Switch ABC3 TC to TDFFTC SWIFT Study SWIFT

Switch ABC/3 TC to TDF/FTC § SWIFT Study
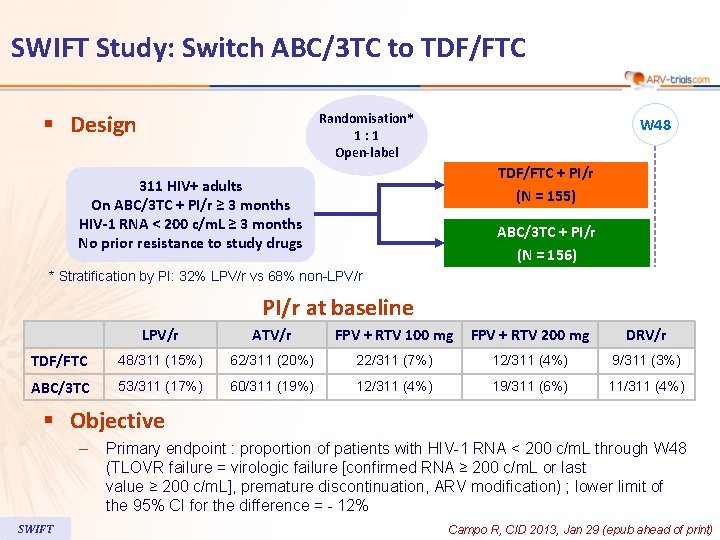
SWIFT Study: Switch ABC/3 TC to TDF/FTC § Design Randomisation* 1 : 1 Open-label W 48 TDF/FTC + PI/r (N = 155) 311 HIV+ adults On ABC/3 TC + PI/r ≥ 3 months HIV-1 RNA < 200 c/m. L ≥ 3 months No prior resistance to study drugs ABC/3 TC + PI/r (N = 156) * Stratification by PI: 32% LPV/r vs 68% non-LPV/r PI/r at baseline LPV/r ATV/r FPV + RTV 100 mg FPV + RTV 200 mg DRV/r TDF/FTC 48/311 (15%) 62/311 (20%) 22/311 (7%) 12/311 (4%) 9/311 (3%) ABC/3 TC 53/311 (17%) 60/311 (19%) 12/311 (4%) 19/311 (6%) 11/311 (4%) § Objective – SWIFT Primary endpoint : proportion of patients with HIV-1 RNA < 200 c/m. L through W 48 (TLOVR failure = virologic failure [confirmed RNA ≥ 200 c/m. L or last value ≥ 200 c/m. L], premature discontinuation, ARV modification) ; lower limit of the 95% CI for the difference = - 12% Campo R, CID 2013, Jan 29 (epub ahead of print)
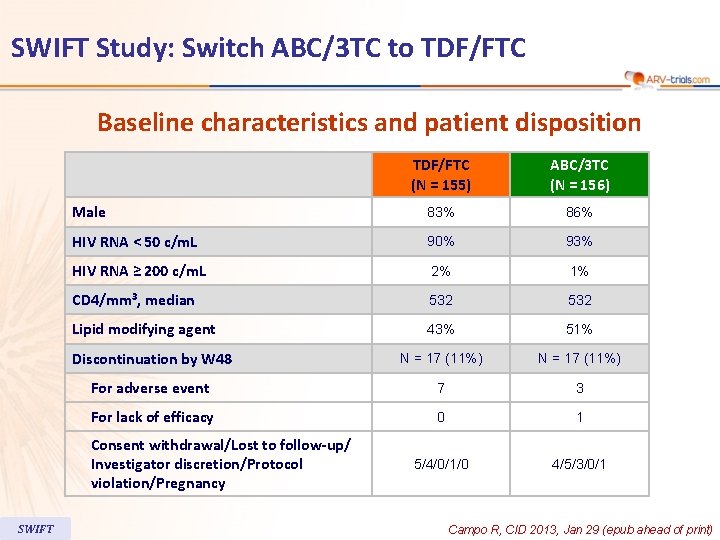
SWIFT Study: Switch ABC/3 TC to TDF/FTC Baseline characteristics and patient disposition TDF/FTC (N = 155) ABC/3 TC (N = 156) Male 83% 86% HIV RNA < 50 c/m. L 90% 93% HIV RNA ≥ 200 c/m. L 2% 1% CD 4/mm 3, median 532 Lipid modifying agent 43% 51% Discontinuation by W 48 N = 17 (11%) For adverse event 7 3 For lack of efficacy 0 1 5/4/0/1/0 4/5/3/0/1 Consent withdrawal/Lost to follow-up/ Investigator discretion/Protocol violation/Pregnancy SWIFT Campo R, CID 2013, Jan 29 (epub ahead of print)
