SUSTAINABLE MATERIALS BASED ON ALIPHATIC POLYESTERS TEACHING OLD

SUSTAINABLE MATERIALS BASED ON ALIPHATIC POLYESTERS : TEACHING OLD CHEMISTRY SOME NEW TRICKS Indian Academy of Science, Bangalore Mid-Year Meeting July 5, 2013 DR. S. SIVARAM A 201, Polymers & Advanced Materials Laboratory, National Chemical Laboratory, Pune-411 008, INDIA Tel : 0091 20 2589 2614 Fax : 0091 20 2589 2615 Email : s. sivaram@ncl. res. in

SUSTAINABLE DEVELOPMENT (CHEMISTRY) “ Development (chemistry or chemical industry) that meet the needs of the present without compromising the ability of future generations to meet their own needs” In other words, each generation must bequeath to its successor at least as large a productive base it inherited from its predecessor Brundtland Report UN World Commission on Environment and Development, 1987 www. un. org/documents/ga/res/42/ares 42 -187. htm We do not inherit the earth from our ancestors; we borrow it from our children. Native American Proverb

ORGANIC CHEMICALS AND MATERIALS REDUCE • Exclusive dependence on fossil fuel based resources • Generation of wastes that need disposal Sustainability is the key concern of science, technology, industry and society today RECYCLE REUSE Can the chemical and materials needs of humankind be based on the concept of sustainability of both resources and environment? RIL, Mumbai 281205

Poly( ethylene terephthalate) Over 30 billion liters of bottled water is consumed annually Every second we throw away about 1500 bottles What is the solution ?
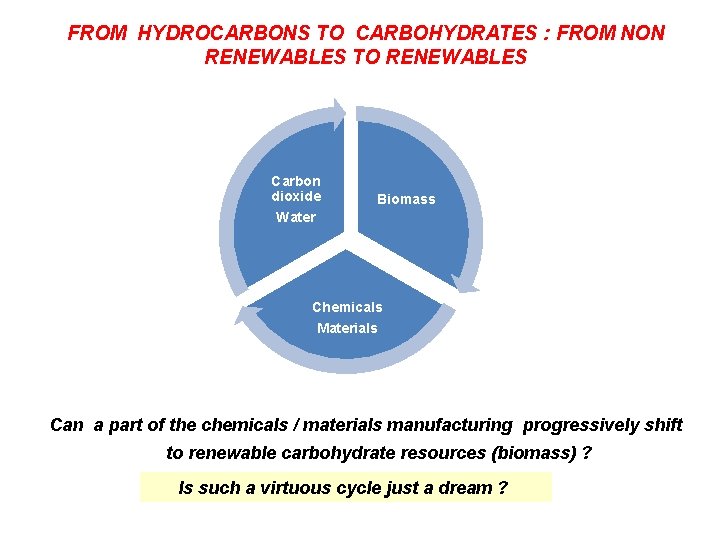
FROM HYDROCARBONS TO CARBOHYDRATES : FROM NON RENEWABLES TO RENEWABLES Carbon dioxide Water Biomass Chemicals Materials Can a part of the chemicals / materials manufacturing progressively shift to renewable carbohydrate resources (biomass) ? Is such a virtuous cycle just a dream ? RIL, Mumbai 281205

SUSTAINABLE POLYMERS FROM BIO-DERIVED AND BIO-RENEWABLE RESOURCES Objectives Expand the chemistry toolbox with new methods and techniques for next generation products Materials Platform Aliphatic Polyesters
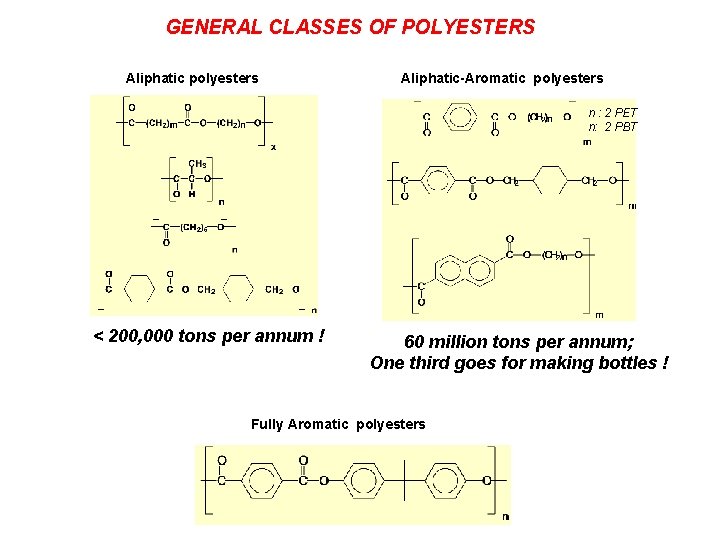
GENERAL CLASSES OF POLYESTERS Aliphatic polyesters Aliphatic-Aromatic polyesters n : 2 PET n: 2 PBT < 200, 000 tons per annum ! 60 million tons per annum; One third goes for making bottles ! Fully Aromatic polyesters

POLY (LACTIC ACID )S : AN ALIPHATIC POLYESTER FROM THE MOST SIMPLE AB MONOMER Insoluble in water, moisture and grease resistant Biodegradable and compostable Clarity and glossiness similar to PET Requires 20 to 50% less fossil fuels to produce than PET Physical properties similar to PET
