Switch from AbacavirLamivudine to Tenofovir DFEmtricitabine SWIFT Switch

Switch from Abacavir-Lamivudine to Tenofovir DF-Emtricitabine SWIFT
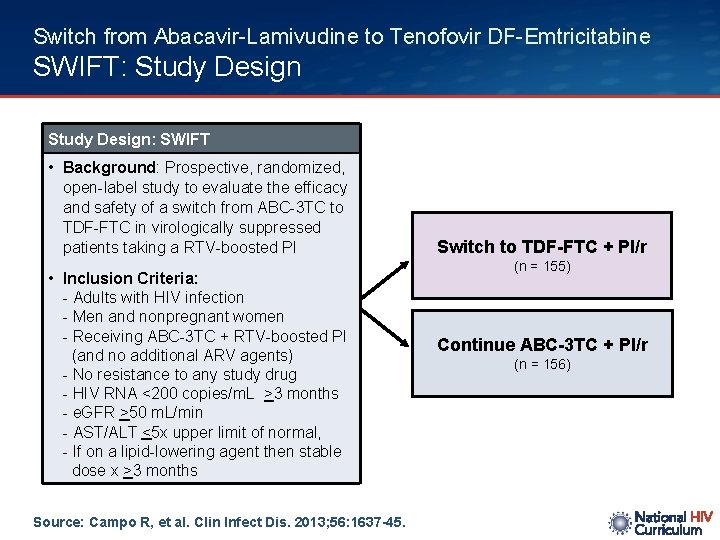
Switch from Abacavir-Lamivudine to Tenofovir DF-Emtricitabine SWIFT: Study Design: SWIFT • Background: Prospective, randomized, open-label study to evaluate the efficacy and safety of a switch from ABC-3 TC to TDF-FTC in virologically suppressed patients taking a RTV-boosted PI • Inclusion Criteria: - Adults with HIV infection - Men and nonpregnant women - Receiving ABC-3 TC + RTV-boosted PI (and no additional ARV agents) - No resistance to any study drug - HIV RNA <200 copies/m. L >3 months - e. GFR >50 m. L/min - AST/ALT <5 x upper limit of normal, - If on a lipid-lowering agent then stable dose x >3 months Source: Campo R, et al. Clin Infect Dis. 2013; 56: 1637 -45. Switch to TDF-FTC + PI/r (n = 155) Continue ABC-3 TC + PI/r (n = 156)
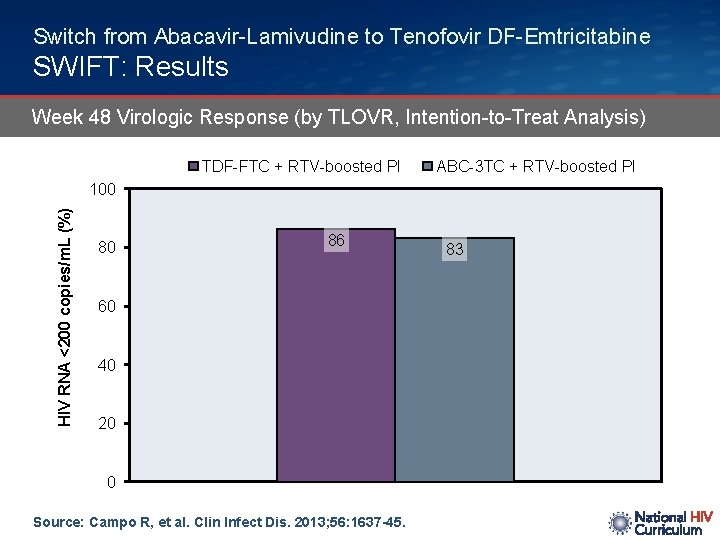
Switch from Abacavir-Lamivudine to Tenofovir DF-Emtricitabine SWIFT: Results Week 48 Virologic Response (by TLOVR, Intention-to-Treat Analysis) TDF-FTC + RTV-boosted PI ABC-3 TC + RTV-boosted PI HIV RNA <200 copies/m. L (%) 100 80 86 60 40 20 0 Source: Campo R, et al. Clin Infect Dis. 2013; 56: 1637 -45. 83
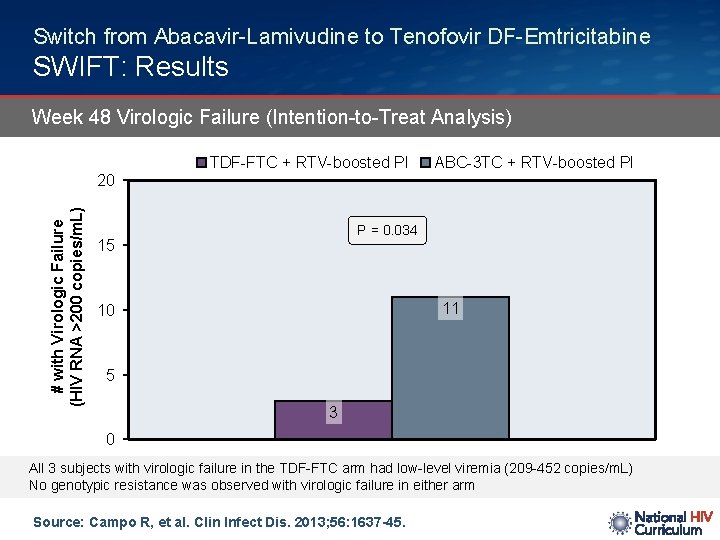
Switch from Abacavir-Lamivudine to Tenofovir DF-Emtricitabine SWIFT: Results Week 48 Virologic Failure (Intention-to-Treat Analysis) # with Virologic Failure (HIV RNA >200 copies/m. L) 20 TDF-FTC + RTV-boosted PI ABC-3 TC + RTV-boosted PI P = 0. 034 15 11 10 5 3 0 All 3 subjects with virologic failure in the TDF-FTC arm had low-level viremia (209 -452 copies/m. L) No genotypic resistance was observed with virologic failure in either arm Source: Campo R, et al. Clin Infect Dis. 2013; 56: 1637 -45.
