SWITCHING FROM FONDAPARINUX TO BIVALIRUDIN OR UFRACTIONATRED HEPARIN

SWITCHING FROM FONDAPARINUX TO BIVALIRUDIN OR UFRACTIONATRED HEPARIN IN PATIENTS WITH ACS WITHOUT STEMI UNDERGOING PCI SWITCH III Ron Waksman, MD on Behalf of the SWITCH III Investigators

Ron Waksman, MD Consulting Fee Abbott Laboratories Biotronik, Inc. Boston Scientific Corporation Medtronic, Inc. Merck and Company, Inc.

Honoraria Abbott Laboratories Boston Scientific Corporation Medtronic, Inc. Consulting Fee Merck and Company, Inc.

Background I • Recent studies have demonstrated that the treatment of patients with low molecular weight heparin followed by early angiography and then by appropriate medical PCI, or CABG MANAGEMENT is safe and effective. • The OASIS 5 study demonstrated the superiority of fondaparinux over enoxaparin for patients presenting with ACS. However, for patients undergoing PCI there was increase of thrombus within the catheter • This suggested that although fondaparinux is beneficial for the overall ACS patient there is a need for additional anticoagulation during PCI for patients presenting on fondaparinux.

Purpose and Aims • The primary objective was to evaluate the safety of switching from fondaparinux to either unfractionated heparin or bivalirudin for patients experiencing acute coronary syndrome undergoing percutaneous coronary angioplasty.

Endpoints • Primary: in hospital major bleed. Major bleed defined as clinically overt bleeding that is: • Fatal • Symptomatic intracranial hemorrhage • Retroperitoneal hemorrhage • Intraocular hemorrhage leading to significant vision loss • Decrease in hemoglobin of at least 3. 0 g/d. L (with each blood transfusion unit counting for 1 g/d. L of Hb) • Bleed requiring transfusion of two or more units of RBCs or equivalent whole blood • Secondary: in hospital death (non-hemorrhagic related), vascular access site complications, myocardial infarction, need for repeat revascularization, procedural complication and catheter thrombosis.
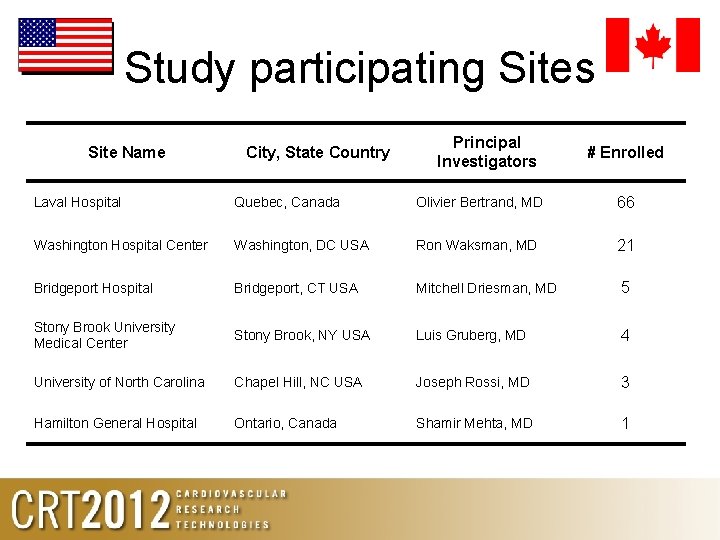
Study participating Sites Site Name City, State Country Principal Investigators # Enrolled Laval Hospital Quebec, Canada Olivier Bertrand, MD 66 Washington Hospital Center Washington, DC USA Ron Waksman, MD 21 Bridgeport Hospital Bridgeport, CT USA Mitchell Driesman, MD 5 Stony Brook University Medical Center Stony Brook, NY USA Luis Gruberg, MD 4 University of North Carolina Chapel Hill, NC USA Joseph Rossi, MD 3 Hamilton General Hospital Ontario, Canada Shamir Mehta, MD 1

