SUSPENSION SUSPENSION A pharmaceutical suspension may be defined

SUSPENSION

SUSPENSION • A pharmaceutical suspension may be defined as a coarse dispersion containing finely divided insoluble material suspended in a liquid medium. • The physical chemist defines the word “suspension” as two-phase system consisting of an undissloved or immiscible material dispersed in a vehicle (solid, liquid, or gas). • Generally pharmaceutical suspensions contain aqueous dispersion phase however in some cases they may be an oily or organic phase. • The suspensions have dispersed particles above the colloidal size that is mean particle diameter above 1µm.

Examples of Pharmaceutical Suspensions q. Antacid oral suspensions Antibacterial oral suspension q. Dry powders for oral suspension (antibiotic) q. Analgesic oral suspension q. Anthelmentic oral suspension q. Anticonvulsant oral suspension q. Antifungal oral suspension

Pharmaceutical applications of suspensions 1. Insoluble drug or poorly soluble drugs which required to be given orally in liquid dosage forms ( in case of children, elderly, and patients have difficulty in swallowing solids dosage forms) 2. To over come the instability of certain drug in aqueous solution: a) Insoluble derivative formulated as suspension An example is oxytetracycline HCL (instable) calcium salt (stable) a) Reduce the contact time between solid drug particles and dispersion media increase the stability of drug like Ampicillin by making it as reconstituted powder. b) A drug that degraded in the presence of water suspended in non -aqueous vehicles. Examples are phenoxymethypencillin/ coconut oil and tetracycline HCL/ oil 3. To mask the taste: Examples are paracetamol suspension (more palatable) and chloramphenicol palmitate.

Pharmaceutical applications of suspensions 4. Some materials are needed to be present as finely divided forms to increase the surface area. Fore example, Mg carbonate and Mg trisilcate are used to adsorb some toxins 5. Suspension can be used topical applications: An example is calamine lotion BP after evaporation of dispersing media; the active agent will be left as light deposit 6. Can be used for parentral administration intramuscular (i. m. ) to control arte of absorption 7. In vaccines e. g. Diphtheria and Tetanus vaccines 8. X-ray contrast media: an example is oral and rectal administration of propyliodone 9. In aerosol suspension of active agents in mixture of propellants

Advantages • Suspension can improve chemical stability of certain drug. E. g. Procaine penicillin G • Drug in suspension exhibits higher rate of bioavailability than other dosage forms. bioavailability is in following order, Solution > Suspension > Capsule > Compressed Tablet > Coated tablet • Duration and onset of action can be controlled. E. g. Protamine Zinc-Insulin suspension • Suspension can mask the unpleasant/ bitter taste of drug. E. g. Chloramphenicol

Disadvantages • Physical stability, sedimentation and compaction causes problems. • It is bulky sufficient care must be taken during handling and transport. • It is difficult to formulate • Uniform and accurate dose can not be achieved unless suspension are packed in unit dosage form

