Switch from Boosted PI 2 NRTIs to BICTAFFTC

Switch from Boosted PI + 2 NRTI’s to BIC-TAF-FTC with Viral Suppression GS-380 -1878

Switch from Boosted PI to BIC-TAF-FTC GS-380 -1878: Design GS-380 -1878: Study Design • Background: Randomized, phase 3, multicenter, open-label switch study evaluating the efficacy and safety of switching adults with viral suppression taking a boosted PI plus 2 NRTI’s to BIC-TAF-FTC • Inclusion Criteria - Age >18 - HIV RNA <50 copies/m. L for ≥ 6 months - On stable antiretroviral regimen for ≥ 6 months - No history of treatment failure - No prior treatment with an INSTI - e. GFR >50 m. L/min - HBV and HCV allowed - Taking atazanavir or darunavir (each boosted by ritonavir or cobicistat) + TDF-FTC or ABC-3 TC Source: Daar E, et al. Lancet HIV. 2018; 5: e 347 -e 356. Switch Regimen Bictegravir-TAF-FTC (n = 290) Maintain Regimen Boosted PI + 2 NRTI’s (n = 287)
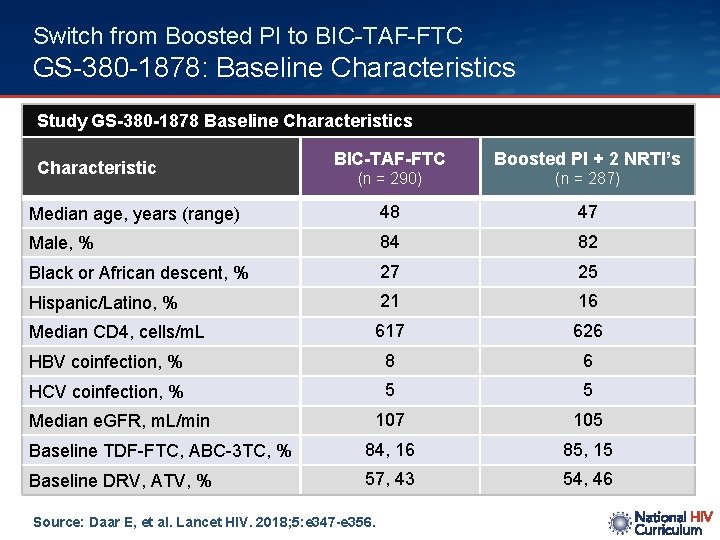
Switch from Boosted PI to BIC-TAF-FTC GS-380 -1878: Baseline Characteristics Study GS-380 -1878 Baseline Characteristics BIC-TAF-FTC Boosted PI + 2 NRTI’s (n = 290) (n = 287) Median age, years (range) 48 47 Male, % 84 82 Black or African descent, % 27 25 Hispanic/Latino, % 21 16 Median CD 4, cells/m. L 617 626 HBV coinfection, % 8 6 HCV coinfection, % 5 5 107 105 Baseline TDF-FTC, ABC-3 TC, % 84, 16 85, 15 Baseline DRV, ATV, % 57, 43 54, 46 Characteristic Median e. GFR, m. L/min Source: Daar E, et al. Lancet HIV. 2018; 5: e 347 -e 356.