Main Inclusion Criteria • • • Patient is male or female, >18 years of age Patient presents with ACS, unstable angina, or NSTEMI defined as at least one of criteria: Elevated CK-MB or Troponin I or T ECG changes indicative of ischemia Patient is scheduled for angiography, with possible angioplasty, evaluation of their coronary disease Patient is eligible for PCI Patient requires PCI of at least one coronary artery Target lesion stenosis is <100%

Major Exclusion Criteria • • Patients with STEMI within 48 hours Patient weighs more than 400 lbs Patient presents on or has received UFH, bivalirudin, GP IIb/IIIa inhibitors or LMWH within 24 hours Patient with known conditions of bleeding diathesis or actively bleeding within the previous 6 months Patient with cardiogenic shock or required IABP Patient on Coumadin therapy Patient who had a major or minor stroke or major surgery within past 6 months Patient with creatinine ≥ 3. 0, s/p renal transplant, or on dialysis Patient with platelet count <100, 000 Known allergy to fondaparinux, ASA, Plavix, Ticlid, heparin, Angiomax or contrast that cannot be managed medically Contraindication to LMWH, UFH or Angiomax
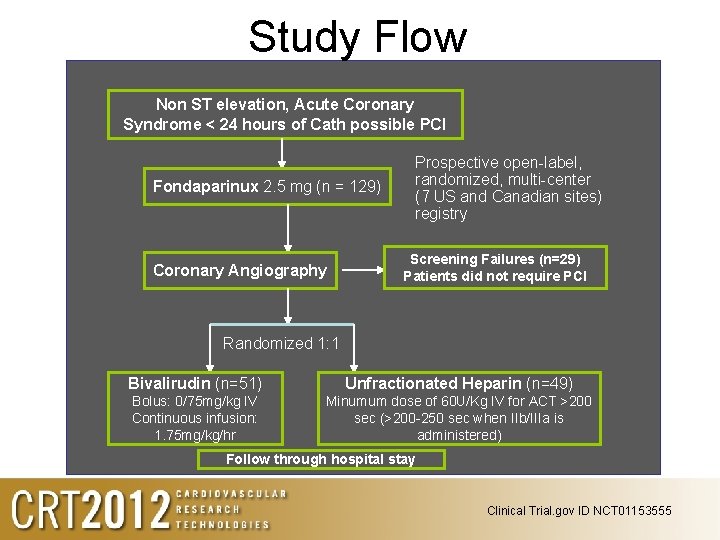
Study Flow Non ST elevation, Acute Coronary Syndrome < 24 hours of Cath possible PCI Fondaparinux 2. 5 mg (n = 129) Coronary Angiography Prospective open-label, randomized, multi-center (7 US and Canadian sites) registry Screening Failures (n=29) Patients did not require PCI Randomized 1: 1 Bivalirudin (n=51) Unfractionated Heparin (n=49) Bolus: 0/75 mg/kg IV Continuous infusion: 1. 75 mg/kg/hr Minumum dose of 60 U/Kg IV for ACT >200 sec (>200 -250 sec when IIb/IIIa is administered) Follow through hospital stay Clinical Trial. gov ID NCT 01153555
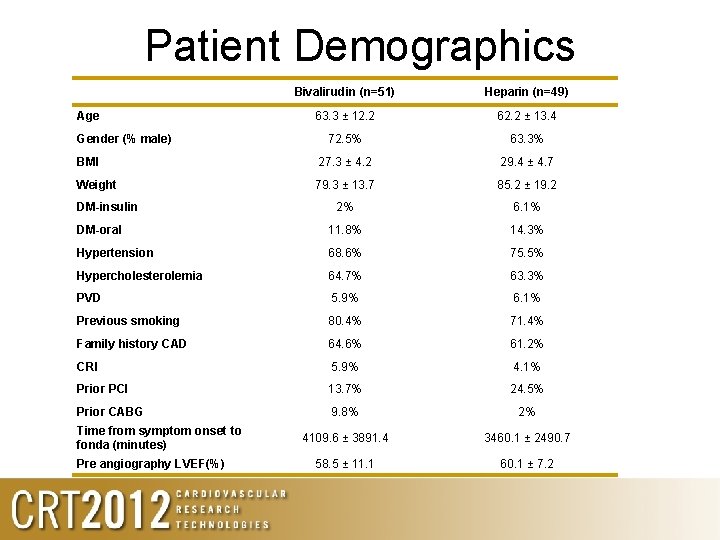
Patient Demographics Bivalirudin (n=51) Heparin (n=49) 63. 3 ± 12. 2 62. 2 ± 13. 4 72. 5% 63. 3% BMI 27. 3 ± 4. 2 29. 4 ± 4. 7 Weight 79. 3 ± 13. 7 85. 2 ± 19. 2 2% 6. 1% DM-oral 11. 8% 14. 3% Hypertension 68. 6% 75. 5% Hypercholesterolemia 64. 7% 63. 3% PVD 5. 9% 6. 1% Previous smoking 80. 4% 71. 4% Family history CAD 64. 6% 61. 2% CRI 5. 9% 4. 1% Prior PCI 13. 7% 24. 5% Prior CABG 9. 8% 2% 4109. 6 ± 3891. 4 3460. 1 ± 2490. 7 58. 5 ± 11. 1 60. 1 ± 7. 2 Age Gender (% male) DM-insulin Time from symptom onset to fonda (minutes) Pre angiography LVEF(%)