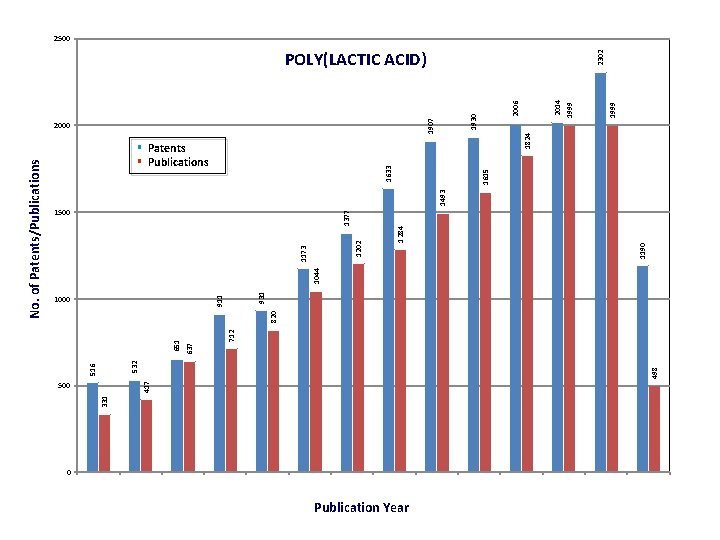
331 500 417 498 532 516 712 651 637 820 1000 931 911 1044 1493 1615 1633 2000 0 Publication Year 1824 1999 2014 2006 1930 1907 2302 POLY(LACTIC ACID) 1190 1500 1377 § Patents § Publications 1284 1202 1173 No. of Patents/Publications 2500
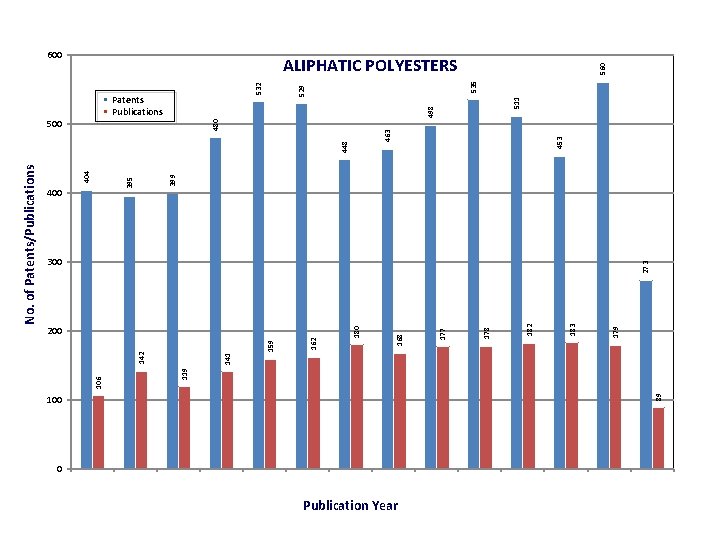
200 100 0 Publication Year 300 273 453 463 448 511 498 535 529 532 560 ALIPHATIC POLYESTERS 179 183 182 178 177 168 180 480 500 162 159 399 § Patents § Publications 89 141 142 404 395 400 119 106 No. of Patents/Publications 600

ALIPHATIC POLYESTERS : PIONEERING CHEMISTRY OF WALLACE HUME CAROTHERS STUDIES ON POLYMERIZATION AND RING FORMATION Twenty eight papers from 1929 to 1935 Carothers addressed one important question: (a) If two bifunctional molecules, e. g. , one dibasic acid and one glycol or diamide, react, two possibilities occur. The reaction can result (1) in a chain polymer of lower or higher molecular weight, which still bears either hydroxyl or carboxyl terminal groups or (2) in a smaller or larger ring, which does not contain the reactive group. 1896 -1937 54 papers 400 pages Enough for one lifetime ! Under what conditions does either of these two possibilities take place and what is the molecular weight of the resulting compound? Published 1940
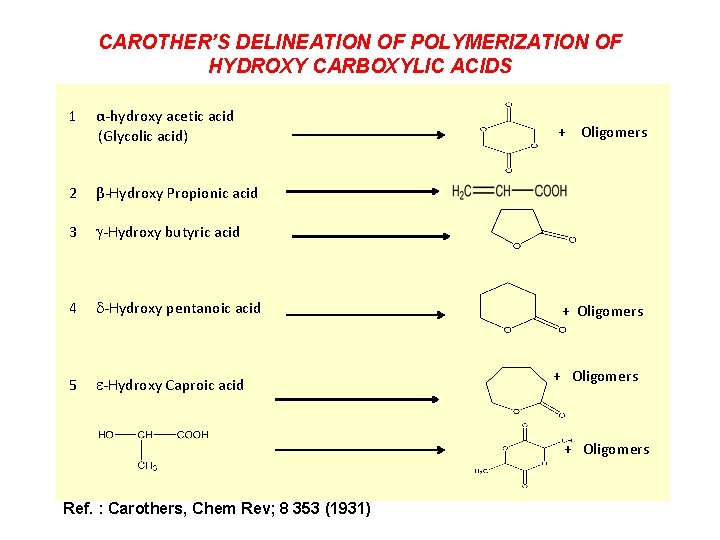
CAROTHER’S DELINEATION OF POLYMERIZATION OF HYDROXY CARBOXYLIC ACIDS 1 α-hydroxy acetic acid (Glycolic acid) 2 β-Hydroxy Propionic acid 3 -Hydroxy butyric acid 4 -Hydroxy pentanoic acid 5 -Hydroxy Caproic acid + Oligomers Ref. : Carothers, Chem Rev; 8 353 (1931)
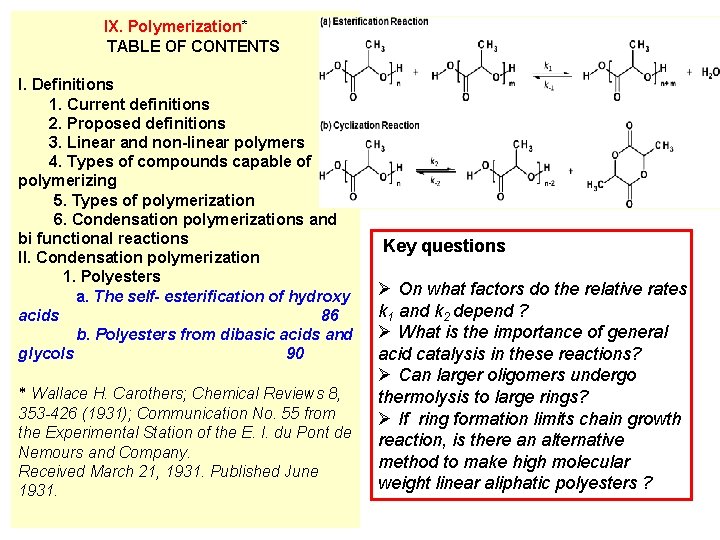
IX. Polymerization* TABLE OF CONTENTS I. Definitions 1. Current definitions 2. Proposed definitions 3. Linear and non-linear polymers 4. Types of compounds capable of polymerizing 5. Types of polymerization 6. Condensation polymerizations and bi functional reactions II. Condensation polymerization 1. Polyesters a. The self- esterification of hydroxy acids 86 b. Polyesters from dibasic acids and glycols 90 * Wallace H. Carothers; Chemical Reviews 8, 353 -426 (1931); Communication No. 55 from the Experimental Station of the E. I. du Pont de Nemours and Company. Received March 21, 1931. Published June 1931. Key questions Ø On what factors do the relative rates k 1 and k 2 depend ? Ø What is the importance of general acid catalysis in these reactions? Ø Can larger oligomers undergo thermolysis to large rings? Ø If ring formation limits chain growth reaction, is there an alternative method to make high molecular weight linear aliphatic polyesters ?