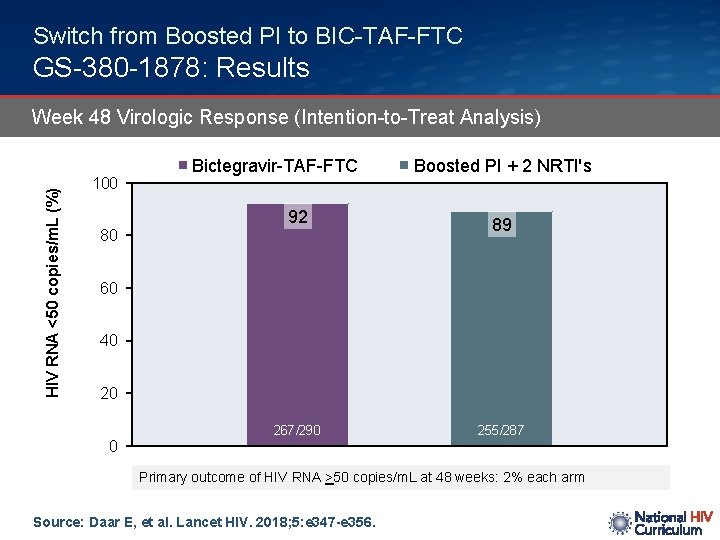
Switch from Boosted PI to BIC-TAF-FTC GS-380 -1878: Results HIV RNA <50 copies/m. L (%) Week 48 Virologic Response (Intention-to-Treat Analysis) 100 Bictegravir-TAF-FTC 92 80 Boosted PI + 2 NRTI's 89 60 40 20 0 267/290 255/287 Primary outcome of HIV RNA >50 copies/m. L at 48 weeks: 2% each arm Source: Daar E, et al. Lancet HIV. 2018; 5: e 347 -e 356.
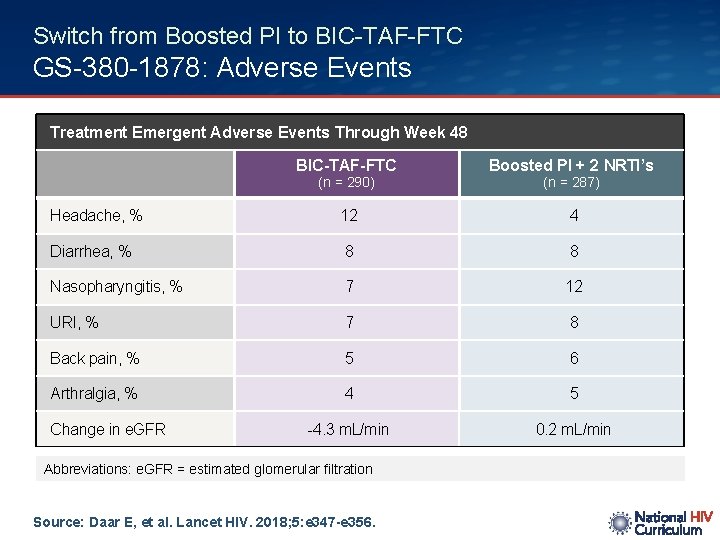
Switch from Boosted PI to BIC-TAF-FTC GS-380 -1878: Adverse Events Treatment Emergent Adverse Events Through Week 48 BIC-TAF-FTC Boosted PI + 2 NRTI’s Headache, % 12 4 Diarrhea, % 8 8 Nasopharyngitis, % 7 12 URI, % 7 8 Back pain, % 5 6 Arthralgia, % 4 5 -4. 3 m. L/min 0. 2 m. L/min (n = 290) Change in e. GFR Abbreviations: e. GFR = estimated glomerular filtration Source: Daar E, et al. Lancet HIV. 2018; 5: e 347 -e 356. (n = 287)
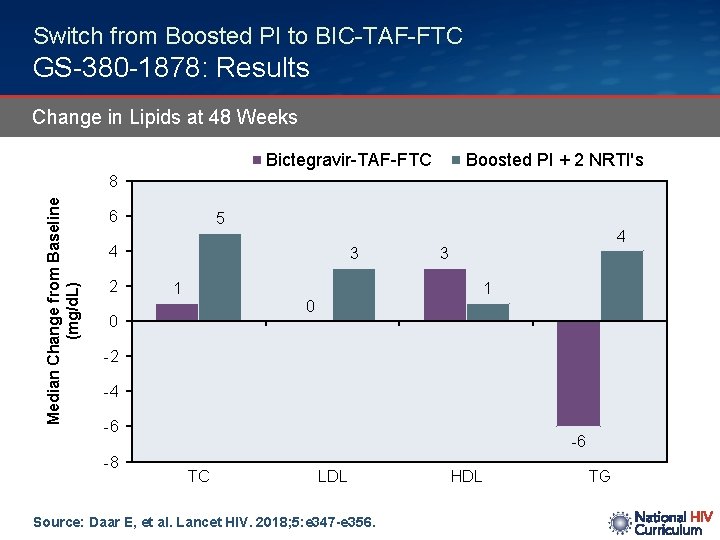
Switch from Boosted PI to BIC-TAF-FTC GS-380 -1878: Results Change in Lipids at 48 Weeks Bictegravir-TAF-FTC Boosted PI + 2 NRTI's Median Change from Baseline (mg/d. L) 8 6 5 4 2 3 1 4 3 1 0 0 -2 -4 -6 -8 -6 TC LDL Source: Daar E, et al. Lancet HIV. 2018; 5: e 347 -e 356. HDL TG