Intraprocedural Anticoagulation Bivalirudin (n=51) Heparin (n=49) Unfractionalted Heparin* 5. 9% 98. 0% Bivalirudin* 98. 0% 2. 0% LMWH 0. 0% GP IIb/IIIA Inhibitor 3. 9% 12. 2% Lytic 0. 0% *one patient in each group received the other randomization assignment. Two additional patients receive UFH prior to randomization drug administration at the physicians discretion.
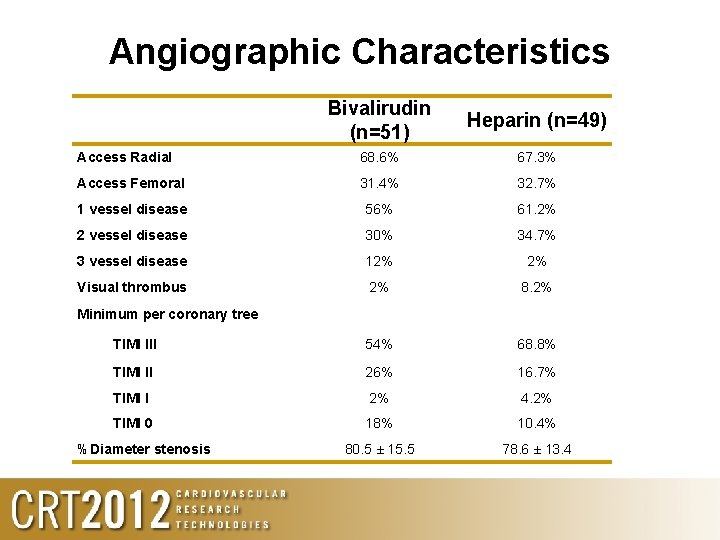
Angiographic Characteristics Bivalirudin (n=51) Heparin (n=49) Access Radial 68. 6% 67. 3% Access Femoral 31. 4% 32. 7% 1 vessel disease 56% 61. 2% 2 vessel disease 30% 34. 7% 3 vessel disease 12% 2% Visual thrombus 2% 8. 2% TIMI III 54% 68. 8% TIMI II 26% 16. 7% TIMI I 2% 4. 2% TIMI 0 18% 10. 4% 80. 5 ± 15. 5 78. 6 ± 13. 4 Minimum per coronary tree %Diameter stenosis
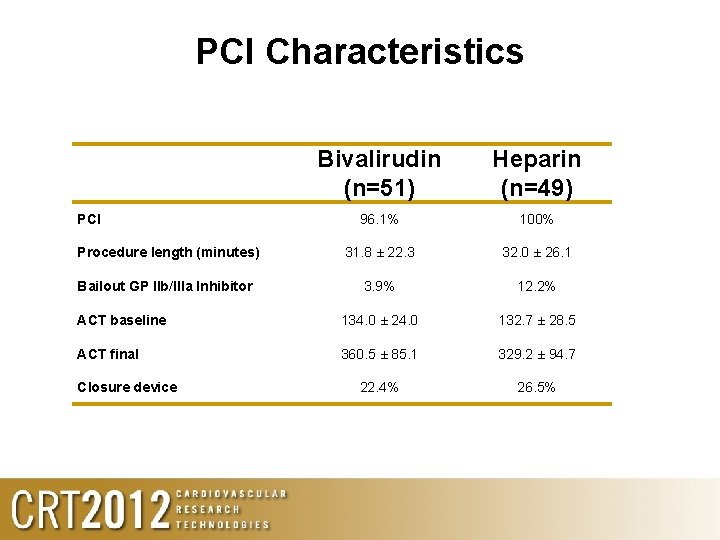
PCI Characteristics Bivalirudin (n=51) Heparin (n=49) 96. 1% 100% Procedure length (minutes) 31. 8 ± 22. 3 32. 0 ± 26. 1 Bailout GP IIb/IIIa Inhibitor 3. 9% 12. 2% ACT baseline 134. 0 ± 24. 0 132. 7 ± 28. 5 ACT final 360. 5 ± 85. 1 329. 2 ± 94. 7 22. 4% 26. 5% PCI Closure device
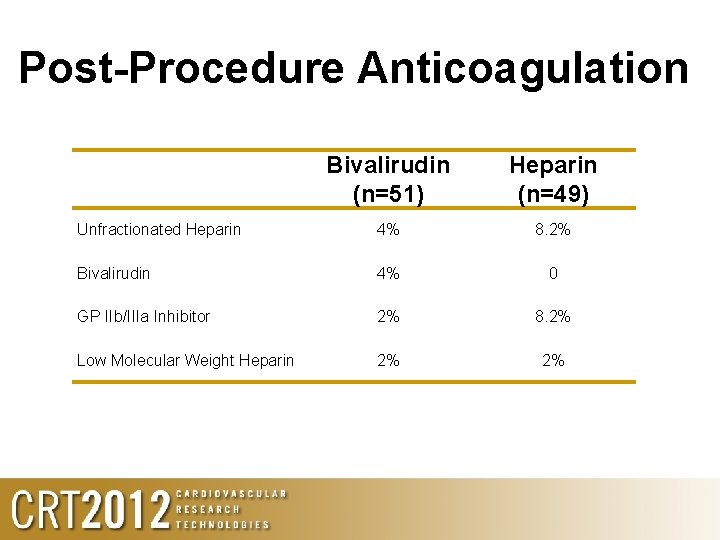