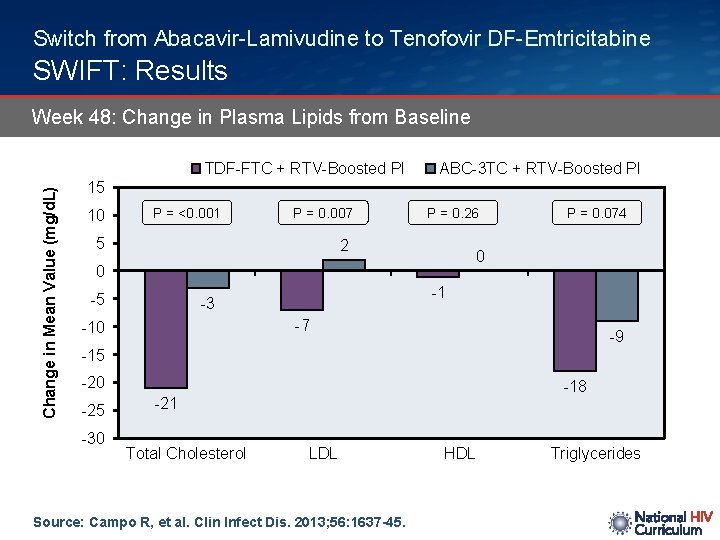
Switch from Abacavir-Lamivudine to Tenofovir DF-Emtricitabine SWIFT: Results Week 48: Change in Plasma Lipids from Baseline Change in Mean Value (mg/d. L) TDF-FTC + RTV-Boosted PI ABC-3 TC + RTV-Boosted PI 15 10 P = <0. 001 P = 0. 007 5 P = 0. 26 2 0 0 -5 P = 0. 074 -1 -3 -7 -10 -9 -15 -20 -25 -30 -18 -21 Total Cholesterol LDL Source: Campo R, et al. Clin Infect Dis. 2013; 56: 1637 -45. HDL Triglycerides
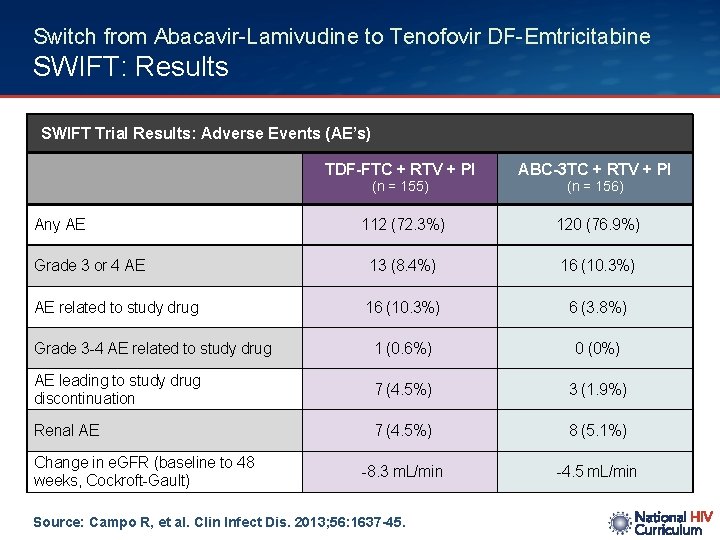
Switch from Abacavir-Lamivudine to Tenofovir DF-Emtricitabine SWIFT: Results SWIFT Trial Results: Adverse Events (AE’s) TDF-FTC + RTV + PI ABC-3 TC + RTV + PI 112 (72. 3%) 120 (76. 9%) Grade 3 or 4 AE 13 (8. 4%) 16 (10. 3%) AE related to study drug 16 (10. 3%) 6 (3. 8%) Grade 3 -4 AE related to study drug 1 (0. 6%) 0 (0%) AE leading to study drug discontinuation 7 (4. 5%) 3 (1. 9%) Renal AE 7 (4. 5%) 8 (5. 1%) -8. 3 m. L/min -4. 5 m. L/min (n = 155) Any AE Change in e. GFR (baseline to 48 weeks, Cockroft-Gault) Source: Campo R, et al. Clin Infect Dis. 2013; 56: 1637 -45. (n = 156)

Switch from Abacavir-Lamivudine to Tenofovir DF-Emtricitabine SWIFT: Results Conclusion: “Switching to FTC/TDF from 3 TC/ABC maintained virologic suppression, had fewer VFs, improved lipid parameters and Framingham scores but decreased e. GFR. ” Source: Campo R et al. Clin Infect Dis. 2013; 56: 1637 -45.

Acknowledgment The National HIV Curriculum is an AIDS Education and Training Center (AETC) Program resource funded by the United States Health Resources and Services Administration. The project is led by the University of Washington and the AETC National Coordinating Resource Center. The content in this slide set does not represent the official views of the U. S. Department of Health and Human Services, Health Resources & Services Administration.
- Slides: 8