DTG/3 TC THROUGH WEEK 48 DTG/3 TC (N=369) TAF-based regimen (N=372) Confirmed virologic withdrawal (CVW)a 0 1 (<1)b Observed resistance mutation at failurec 0 0 n (%) a. One assessment with HIV-1 RNA ≥ 200 c/m. L after Day 1 with an immediately prior HIV-1 RNA ≥ 50 c/m. L. interrupted before suspected virologic withdrawal (VL, 38, 042 c/m. L) and resumed 3 weeks before VL retest (297 c/m. L). c. Plasma HIV-1 RNA resistance genotype at failure is compared with baseline PBMC pro-viral resistance genotype. b. Treatment van Wyk et al. IAS 2019; Mexico City, Mexico. Slides WEAB 0403 LB. 10 th IAS Conference on HIV Science; July 21– 24, 2019; Mexico City, Mexico 9
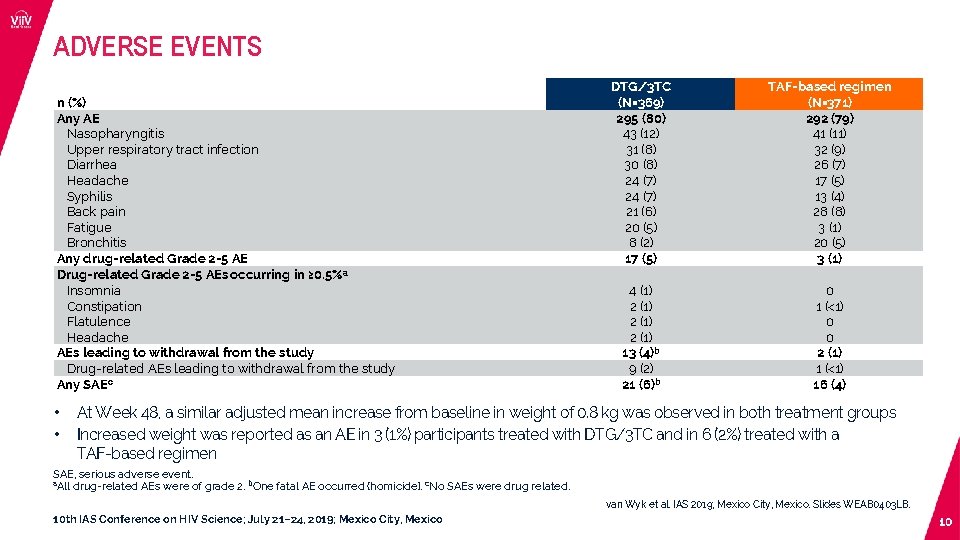
ADVERSE EVENTS n (%) Any AE Nasopharyngitis Upper respiratory tract infection Diarrhea Headache Syphilis Back pain Fatigue Bronchitis Any drug-related Grade 2 -5 AE Drug-related Grade 2 -5 AEs occurring in ≥ 0. 5%a Insomnia Constipation Flatulence Headache AEs leading to withdrawal from the study Drug-related AEs leading to withdrawal from the study Any SAEc • • DTG/3 TC (N=369) 295 (80) 43 (12) 31 (8) 30 (8) 24 (7) 21 (6) 20 (5) 8 (2) 17 (5) TAF-based regimen (N=371) 292 (79) 41 (11) 32 (9) 26 (7) 17 (5) 13 (4) 28 (8) 3 (1) 20 (5) 3 (1) 4 (1) 2 (1) 13 (4)b 9 (2) 21 (6)b 0 1 (<1) 0 0 2 (1) 1 (<1) 16 (4) At Week 48, a similar adjusted mean increase from baseline in weight of 0. 8 kg was observed in both treatment groups Increased weight was reported as an AE in 3 (1%) participants treated with DTG/3 TC and in 6 (2%) treated with a TAF-based regimen SAE, serious adverse event. a. All drug-related AEs were of grade 2. b. One fatal AE occurred (homicide). c. No SAEs were drug related. van Wyk et al. IAS 2019; Mexico City, Mexico. Slides WEAB 0403 LB. 10 th IAS Conference on HIV Science; July 21– 24, 2019; Mexico City, Mexico 10
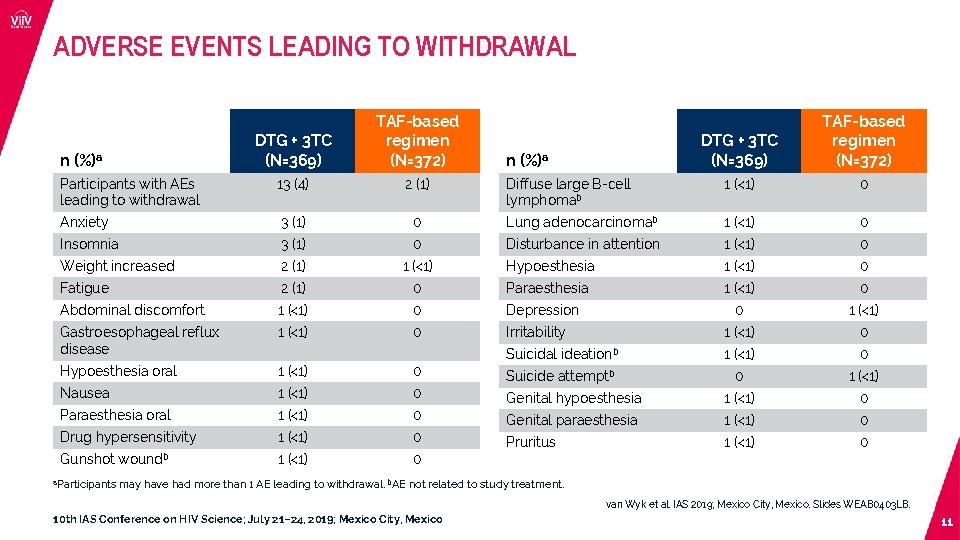