Switch from Boosted PI to BIC-TAF-FTC GS-380 -1878: Conclusions Interpretation: “Fixed-dose bictegravir, emtricitabine, and tenofovir alafenamide might be a safe and efficacious alternative to continued boosted protease inhibitor therapy in adults with HIV-1 infection. ” Source: Daar E, et al. Lancet HIV. 2018; 5: e 347 -e 356.

BIC-TAF-FTC Switch Studies (1844 and 1878) Impact of Archived M 184 V Mutation

Switching to Bictegravir-TAF-FTC with Archived M 184 V Studies 1844 and 1878: Design 1844 & 1878: Analysis of Switch Studies Switch Regimen • Background: Preexisiting resistance data were assessed from 2 phase 3 studies that analyzed switching antiretroviral regimens in adults with suppressed HIV RNA levels (for ≥ 6 months) to bictegravir-tenofovir alafenamide-emtricitabine (BIC-TAF-FTC) versus continuing regimen • Analysis for Resistance Criteria - Historical genotypes - Retrospective proviral archived DNA genotype - Resistance data obtained for 95% (543/570) of participants who switched to BIC-TAF-FTC Bictegravir-TAF-FTC (n=570) Maintain Regimen 1844: Dolutegravir + ABC-3 TC (n=285) 1878: Boosted PI* + 2 NRTIs (n=281) *56% boosted Darunavir Source: Andreatta K, et al. J Antimicrob Chemo. . 74: 3555 -64; 2019

Switching to Bictegravir-TAF-FTC with Archived Resistance Studies 1844 and 1878: Results Pre-existing resistance substitutions and virologic outcomes at 48 weeks Percentage with HIV RNA <50 copies/m. L by baseline resistance mutation BIC-FTC-TAF (n = 543) Common NRTI Substitutions M 184 V/I 96. 3% (52/54) K 65 R/N 100. 0% (7/7) Any TAM 95. 8% (46/48) 1 or 2 TAM’s 94. 3% (33/35) 3 or more TAM’s 100. 0% (13/13) Primary INSTI Substitutions T 97 A 100% (9/9) E 92 G or Y 143 H or S 147 G or Q 148 H 100% (4/4) Source: Andreatta K, et al. J Antimicrob Chemo. . 74: 3555 -64; 2019

Switching to Bictegravir-TAF-FTC with Archived M 184 V Studies 1844 and 1878: Key Points • Baseline M 184 V/I in 10% of switch group (BIC-TAF-FTC) • 96% (52/54) with archived M 184 V had HIV RNA <50 copies/m. L for up to 48 weeks on BIC-TAF-FTC Source: Andreatta K, et al. J Antimicrob Chemo. . 74: 3555 -64; 2019

Switching to Bictegravir-TAF-FTC with Archived M 184 V Studies 1878 and 1844: Conclusions Interpretation: “Pre-existing resistance substitutions, notably M 184 V/I, were unexpectedly common among suppressed participants who switched to BIC/FTC/TAF. High rates of virological suppression were maintained in the overall study population and in those with pre-existing resistance, including M 184 V/I, for up to 48 weeks of BIC/FTC/TAF treatment with no resistance development. These results indicate that BIC/FTC/TAF is an effective treatment option for suppressed patients, including those with evidence of archived NRTI resistance. ” Source: Andreatta K, et al. J Antimicrob Chemo. 2019; pii: dkz 347. [Epub ahead of print]

Acknowledgment The National HIV Curriculum is an AIDS Education and Training Center (AETC) Program supported by the Health Resources and Services Administration (HRSA) of the U. S. Department of Health and Human Services (HHS) as part of an award totaling $800, 000 with 0% financed with non-governmental sources. This project is led by the University of Washington’s Infectious Diseases Education and Assessment (IDEA) Program. The content in this presentation are those of the author(s) and do not necessarily represent the official views of, nor an endorsement, by HRSA, HHS, or the U. S. Government.
- Slides: 13