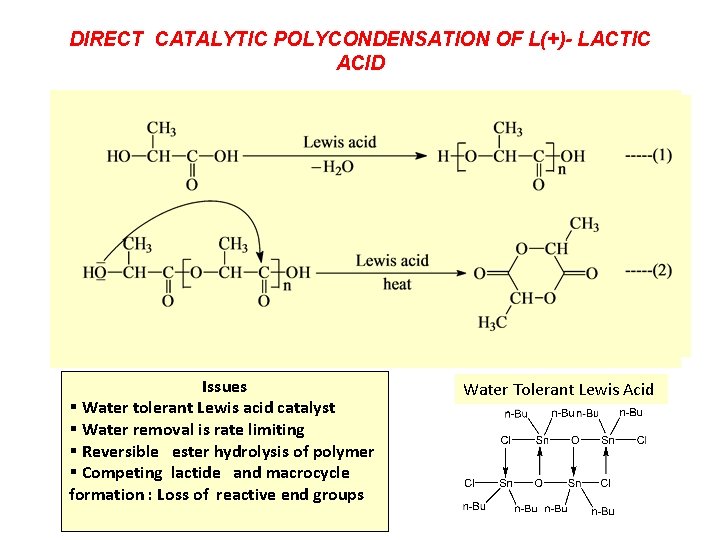
DIRECT CATALYTIC POLYCONDENSATION OF L(+)- LACTIC ACID Issues § Water tolerant Lewis acid catalyst § Water removal is rate limiting § Reversible ester hydrolysis of polymer § Competing lactide and macrocycle formation : Loss of reactive end groups Water Tolerant Lewis Acid
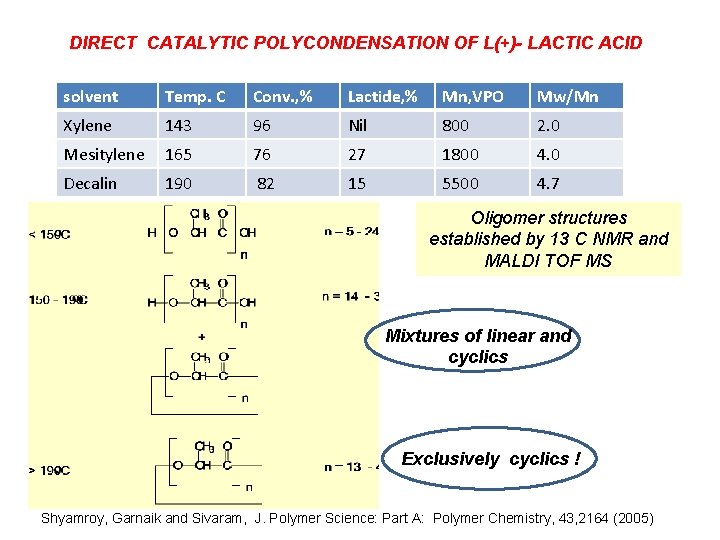
DIRECT CATALYTIC POLYCONDENSATION OF L(+)- LACTIC ACID solvent Temp. C Conv. , % Lactide, % Mn, VPO Mw/Mn Xylene 143 96 Nil 800 2. 0 Mesitylene 165 76 27 1800 4. 0 Decalin 190 82 15 5500 4. 7 Oligomer structures established by 13 C NMR and MALDI TOF MS Mixtures of linear and cyclics Exclusively cyclics ! Shyamroy, Garnaik and Sivaram, J. Polymer Science: Part A: Polymer Chemistry, 43, 2164 (2005)
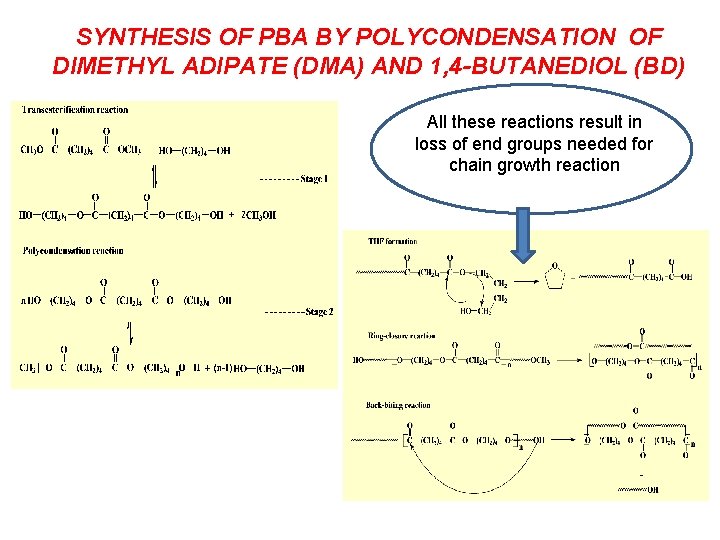
SYNTHESIS OF PBA BY POLYCONDENSATION OF DIMETHYL ADIPATE (DMA) AND 1, 4 -BUTANEDIOL (BD) All these reactions result in loss of end groups needed for chain growth reaction
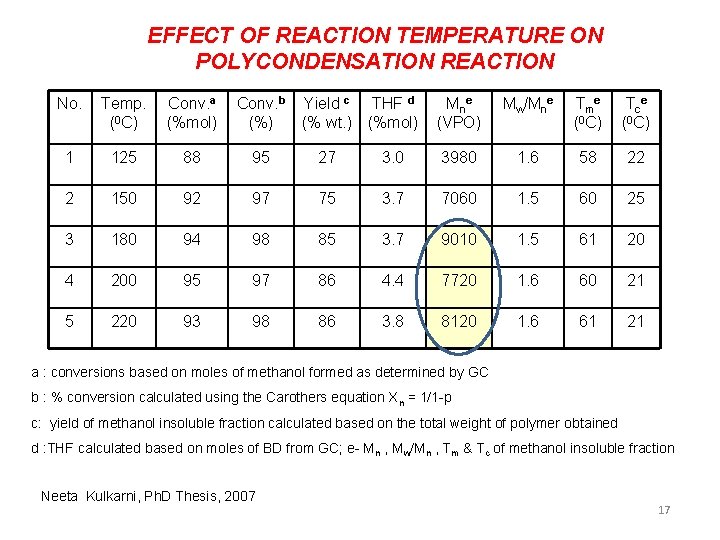
EFFECT OF REACTION TEMPERATURE ON POLYCONDENSATION REACTION No. Temp. (0 C) Conv. a (%mol) Conv. b (%) Yield c THF d (% wt. ) (%mol) Mn e (VPO) Mw/Mne Tme (0 C) Tce (0 C) 1 125 88 95 27 3. 0 3980 1. 6 58 22 2 150 92 97 75 3. 7 7060 1. 5 60 25 3 180 94 98 85 3. 7 9010 1. 5 61 20 4 200 95 97 86 4. 4 7720 1. 6 60 21 5 220 93 98 86 3. 8 8120 1. 6 61 21 a : conversions based on moles of methanol formed as determined by GC b : % conversion calculated using the Carothers equation Xn = 1/1 -p c: yield of methanol insoluble fraction calculated based on the total weight of polymer obtained d : THF calculated based on moles of BD from GC; e- Mn , Mw/Mn , Tm & Tc of methanol insoluble fraction Neeta Kulkarni, Ph. D Thesis, 2007 17
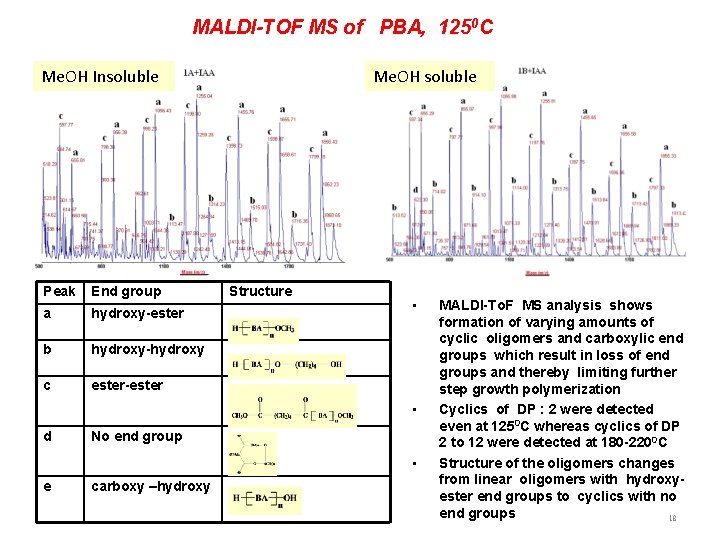
MALDI-TOF MS of PBA, 1250 C Me. OH Insoluble Peak End group a hydroxy-ester b hydroxy-hydroxy c ester-ester Me. OH soluble Structure • • d No end group • e carboxy –hydroxy MALDI-To. F MS analysis shows formation of varying amounts of cyclic oligomers and carboxylic end groups which result in loss of end groups and thereby limiting further step growth polymerization Cyclics of DP : 2 were detected even at 1250 C whereas cyclics of DP 2 to 12 were detected at 180 -2200 C Structure of the oligomers changes from linear oligomers with hydroxyester end groups to cyclics with no end groups 18