ADVERSE EVENTS LEADING TO WITHDRAWAL DTG + 3 TC (N=369) TAF-based regimen (N=372) Diffuse large B-cell lymphomab 1 (<1) 0 0 Lung adenocarcinomab 1 (<1) 0 3 (1) 0 Disturbance in attention 1 (<1) 0 Weight increased 2 (1) 1 (<1) Hypoesthesia 1 (<1) 0 Fatigue 2 (1) 0 Paraesthesia 1 (<1) 0 Abdominal discomfort 1 (<1) 0 Depression 0 1 (<1) Gastroesophageal reflux disease 1 (<1) 0 Irritability 1 (<1) 0 Suicidal ideationb 1 (<1) 0 Hypoesthesia oral 1 (<1) 0 Suicide attemptb 0 1 (<1) Nausea 1 (<1) 0 Genital hypoesthesia 1 (<1) 0 Paraesthesia oral 1 (<1) 0 Genital paraesthesia 1 (<1) 0 Drug hypersensitivity 1 (<1) 0 Pruritus 1 (<1) 0 DTG + 3 TC (N=369) TAF-based regimen (N=372) Participants with AEs leading to withdrawal 13 (4) 2 (1) Anxiety 3 (1) Insomnia n (%)a Gunshot a. Participants woundb n (%)a may have had more than 1 AE leading to withdrawal. b. AE not related to study treatment. van Wyk et al. IAS 2019; Mexico City, Mexico. Slides WEAB 0403 LB. 10 th IAS Conference on HIV Science; July 21– 24, 2019; Mexico City, Mexico 11
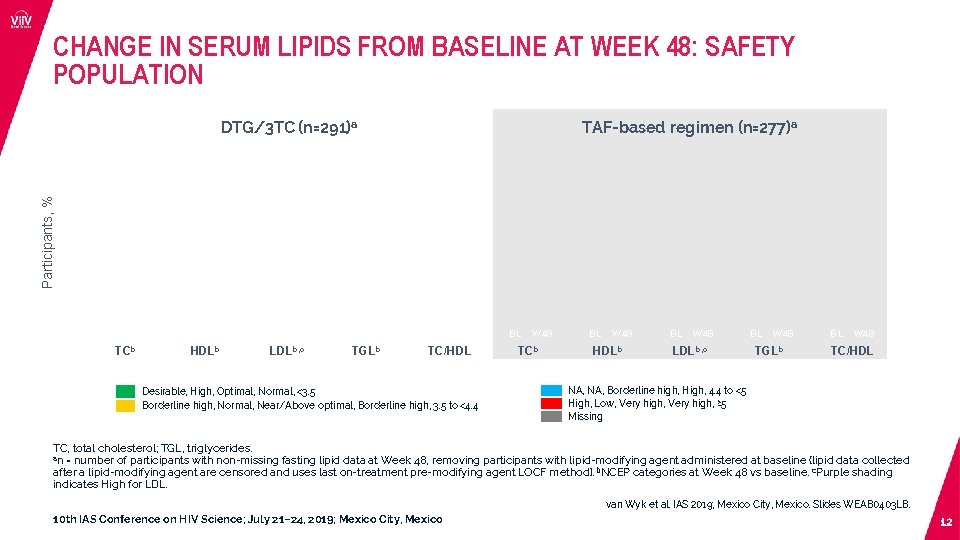
CHANGE IN SERUM LIPIDS FROM BASELINE AT WEEK 48: SAFETY POPULATION TAF-based regimen (n=277)a Participants, % DTG/3 TC (n=291)a BL W 48 TCb BL W 48 HDLb BL W 48 LDLb, c BL W 48 TGLb BL W 48 TC/HDL Desirable, High, Optimal, Normal, <3. 5 Borderline high, Normal, Near/Above optimal, Borderline high, 3. 5 to <4. 4 BL W 48 TCb BL W 48 HDLb BL W 48 LDLb, c BL W 48 TGLb BL W 48 TC/HDL NA, Borderline high, High, 4. 4 to <5 High, Low, Very high, ≥ 5 Missing TC, total cholesterol; TGL, triglycerides. an = number of participants with non-missing fasting lipid data at Week 48, removing participants with lipid-modifying agent administered at baseline (lipid data collected after a lipid-modifying agent are censored and uses last on-treatment pre-modifying agent LOCF method). b. NCEP categories at Week 48 vs baseline. c. Purple shading indicates High for LDL. van Wyk et al. IAS 2019; Mexico City, Mexico. Slides WEAB 0403 LB. 10 th IAS Conference on HIV Science; July 21– 24, 2019; Mexico City, Mexico 12