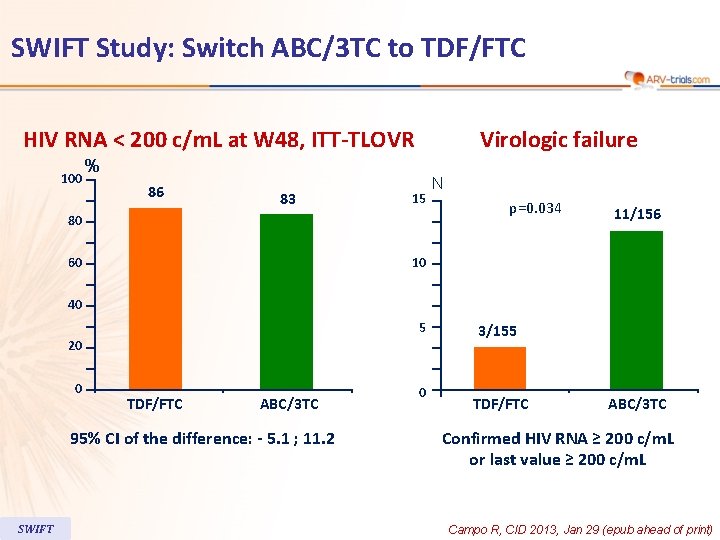
SWIFT Study: Switch ABC/3 TC to TDF/FTC HIV RNA < 200 c/m. L at W 48, ITT-TLOVR 100 Virologic failure % 86 83 15 80 60 N p=0. 034 11/156 10 40 5 20 0 TDF/FTC ABC/3 TC 95% CI of the difference: - 5. 1 ; 11. 2 SWIFT 0 3/155 TDF/FTC ABC/3 TC Confirmed HIV RNA ≥ 200 c/m. L or last value ≥ 200 c/m. L Campo R, CID 2013, Jan 29 (epub ahead of print)
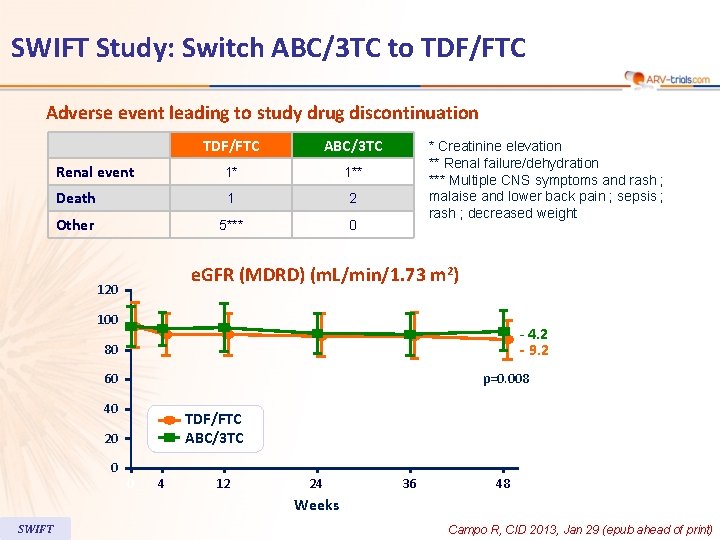
SWIFT Study: Switch ABC/3 TC to TDF/FTC Adverse event leading to study drug discontinuation TDF/FTC ABC/3 TC Renal event 1* 1** Death 1 2 Other 5*** 0 * Creatinine elevation ** Renal failure/dehydration *** Multiple CNS symptoms and rash ; malaise and lower back pain ; sepsis ; rash ; decreased weight e. GFR (MDRD) (m. L/min/1. 73 m 2) 120 100 - 4. 2 - 9. 2 80 60 p=0. 008 40 TDF/FTC ABC/3 TC 20 0 0 4 12 24 36 48 Weeks SWIFT Campo R, CID 2013, Jan 29 (epub ahead of print)
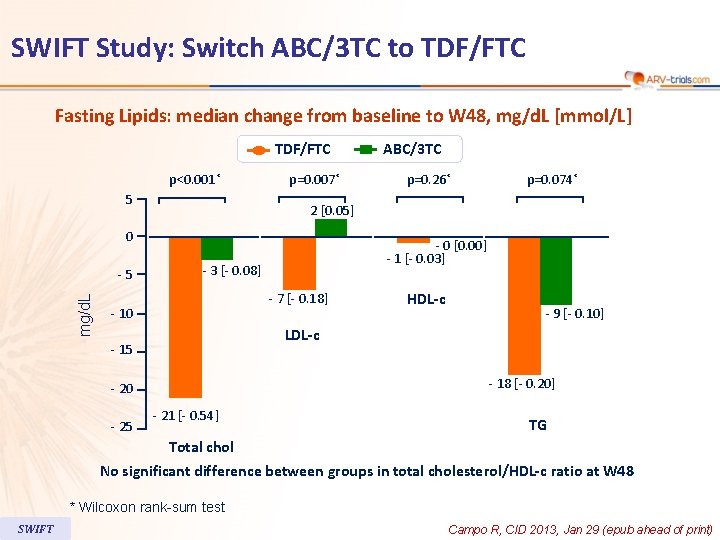
SWIFT Study: Switch ABC/3 TC to TDF/FTC Fasting Lipids: median change from baseline to W 48, mg/d. L [mmol/L] TDF/FTC p<0. 001* 5 p=0. 007* mg/d. L p=0. 074* - 0 [0. 00] - 1 [- 0. 03] - 3 [- 0. 08] - 7 [- 0. 18] - 10 HDL-c - 9 [- 0. 10] LDL-c - 15 - 18 [- 0. 20] - 20 - 25 p=0. 26* 2 [0. 05] 0 -5 ABC/3 TC - 21 [- 0. 54] TG Total chol No significant difference between groups in total cholesterol/HDL-c ratio at W 48 * Wilcoxon rank-sum test SWIFT Campo R, CID 2013, Jan 29 (epub ahead of print)
- Slides: 6