Classification • Based On General Classes – Oral suspension – Externally applied suspension – Parenteral suspension • Based On Proportion Of Solid Particles – Dilute suspension (2 to 10 percnt; w/v solid) – Concentrated suspension (50 percnt; w/v solid) • Based On Electrokinetic Nature Of Solid Particles – Flocculated suspension – Deflocculated suspension • Based On Size Of Solid Particles – Colloidal suspension (< 1 micron) – Coarse suspension (>1 micron) – Nano suspension (10 ng)
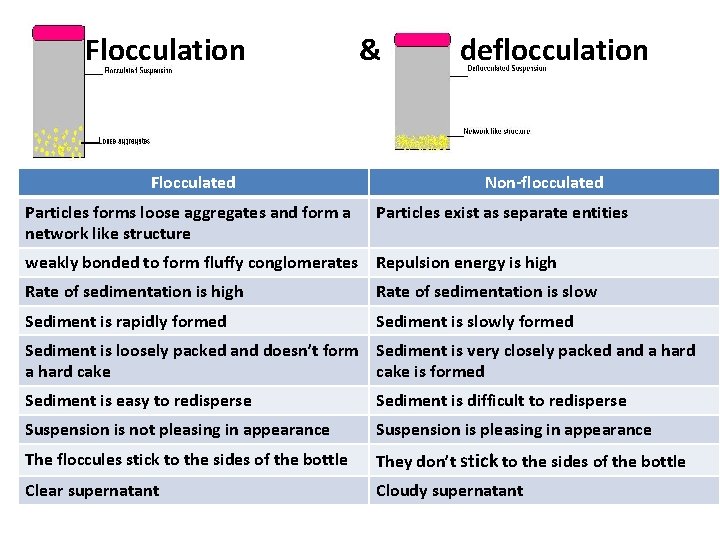
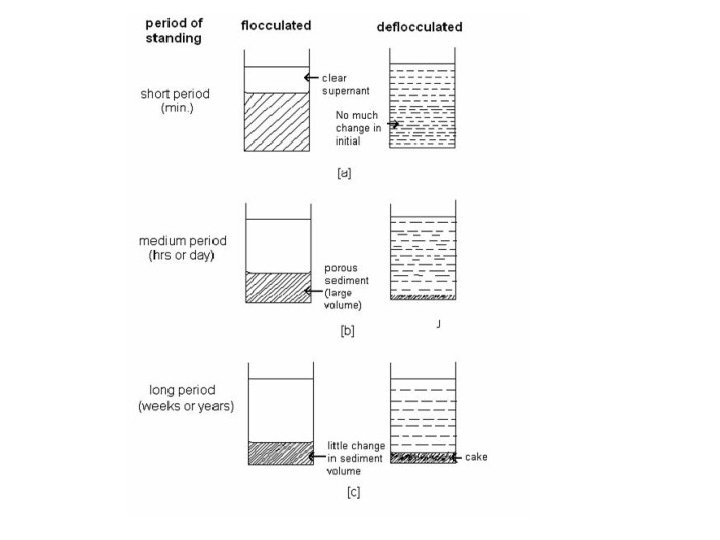
Flocculation Flocculated Particles forms loose aggregates and form a network like structure & deflocculation Non-flocculated Particles exist as separate entities weakly bonded to form fluffy conglomerates Repulsion energy is high Rate of sedimentation is slow Sediment is rapidly formed Sediment is slowly formed Sediment is loosely packed and doesn’t form Sediment is very closely packed and a hard cake is formed Sediment is easy to redisperse Sediment is difficult to redisperse Suspension is not pleasing in appearance Suspension is pleasing in appearance The floccules stick to the sides of the bottle They don’t stick to the sides of the bottle Clear supernatant Cloudy supernatant

Features Desired In Pharmaceutical Suspensions • The suspended particles should not settle rapidly and sediment produced, must be easily re-suspended by the use of moderate amount of shaking. • It should be easy to pour yet not watery and no grittiness. • It should have pleasing odour, colour and palatability. • Good syringeability. • It should be physically, chemically and microbiologically stable. • Parenteral/Ophthalmic suspension should be sterilizable.

Formulation Additives In addition to vehicle, stabilizer, sweetening and flavouring agents, which are common in liquid dosage forms, the following additives are required to prepare suspensions which include: 1. Suspending and Thickening agents 2. Wetting Agents 3. Dispersing agent 4. Flocculating Agent 1. Suspending and Thickening agents: They are added with the objective to increase apparent viscosity of the continuous, phase thus preventing rapid sedimentation of the dispersed particles. a) Natural Polysaccharides : Gum acacia, Tragacanth, sod. Alginate, Xanthan Gum b) Semi-Synthetic Polysaccharides: Sodium Carboxymethyl cellulose, Methyl cellulose, Hydroxypropyl methyl cellulose, microcrystalline cellulose c) Clays: Aluminium Magnesium Silicate, Bentonite, Hectorite d) Synthetic Agents: Carbomer, Colloidal Silicon dioxide