Post-Procedure Anticoagulation Bivalirudin (n=51) Heparin (n=49) Unfractionated Heparin 4% 8. 2% Bivalirudin 4% 0 GP IIb/IIIa Inhibitor 2% 8. 2% Low Molecular Weight Heparin 2% 2%
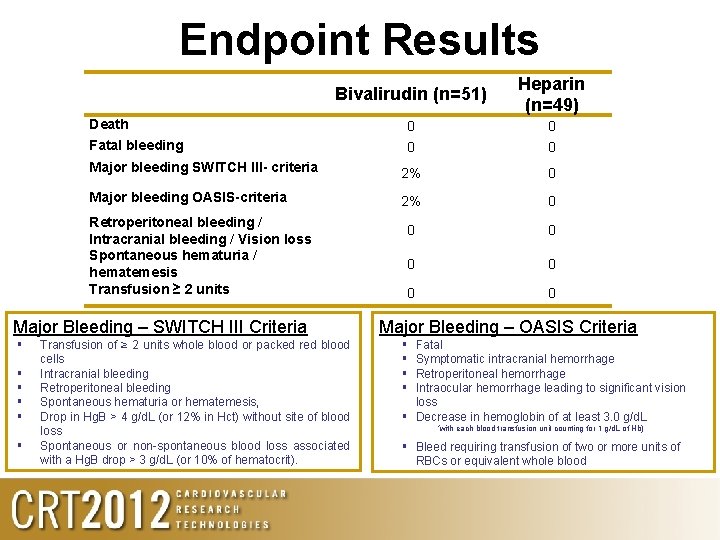
Endpoint Results Bivalirudin (n=51) Heparin (n=49) Death 0 0 Fatal bleeding 0 0 Major bleeding SWITCH III- criteria 2% 0 Major bleeding OASIS-criteria 2% 0 0 0 0 Retroperitoneal bleeding / Intracranial bleeding / Vision loss Spontaneous hematuria / hematemesis Transfusion ≥ 2 units Major Bleeding – SWITCH III Criteria Transfusion of ≥ 2 units whole blood or packed red blood cells Intracranial bleeding Retroperitoneal bleeding Spontaneous hematuria or hematemesis, Drop in Hg. B > 4 g/d. L (or 12% in Hct) without site of blood loss Spontaneous or non-spontaneous blood loss associated with a Hg. B drop > 3 g/d. L (or 10% of hematocrit). Major Bleeding – OASIS Criteria Fatal Symptomatic intracranial hemorrhage Retroperitoneal hemorrhage Intraocular hemorrhage leading to significant vision loss Decrease in hemoglobin of at least 3. 0 g/d. L *with each blood transfusion unit counting for 1 g/d. L of Hb) Bleed requiring transfusion of two or more units of RBCs or equivalent whole blood