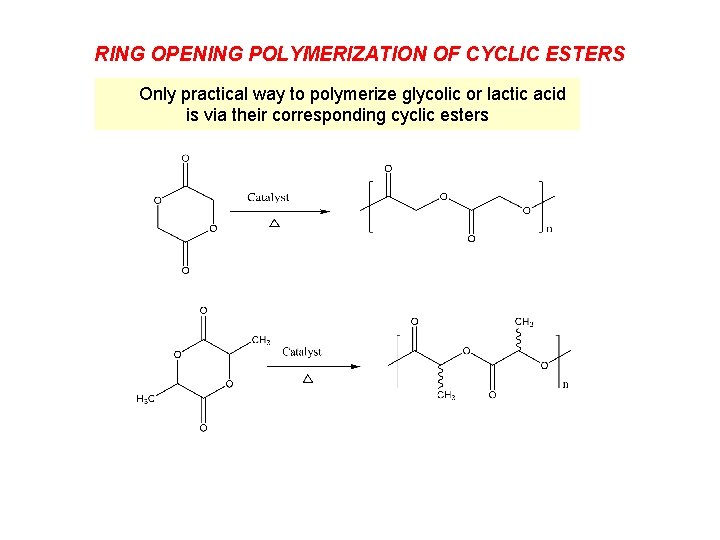
RING OPENING POLYMERIZATION OF CYCLIC ESTERS Only practical way to polymerize glycolic or lactic acid is via their corresponding cyclic esters
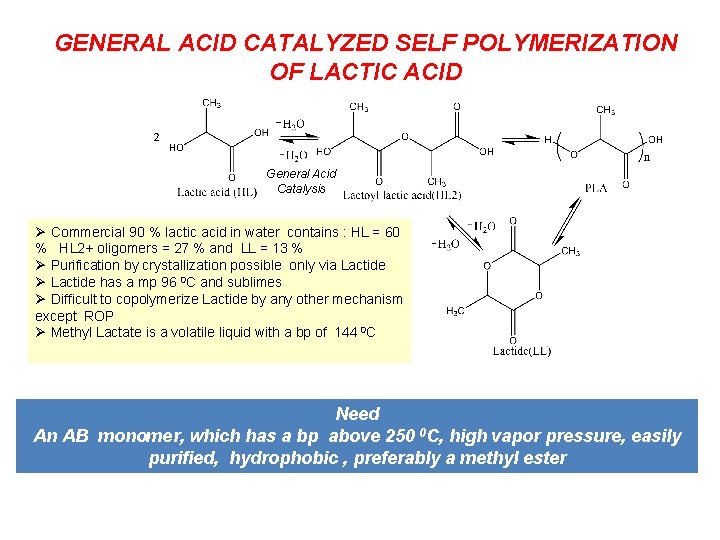
GENERAL ACID CATALYZED SELF POLYMERIZATION OF LACTIC ACID General Acid Catalysis Ø Commercial 90 % lactic acid in water contains : HL = 60 % HL 2+ oligomers = 27 % and LL = 13 % Ø Purification by crystallization possible only via Lactide Ø Lactide has a mp 96 0 C and sublimes Ø Difficult to copolymerize Lactide by any other mechanism except ROP Ø Methyl Lactate is a volatile liquid with a bp of 144 0 C Need An AB monomer, which has a bp above 250 0 C, high vapor pressure, easily purified, hydrophobic , preferably a methyl ester
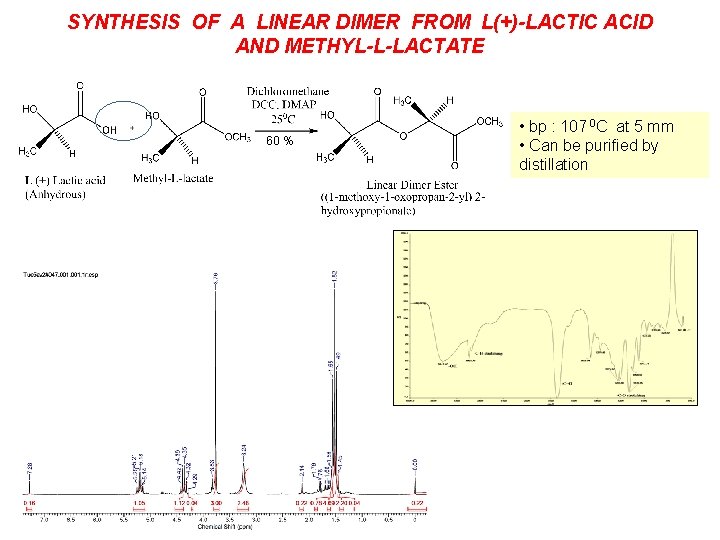
SYNTHESIS OF A LINEAR DIMER FROM L(+)-LACTIC ACID AND METHYL-L-LACTATE 60 % • bp : 107 0 C at 5 mm • Can be purified by distillation
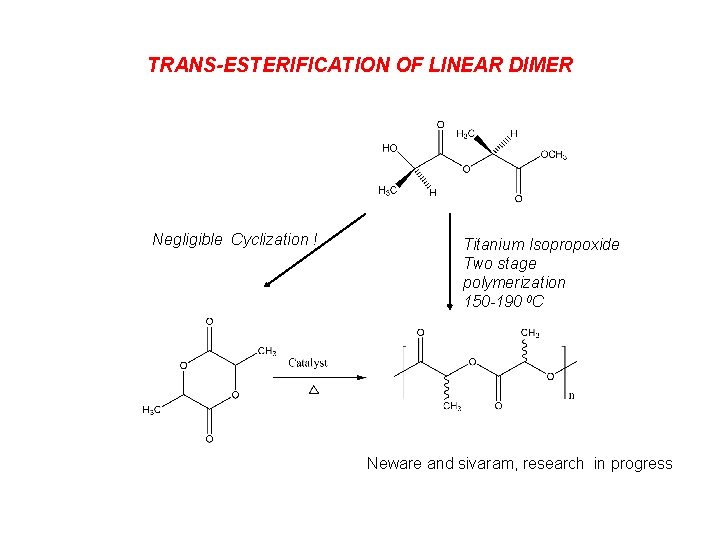
TRANS-ESTERIFICATION OF LINEAR DIMER Negligible Cyclization ! Titanium Isopropoxide Two stage polymerization 150 -190 0 C Neware and sivaram, research in progress

SYNTHESIS OF HIGH MOLECULAR WEIGHT LINEAR ALIPHATIC POLYESTERS Ø Coupling of preformed oligomer chains with suitably disposed end groups Ø Converting a reversible chain growth reaction into an irreversible reaction Ø Use of conformationally constrained cyclo-aliphatic diacid or diol monomers
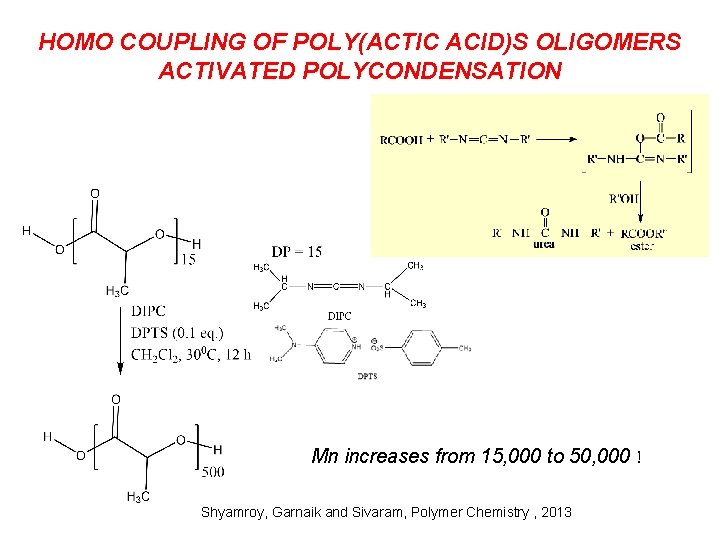
HOMO COUPLING OF POLY(ACTIC ACID)S OLIGOMERS ACTIVATED POLYCONDENSATION Mn increases from 15, 000 to 50, 000 ! Shyamroy, Garnaik and Sivaram, Polymer Chemistry , 2013
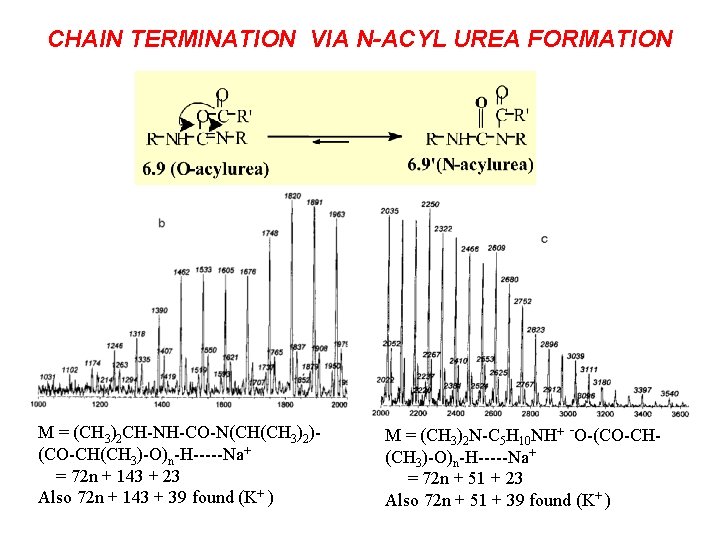
CHAIN TERMINATION VIA N-ACYL UREA FORMATION M = (CH 3)2 CH-NH-CO-N(CH(CH 3)2)(CO-CH(CH 3)-O)n-H-----Na+ = 72 n + 143 + 23 Also 72 n + 143 + 39 found (K+ ) M = (CH 3)2 N-C 5 H 10 NH+ -O-(CO-CH(CH 3)-O)n-H-----Na+ = 72 n + 51 + 23 Also 72 n + 51 + 39 found (K+ )
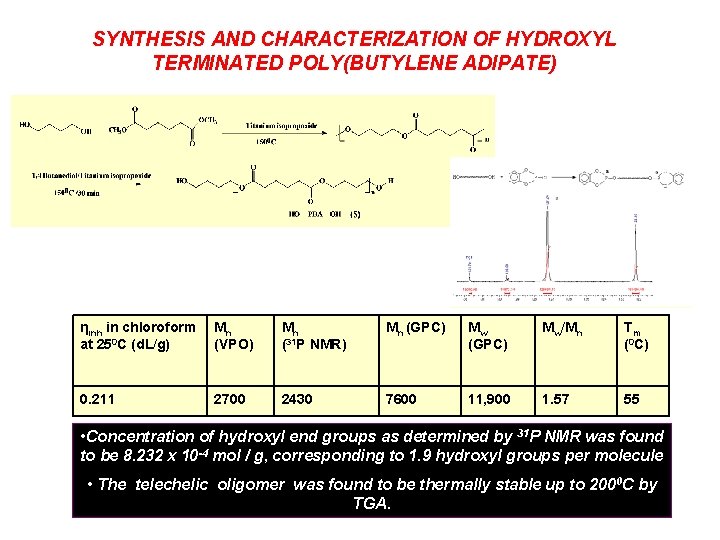
SYNTHESIS AND CHARACTERIZATION OF HYDROXYL TERMINATED POLY(BUTYLENE ADIPATE) ηinh in chloroform at 250 C (d. L/g) Mn (VPO) Mn (31 P NMR) Mn (GPC) Mw/Mn Tm (0 C) 0. 211 2700 2430 7600 11, 900 1. 57 55 • Concentration of hydroxyl end groups as determined by 31 P NMR was found to be 8. 232 x 10 -4 mol / g, corresponding to 1. 9 hydroxyl groups per molecule • The telechelic oligomer was found to be thermally stable up to 2000 C by TGA.