Formulation Additives 2. Wetting Agents: Wetting agents are additives which are usually added to decrease this hydrophobicity. These agents generally get adsorbed at the solid-liquid interface and promote wetting of the solid particles by the liquid of the dispersion medium. a) Surfactants: polysorbates, sorbitan, esters, sodium lauryl sulfate, sodium dioctyl sulfosuccinate b) Hydrophilic Polymers: acacia, bentonite, colloidal silicon dioxide and cellulose derivatives c) Hydrophilic Liquids: alcohol, glycerol, propylene glycol 3. Dispersing agent: These additives are generally added as an aid to uniform distribution and dispersion of solid particles of the dispersed phase. Wetting agents such as surfactants are often employed as dispersing agents. 4. Flocculating Agent: These are substances added to cause controlled aggregation of the particles of the dispersed phase in a suspension. Examples of such agents include surfactants, electrolytes and hydrophilic polymers.
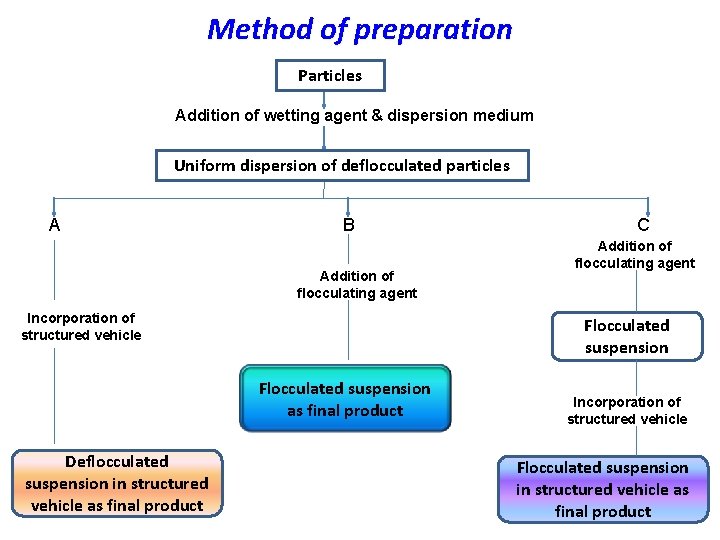
Method of preparation Particles Addition of wetting agent & dispersion medium Uniform dispersion of deflocculated particles A B Addition of flocculating agent Incorporation of structured vehicle Addition of flocculating agent Flocculated suspension as final product Deflocculated suspension in structured vehicle as final product C Incorporation of structured vehicle Flocculated suspension in structured vehicle as final product

Formulation of Suspensions • Suspensions containing diffusible solids • Suspensions containing indiffusible solids • Suspensions containing poorly wettable solids • Suspensions of precipitate forming liquids • Suspensions produced by chemical reactions

• Suspensions containing diffusible solids consist of solids insoluble in water but easily wettable. • On shaking with water solid particles diffuse readily through out the liquid and remain suspended for a long time. • The suspensions containing diffusible solids are prepared by triturating the solids in a mortar with sufficient quantity of vehicle to form a smooth cream. • Any soluble nonvolatile substance is then added by separately dissolving them in a small quantity of vehicle. • More vehicles are then added any foreign particle is strained through a muslin cloth. • Any volatile component is added at this stage and adding the required quantity of vehicle makes up the final volume. • Example: Magnesium Trisilicate Mixture

• Suspensions containing indiffusible solids consist of substances, which do not remain distributed in the dispersion medium when shaken for long time to ensure uniformity of dose. • They are prepared by adding a suitable thickening agent to the vehicle, which increases the viscosity of the vehicle and delays the separation or sedimentation of indiffusible particles. Example: Calamine Lotion

• Suspensions containing poorly wettable solids consist of substances, which are poorly soluble, and at the same time poorly wetted by the dispersion medium, and clump together with the difficulty to disperse. • They are prepared by including suitable wetting agent in the formulation. These agents get adsorbed at the solid/liquid interface and promote wetting of the solid particles by the liquid of the dispersion medium. • Example: Sulphur Lotion

• Suspensions of precipitate forming liquids consist of liquid tinctures which are alcoholic or hydroalcoholic extract of vegetable drugs which contain resinous material. • When tinctures are added to water they precipitate. Precipitates are indiffusible and stick to the walls of the container. • They are prepared by adding a suitable thickening agent prior to the addition of the precipitate forming liquid. Example: Lobelia and Stramonium Mixture

• Suspensions produced by chemical reactions are prepared by mixing two dilute solutions of reactants to form a fine precipitate. • Generally precipitates so formed are diffusible and no suspending agent is required. • If precipitate is indiffusible a suitable thickening or suspending agent may be added. • They are prepared by dissolving the reactants separately in approximately half volumes of the vehicle and the two portions are then mixed together. • Example: Zinc Sulphide Lotion

Preparation of Suspensions • Reduce drug powder to desired size • Add drug and wetting agent to solution • Prepare solution of suspending agent • Add other ingredients – electrolytes, color, flavor • Homogenize medium • Package

Structured Vehicle • Structured vehicles called also thickening or suspending agents. They are aqueous solutions of natural and synthetic gums. These are used to increase the viscosity of the suspension. • Methyl cellulose, carboxymethyl cellulose, sodium carboxymethyl cellulose, acacia, gelatin and tragacanth are the most commonly used structured vehicle in the pharmaceutical suspensions. These are non-toxic, pharmacologically inert, and compatible with a wide range of active and inactive ingredients. • These structured vehicles entrapped the particle and reduces the sedimentation of particles. Although, these structured vehicles reduces the sedimentation of particles, not necessarily completely eliminate the particle settling. Thus, the use of deflocculated particles in a structure vehicle may form solid hard cake upon long storage. • The risk of caking may be eliminated by forming flocculated particles in a structured vehicle. • Note that too high viscosity isn’t desirable and it causes difficulty in pouring and administration. Also, it may affect drug absorption since they adsorb on the surface of particle and suppress the dissolution rate. • Structured vehicles are pseudoplastic or plastic in their rheological behaviors