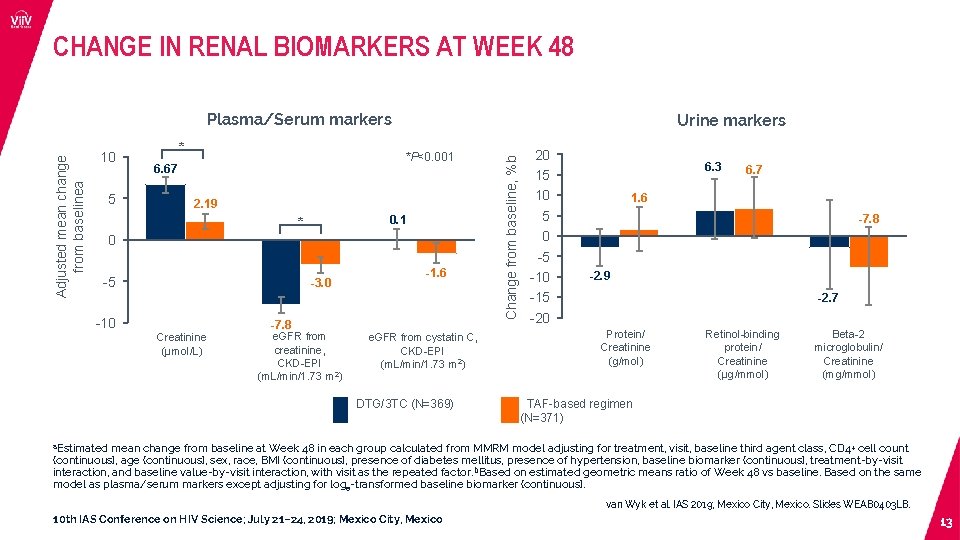
CHANGE IN RENAL BIOMARKERS AT WEEK 48 10 5 * Urine markers *P<0. 001 6. 67 2. 19 0. 1 * 0 -5 -3. 0 -1. 6 -7. 8 Creatinine (µmol/L) e. GFR from creatinine, CKD-EPI (m. L/min/1. 73 m 2) e. GFR from cystatin C, CKD-EPI (m. L/min/1. 73 m 2) DTG/3 TC (N=369) Change from baseline, %b Adjusted mean change from baselinea Plasma/Serum markers 20 15 10 5 0 -5 -10 -15 -20 6. 3 6. 7 1. 6 -7. 8 -2. 9 -2. 7 Protein/ Creatinine (g/mol) Retinol-binding protein/ Creatinine (µg/mmol) Beta-2 microglobulin/ Creatinine (mg/mmol) TAF-based regimen (N=371) a. Estimated mean change from baseline at Week 48 in each group calculated from MMRM model adjusting for treatment, visit, baseline third agent class, CD 4+ cell count (continuous), age (continuous), sex, race, BMI (continuous), presence of diabetes mellitus, presence of hypertension, baseline biomarker (continuous), treatment-by-visit interaction, and baseline value-by-visit interaction, with visit as the repeated factor. b. Based on estimated geometric means ratio of Week 48 vs baseline. Based on the same model as plasma/serum markers except adjusting for loge-transformed baseline biomarker (continuous). van Wyk et al. IAS 2019; Mexico City, Mexico. Slides WEAB 0403 LB. 10 th IAS Conference on HIV Science; July 21– 24, 2019; Mexico City, Mexico 13
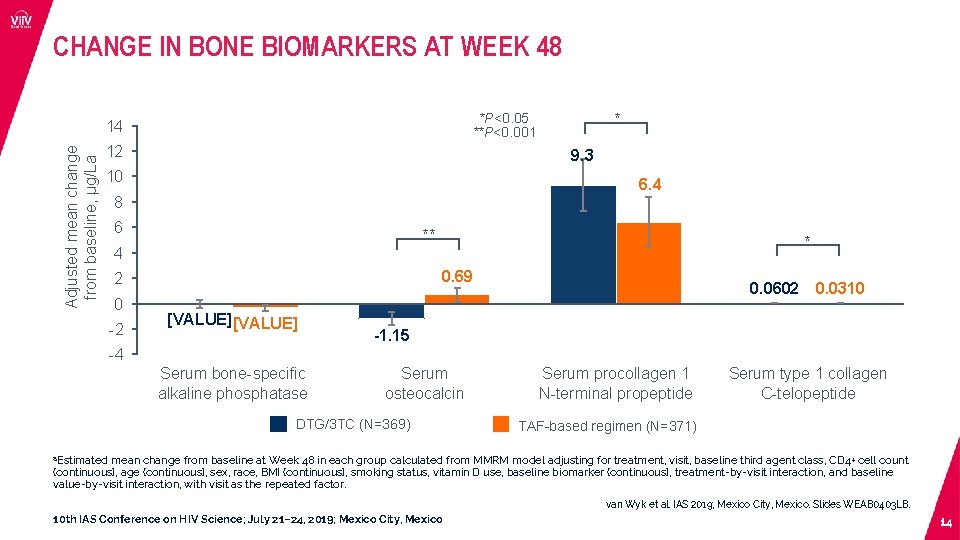
CHANGE IN BONE BIOMARKERS AT WEEK 48 Adjusted mean change from baseline, µg/La * *P<0. 05 **P<0. 001 14 12 9. 3 10 6. 4 8 6 ** 4 0. 69 2 0 -2 * [VALUE] 0. 0602 0. 0310 -1. 15 -4 Serum bone-specific alkaline phosphatase Serum osteocalcin DTG/3 TC (N=369) Serum procollagen 1 N-terminal propeptide Serum type 1 collagen C-telopeptide TAF-based regimen (N=371) a. Estimated mean change from baseline at Week 48 in each group calculated from MMRM model adjusting for treatment, visit, baseline third agent class, CD 4+ cell count (continuous), age (continuous), sex, race, BMI (continuous), smoking status, vitamin D use, baseline biomarker (continuous), treatment-by-visit interaction, and baseline value-by-visit interaction, with visit as the repeated factor. van Wyk et al. IAS 2019; Mexico City, Mexico. Slides WEAB 0403 LB. 10 th IAS Conference on HIV Science; July 21– 24, 2019; Mexico City, Mexico 14

VIROLOGIC RESPONSE BY PRO-VIRAL M 184 V/I STATUS: POST-HOC ANALYSIS OF STORED BASELINE SAMPLES DTG/3 TC (N=318)a TAF-based regimen (N=308)a 4 (1) 3 (1) HIV-1 RNA <50 c/m. L at Week 48 c 4 (100) 3 (100) HIV-1 RNA ≥ 50 c/m. L at Week 48 c 0 0 No pro-viral M 184 V/I at baselineb 314 (99) 305 (99) HIV-1 RNA <50 c/m. L at Week 48 c 314 (100) 303 (99) HIV-1 RNA ≥ 50 c/m. L at Week 48 c 0 2 (1) Participants, n (%) Pro-viral M 184 V/I at baselineb • All participants in the DTG/3 TC group with pro-viral M 184 V/I at baseline maintained HIV-1 RNA <50 c/m. L at Week 48 a. ITT-E population with baseline pro-viral DNA resistance result (Geno. Sure Archive®, Monogram Biosciences) with ≥ 1 post-baseline on-treatment HIV-1 RNA viral load result and excluding participants who have withdrawn from the study because of protocol deviation (n=5). b. Percentages based on N. c. Last available on-treatment HIV-1 RNA viral load through Week 48 analysis using last on-treatment HIV-1 RNA viral load through Week 48 or before discontinuation (percentages based on baseline mutation status n). van Wyk et al. IAS 2019; Mexico City, Mexico. Slides WEAB 0403 LB. 10 th IAS Conference on HIV Science; July 21– 24, 2019; Mexico City, Mexico 15

CONCLUSIONS • Switching to DTG/3 TC FDC was non-inferior to remaining on a TAF-based regimen through Week 48 in ART-experienced, virologically suppressed adults • No confirmed virologic withdrawals in the DTG/3 TC group • Zero resistance development in the DTG/3 TC group • The safety profile of DTG/3 TC FDC was consistent with the DTG and 3 TC labels • These data support the use of DTG/3 TC as a new robust switch option without increased risk of virologic failure or resistance and with reduced ART exposure van Wyk et al. IAS 2019; Mexico City, Mexico. Slides WEAB 0403 LB. 10 th IAS Conference on HIV Science; July 21– 24, 2019; Mexico City, Mexico 16