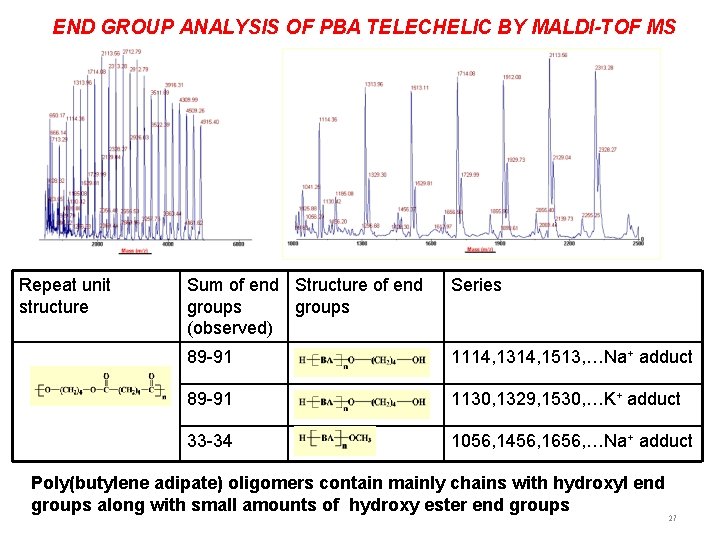
END GROUP ANALYSIS OF PBA TELECHELIC BY MALDI-TOF MS Repeat unit structure Sum of end Structure of end groups (observed) Series 89 -91 1114, 1314, 1513, …Na+ adduct 89 -91 1130, 1329, 1530, …K+ adduct 33 -34 1056, 1456, 1656, …Na+ adduct Poly(butylene adipate) oligomers contain mainly chains with hydroxyl end groups along with small amounts of hydroxy ester end groups 27
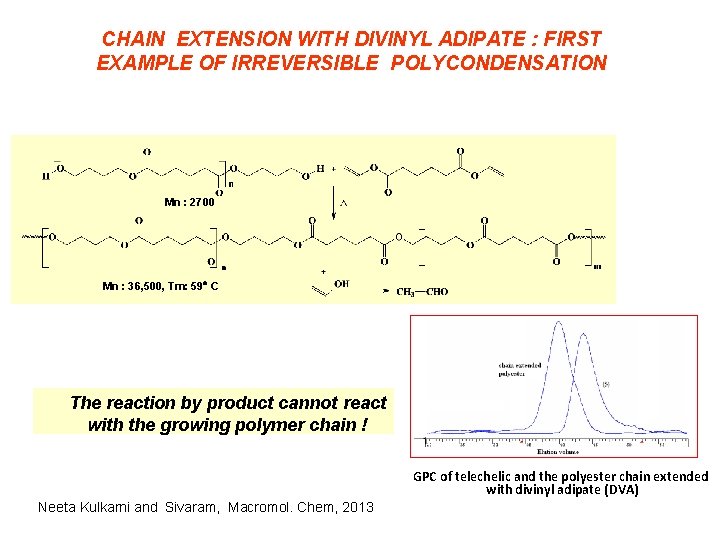
CHAIN EXTENSION WITH DIVINYL ADIPATE : FIRST EXAMPLE OF IRREVERSIBLE POLYCONDENSATION Mn : 2700 Mn : 36, 500, Tm: 59 C The reaction by product cannot react with the growing polymer chain ! GPC of telechelic and the polyester chain extended with divinyl adipate (DVA) Neeta Kulkarni and Sivaram, Macromol. Chem, 2013
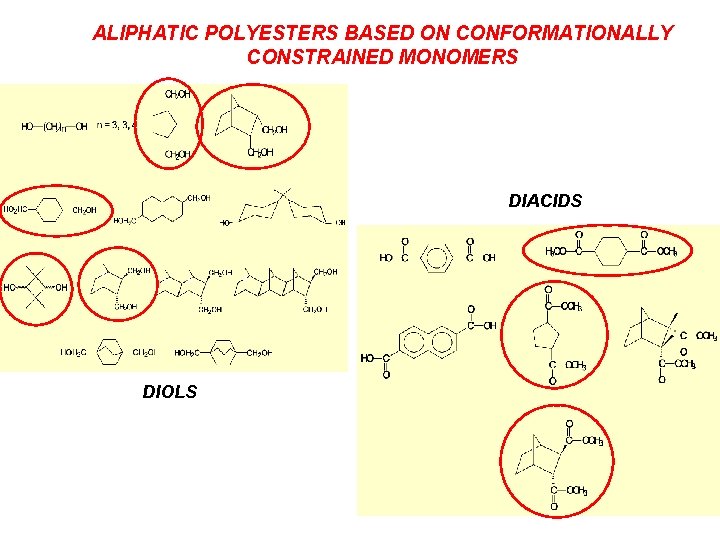
ALIPHATIC POLYESTERS BASED ON CONFORMATIONALLY CONSTRAINED MONOMERS DIACIDS DIOLS
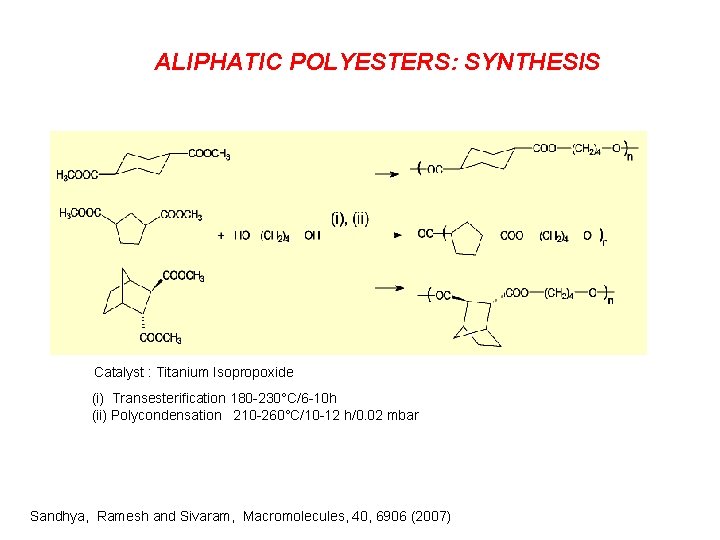
ALIPHATIC POLYESTERS: SYNTHESIS Catalyst : Titanium Isopropoxide (i) Transesterification 180 -230°C/6 -10 h (ii) Polycondensation 210 -260°C/10 -12 h/0. 02 mbar Sandhya, Ramesh and Sivaram, Macromolecules, 40, 6906 (2007)
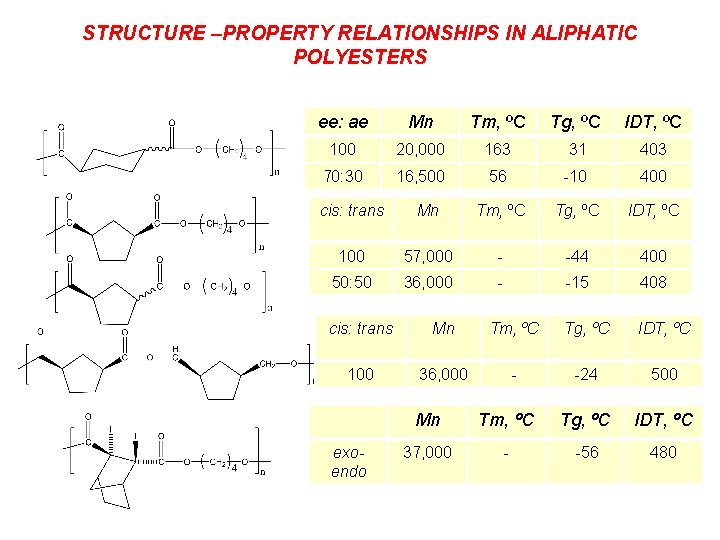
STRUCTURE –PROPERTY RELATIONSHIPS IN ALIPHATIC POLYESTERS ee: ae Mn Tm, ºC Tg, ºC IDT, ºC 100 20, 000 163 31 403 70: 30 16, 500 56 -10 400 cis: trans Mn Tm, ºC Tg, ºC IDT, ºC 100 57, 000 - -44 400 50: 50 36, 000 - -15 408 cis: trans Mn Tm, ºC Tg, ºC IDT, ºC 100 36, 000 - -24 500 exoendo Mn Tm, ºC Tg, ºC IDT, ºC 37, 000 - -56 480