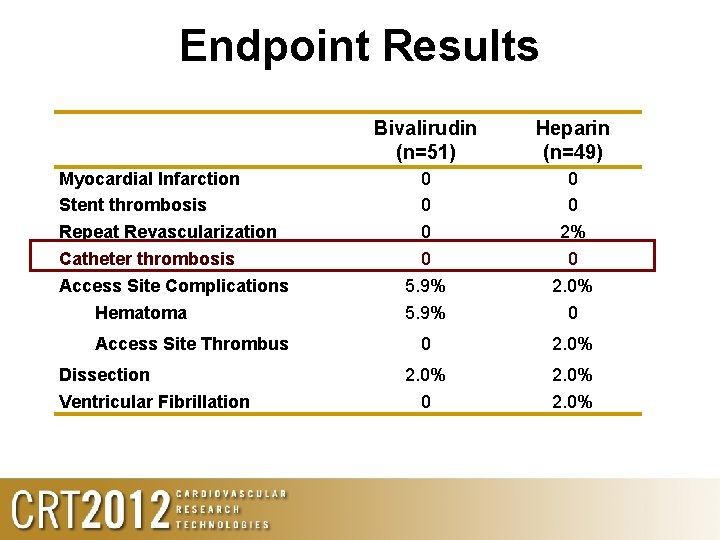
Endpoint Results Myocardial Infarction Stent thrombosis Repeat Revascularization Catheter thrombosis Access Site Complications Hematoma Access Site Thrombus Dissection Ventricular Fibrillation Bivalirudin (n=51) Heparin (n=49) 0 0 5. 9% 0 0 2% 0 2. 0% 0 0 2. 0%

Limitations • This trial included only a small number of patients and is not powered to detect intergroup differences. Nevertheless, the low level of adverse events in the bivalirudin group could still serve as a measure of safety for this treatment approach. • Nearly 2/3 of the patients treated via the radial access which minimized the access bleed

Conclusions • The data from this prospective, multicenter trial suggest that bivalirudin in comparison to standard-dose UFH has a similar safety profile in terms of peri-PCI bleeding and thrombotic events. • The lack of intra catheter thrombus in the Bivalirudin group suggest that it can used safely in ACS patients initially treated with upstream fondaparinux. • Larger randomized trials are warranted to establish the safety and efficacy of treating patients with bivalirudin in this clinical setting
- Slides: 19