ACKNOWLEDGMENTS • We thank the study participants; their families and caregivers; investigators and site staff who participated in the study; and the Vii. V Healthcare, Glaxo. Smith. Kline, Pharmaceutical Product Development, and Phastar study team members Australia Baker Bisshop Bloch Mc. Mahon Moore Pell Roth Schmidt Smith Woolley Belgium De Wit Florence Lacor Vandekerckhove Vandercam Canada Kasper Le. Blanc Routy Sasseville Walmsley France Ajana Bonnet Girard Katlama Philibert Pugliese Yazdanpanah Germany Arasteh Bogner Degen Germany (cont) Jäger Krznaric Lutz Postel Scholten Spinner Stellbrink Stoll Wyen Japan Adachi Igari Yokomaku Netherlands Rijnders Spain Angel-Moreno Antela Arribas Lopez Bernal Morell Bravo Urbieta Crusells Canales Deig Comerma Domingo Force Galinda Puerto Gil Anguita Górgolas Martinez Chamorro Masia Canuto Merino Munoz Montero-Alonso Ocampo Hermida Pasquau Liano Pérez Elias Perez Stachowski Spain (cont) Pineda Podzamczer Palter Portilla Sogorb Rubio Santos Fernandez Santos Gonzalez Sanz Moreno Vera Mendez Vergas Garcia Viciana United Kingdom Arumainayagam Chaponda Clarke Gompels Pett Ross Ustianowski USA Alozie Batra Benson Berhe Bolivar Brennan Brinson Crofoot Cruickshank Cunningham Daar De. Jesus Edelstein Farabi Felizarta Flamm Goldstein Gupta USA (cont) Hagins Henry Johnson Katner Kinder Martorell Mayer Mc. Donald Mc. Kellar Melton Mills Mounzer Ortiz Osiyemi Park Patel Prelutsky Ramgopal USA (cont) Ravi Reddy Rodriguez Ruane Scarsella Schneider Schrader Schreibman Simon Sims Sinclair Stein Thedinger Towner Vanig Wohlfeiler Wurapa Zane van Wyk et al. IAS 2019; Mexico City, Mexico. Slides WEAB 0403 LB. 10 th IAS Conference on HIV Science; July 21– 24, 2019; Mexico City, Mexico 17
- Slides: 17