PLLA : MAJOR PROPERTY DEFICITS AND METHODS FOR IMPROVEMENT q Slow rate of crystallization Annealing and cold crystallization Nucleation q Very brittle material ; Elongation : 3 -4 %, Plasticization Copolymerization q Poor heat stability Stereo-complexation Copolymerization q Poor chain entanglement in melt state leading to poor melt viscosities Crosslinking Branching CSIR Proprietary
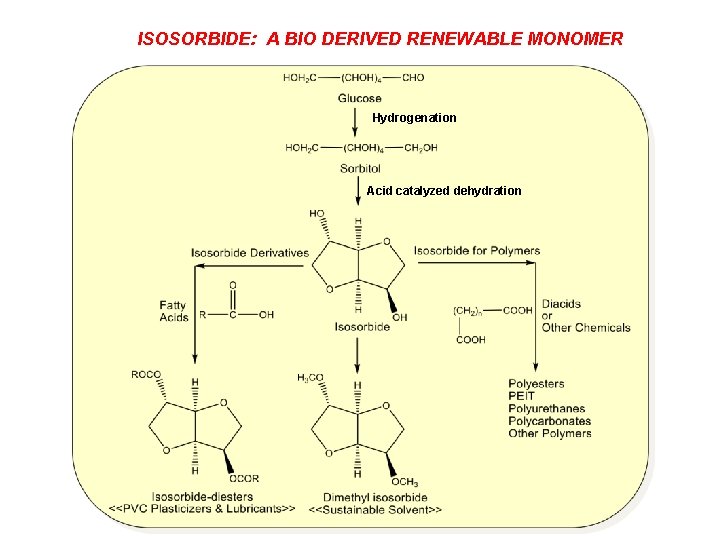
ISOSORBIDE: A BIO DERIVED RENEWABLE MONOMER Hydrogenation Acid catalyzed dehydration
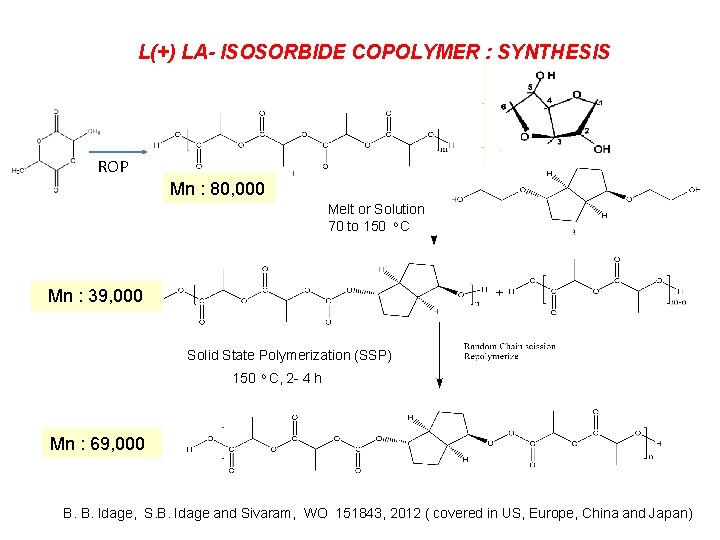
L(+) LA- ISOSORBIDE COPOLYMER : SYNTHESIS ROP Mn : 80, 000 Melt or Solution 70 to 150 o C Mn : 39, 000 Solid State Polymerization (SSP) 150 o C, 2 - 4 h Mn : 69, 000 B. B. Idage, S. B. Idage and Sivaram, WO 151843, 2012 ( covered in US, Europe, China and Japan)
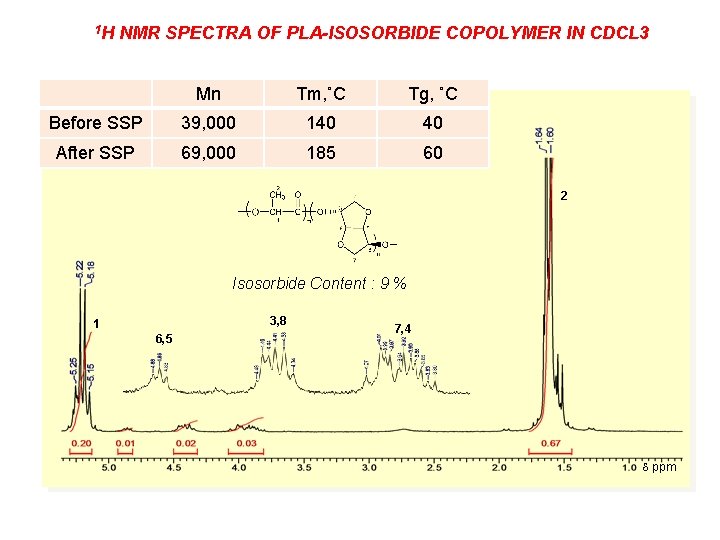
1 H NMR SPECTRA OF PLA-ISOSORBIDE COPOLYMER IN CDCL 3 Mn Tm, ˚C Tg, ˚C Before SSP 39, 000 140 40 After SSP 69, 000 185 60 2 Isosorbide Content : 9 % 3, 8 1 6, 5 7, 4 ppm
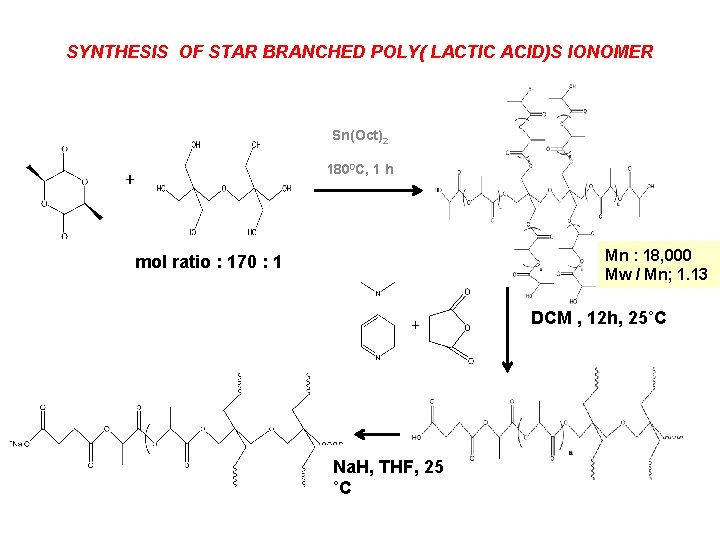
SYNTHESIS OF STAR BRANCHED POLY( LACTIC ACID)S IONOMER Sn(Oct)2 + 1800 C, 1 h Mn : 18, 000 Mw / Mn; 1. 13 mol ratio : 170 : 1 + Na. H, THF, 25 ˚C DCM , 12 h, 25˚C
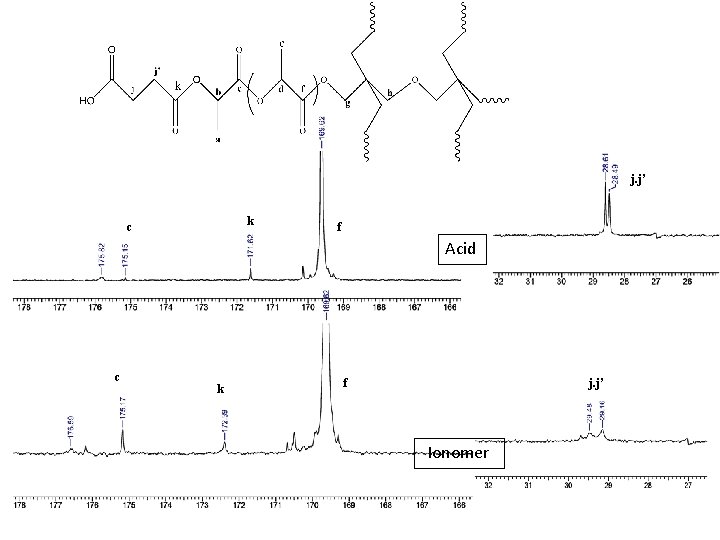
j. j’ k c f Acid c k f j. j’ Ionomer
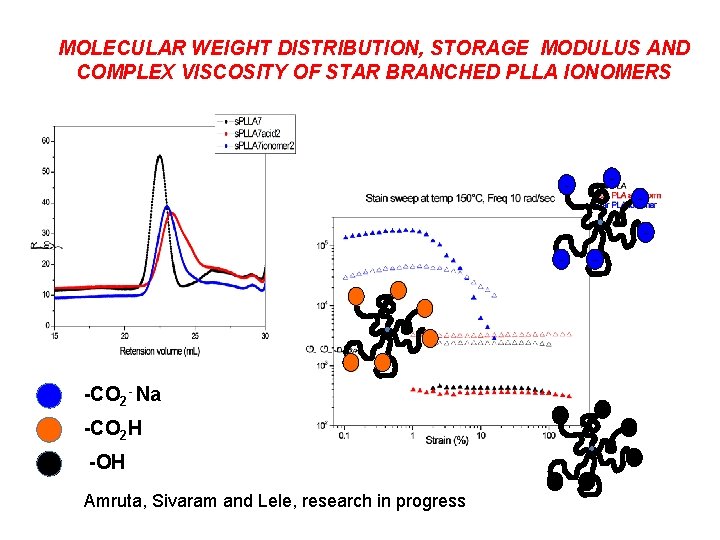
MOLECULAR WEIGHT DISTRIBUTION, STORAGE MODULUS AND COMPLEX VISCOSITY OF STAR BRANCHED PLLA IONOMERS - - -CO 2 - Na -CO 2 H -OH Amruta, Sivaram and Lele, research in progress -

Acknowledgments Students Dr. Sandhya Shankar Dr S. Shyamroy Dr Neeta Kulkarni Mr Yogesh Nevare Ms Amruta Kulkarni Megha Deorkar Maithili Dumbre Ms Dipti Lai Acknowledgments Colleagues Dr B. B. Idage Dr Ms. B. Garnaik Dr. Ashish Lele Financial Support q J. C. Bose Fellowship (DST) q S. S. Bhatnagar Fellowship (CSIR) q CSIR’s TAPSUN Programme

THANK YOU
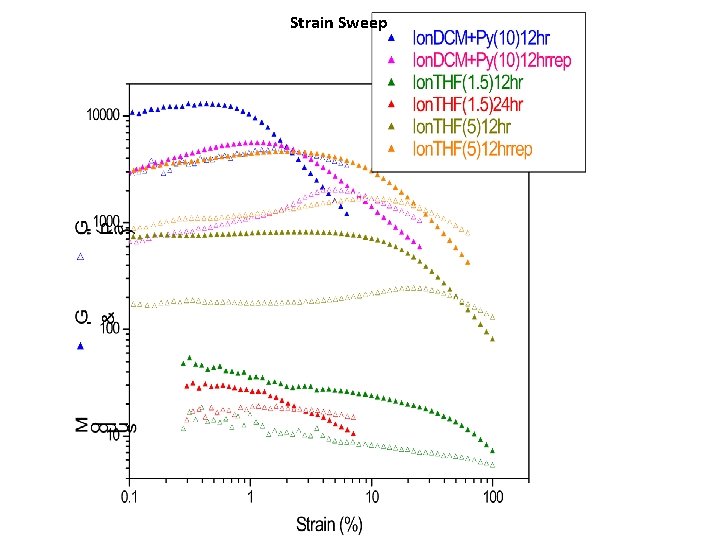
Strain Sweep
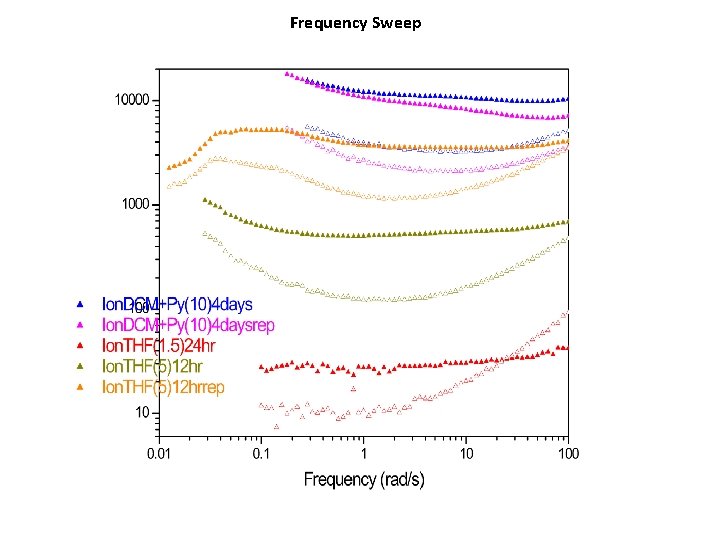
Frequency Sweep
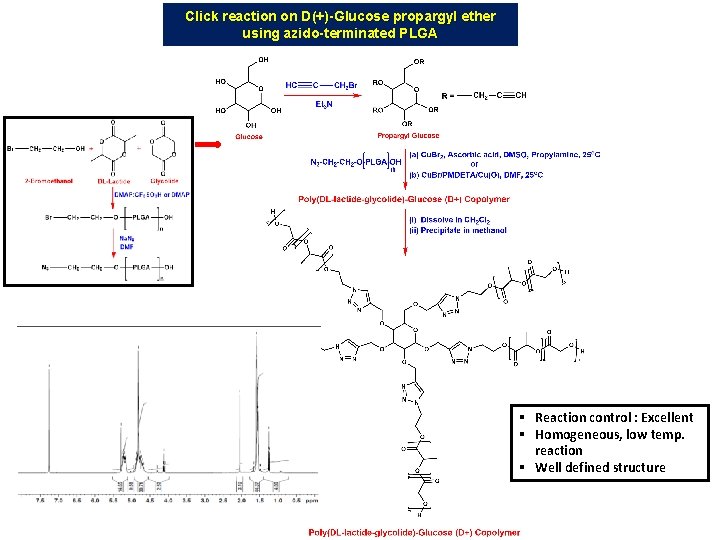
Click reaction on D(+)-Glucose propargyl ether using azido-terminated PLGA § Reaction control : Excellent § Homogeneous, low temp. reaction § Well defined structure
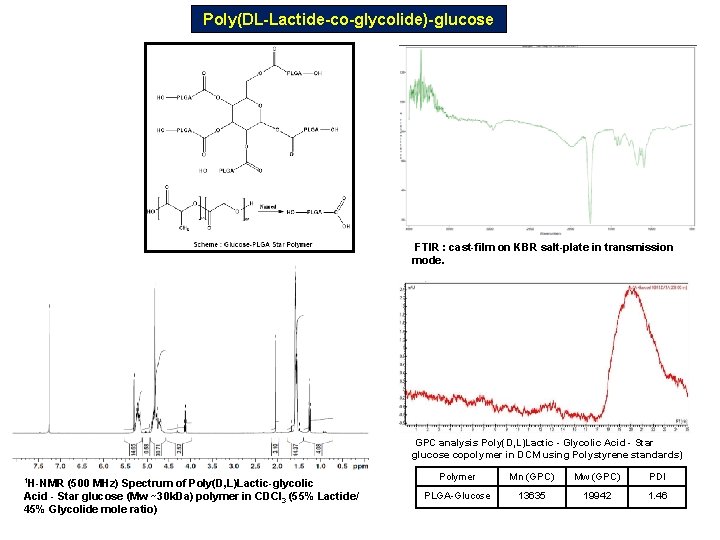
Poly(DL-Lactide-co-glycolide)-glucose FTIR : cast-film on KBR salt-plate in transmission mode. GPC analysis Poly(D, L)Lactic - Glycolic Acid - Star glucose copolymer in DCM using Polystyrene standards) 1 H-NMR (500 MHz) Spectrum of Poly(D, L)Lactic-glycolic Acid - Star glucose (Mw ~30 k. Da) polymer in CDCl 3 (55% Lactide/ 45% Glycolide mole ratio) Polymer Mn (GPC) Mw (GPC) PDI PLGA-Glucose 13635 19942 1. 46
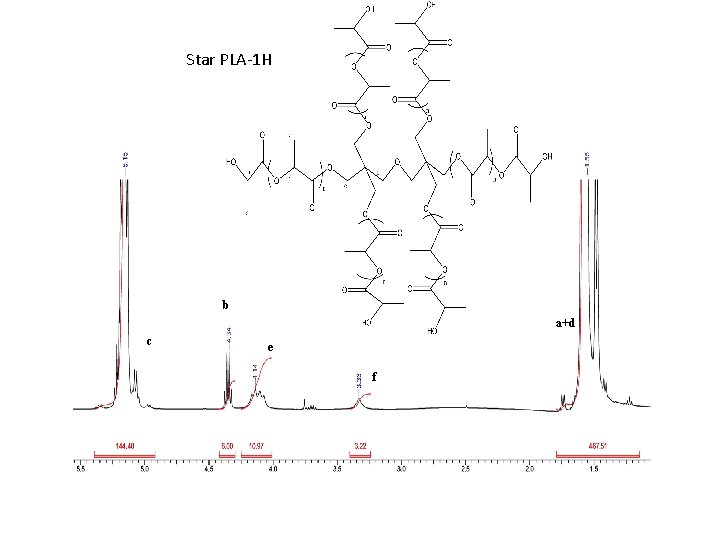
Star PLA-1 H b a+d c e f
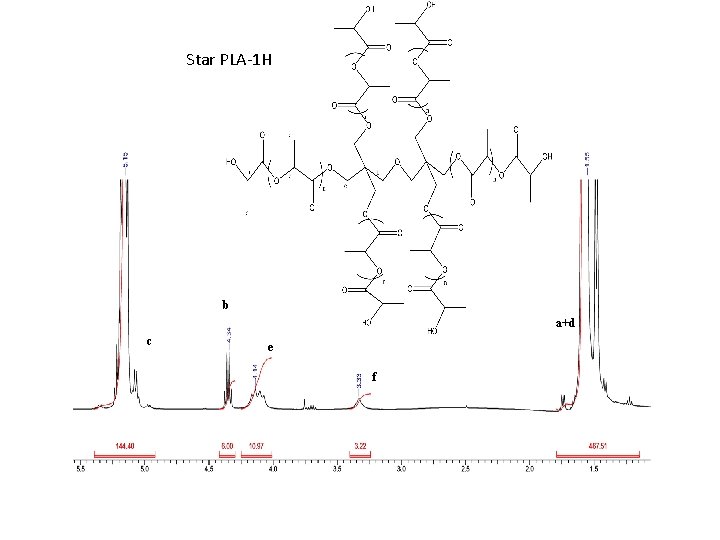
Star PLA-1 H b a+d c e f
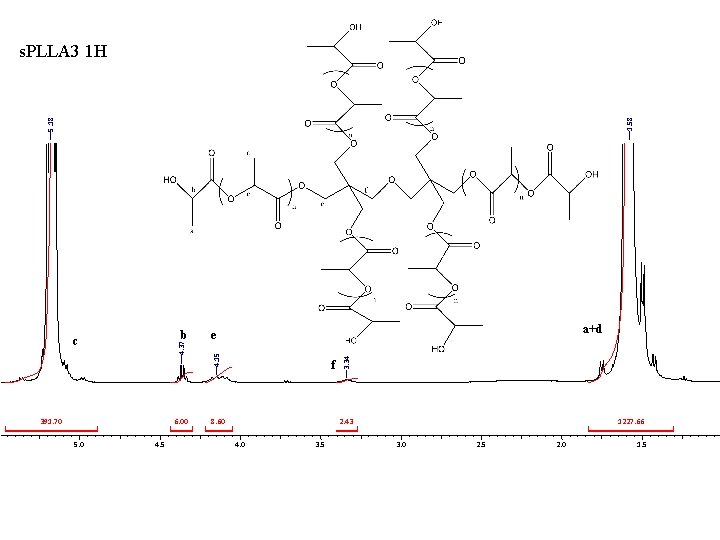
5. 18 1. 58 s. PLLA 3 1 H 6. 00 5. 0 4. 5 f 8. 60 3. 34 4. 37 391. 70 a+d e 4. 15 b c 2. 43 4. 0 3. 5 1227. 66 3. 0 2. 5 2. 0 1. 5
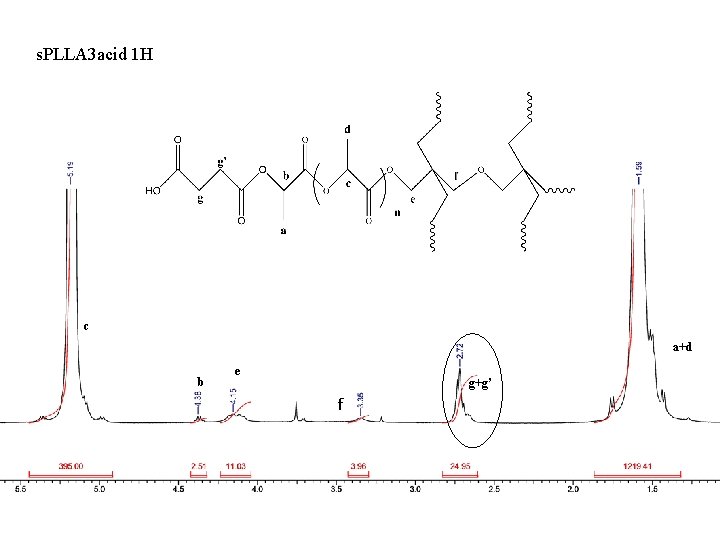
s. PLLA 3 acid 1 H c a+d b e g+g’ f
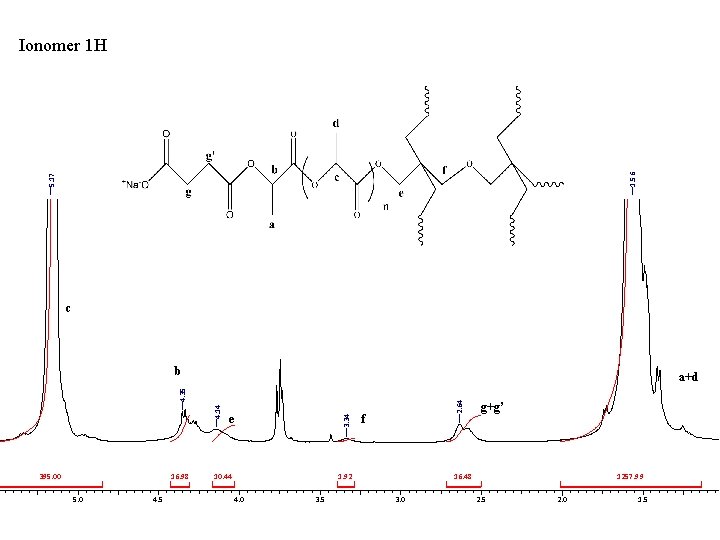
5. 17 1. 56 Ionomer 1 H c b 395. 00 16. 98 5. 0 4. 5 10. 44 1. 92 4. 0 3. 5 2. 64 e 3. 34 4. 14 4. 35 a+d f g+g’ 16. 48 3. 0 1257. 99 2. 5 2. 0 1. 5
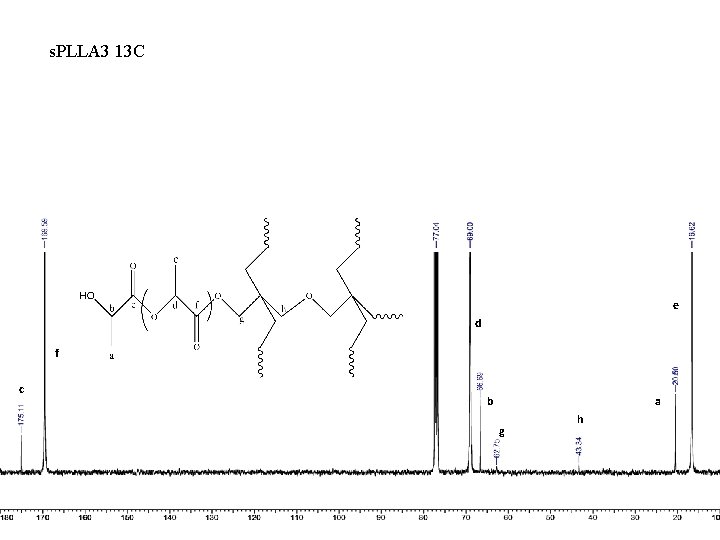
s. PLLA 3 13 C e d f c b a g h
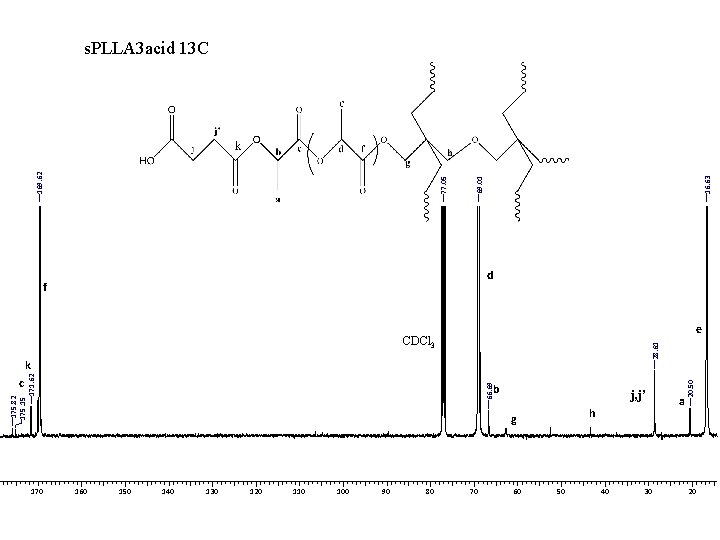
16. 63 69. 01 77. 05 169. 62 s. PLLA 3 acid 13 C d f e 175. 82 175. 15 171. 62 k c 170 66. 69 b j, j’ h g 160 150 140 130 120 110 100 90 80 70 60 50 40 30 a 20. 50 28. 61 CDCl 3 20
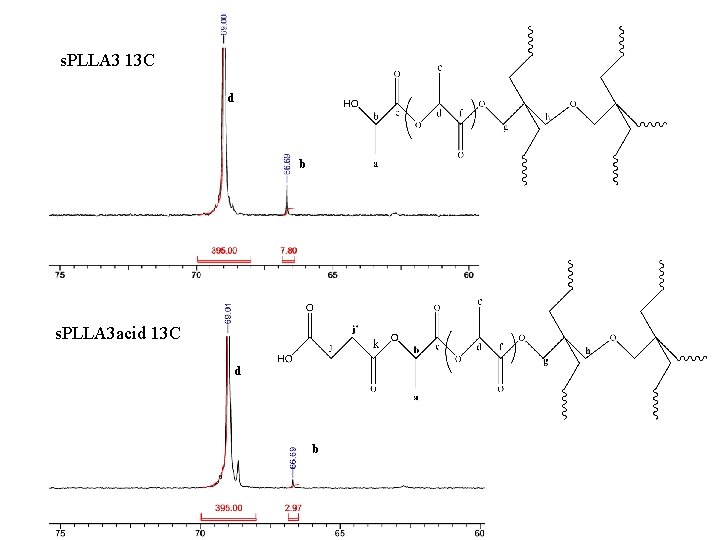
s. PLLA 3 13 C d b s. PLLA 3 acid 13 C d b
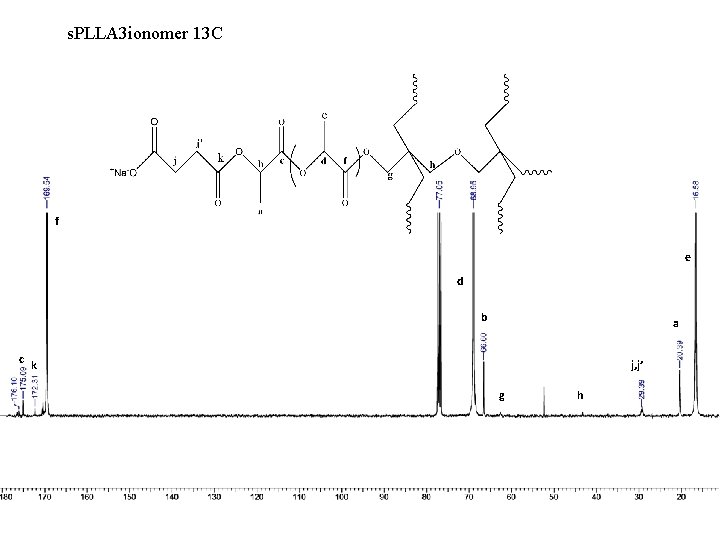
s. PLLA 3 ionomer 13 C f e d b a c k j, j’ g h
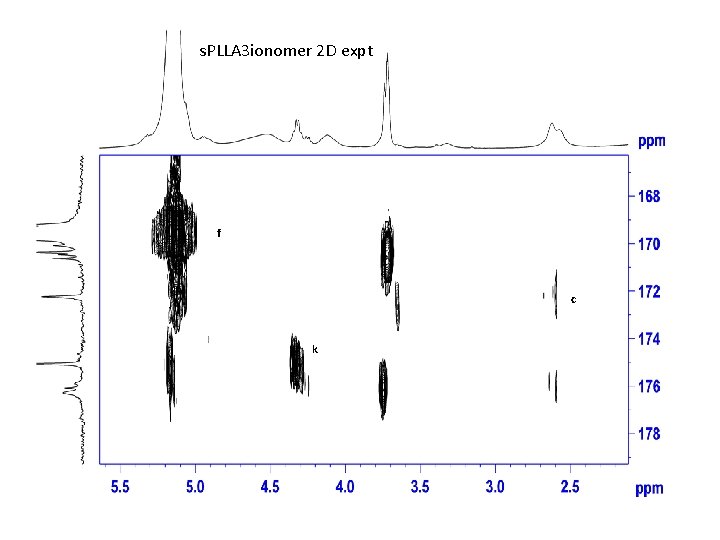
s. PLLA 3 ionomer 2 D expt f c k


SUSTAINABLE POLYMERS FROM BIO-DERIVED AND BIO-RENEWABLE RESOURCES Objectives Perform curiosity-driven and use-inspired research to expand the sustainable chemistry toolbox with new methods and new techniques for next generation products Materials Platform Aliphatic Polyesters
- Slides: 58