Preparation Of Structured Vehicle • Structured vehicles are prepared with the help of Hydrocolloids. • In a particular medium, they first hydrolyzed and swell to great degree and increase viscosity at the lower concentration. In addition, it can act as a ‘Protective colloid’ and stabilize charge. • Density of structured vehicle also can be increased by: – – Polyvinylpyrrolidone Sugars Polyethylene glycols Glycerin

Packaging and Storage of Suspensions 1. Should be packaged in wide mouth containers having adequate air space above the liquid. 2. Should be stored in tight containers protected from: freezing, excessive heat & light 3. Label: "Shake Before Use" to ensure uniform distribution of solid particles and thereby uniform and proper dosage. 4. Stored in room temperature if it is dry powder (25 0 C). It should be stored in the refrigerator after opening or reconstitute (freezing should be avoided to prevent aggregation)

Stability of Suspensions A-Physical stability – Appearance, color, odor and taste – p. H – Specific gravity – Sedimentation arte – Sedimentation volume – Zeta potential measurement – Compatibility with container – Compatibility with cap liner – Microscopic examination – Determination crystal size – Determination uniform drug distribution B-Chemical stability: – Degradation of active ingredient – Viscosity change – antimicrobial activity: • Incompatibility with preservative • Degradation of preservative • Adsorption of preservative onto drug particle

Stability of Suspensions • The physical stability of a pharmaceutical suspension is the condition in which the particles do not aggregate and in which they remain uniformly distributed throughout the dispersions. • In order to achieve this ideal situation the suspension should have additive, which are added to achieve ease in resuspension by a moderate amount of agitation. • Taking a case example: In case of dispersion of positively charged particles that is flocculated by addition of an aninonic electrolyte like monobasic potassium phosphate. The physical stability of the system is enhanced by addition of carboxymethylcellulose, Carbopol 934, veegum, tragacanth or bentonite either alone or in combination. No physical incompatibility is recorded as majority of hydrophilic colloids are negatively charged and are compatible with anionic flocculating agents. • When a flocculated suspension of negatively charged particles with a cationic electrolyte is prepared (aluminum chloride) the addition of hydrocolloid may result in an incompatible product resulting in stingy mass, which has no suspending action, and settle rapidly. In such a condition protective agent is added to change the sign on the particles from the negative to positive is employed which can also be achieved by the adsorption onto the particle surface by fatty acid amine or gelatin. • Thus an anionic electrolyte is used to produce floccules that are compatible with negatively charged suspending agent.

Routes of administration of suspension • Suspensions are used to administer insoluble and distasteful substances in a form that is pleasant to taste by providing a suitable form, for the application of dermatological materials to the skin and mucous membrane and for parenteral usage. • Thus suspensions can be administered by oral, topical parenteral and ophthalmic application

Oral suspensions • Patients who have problems in swallowing solid dosage forms require drugs to be dispersed in a liquid. • Oral suspensions permit the formulation of poorly soluble drugs in the form of liquid dosage form. • As these suspensions are to be taken by oral route therefore they must contain suitable flavoring and sweetening agents. • Drugs, which possess unpleasant taste in solution dosage form like paracetamol, chloramphenicol palmitate etc. can be formulated as palatable suspension as they are suitable for administration to peadiatric patients. • Finely divided solids like kaolin, magnesium carbonate etc. , when administered in the form of suspensions will be available to a higher surface area for adsorptive and neutralizing actions in the gastrointestinal tract.

Topical suspensions • These suspensions are meant for external application and therefore should be free from gritty particles. • There consistency may range from fluid to paste. • Example of fluid suspension includes calamine lotion, which leave a deposit of calamine on the skin after evaporation of the aqueous dispersion phase. Zinc cream has a consistency of semisolid. Zinc cream consists of high percentage of powders dispersed in an oily (paraffin) phase.

Parenteral suspensions • These suspensions should be sterile and should possess property of syringability. • Parenteral suspensions are also used to control the rate of absorption. As the absorption rate of the drug is dependent on the dissolution rate of the solid. Therefore by varying the size of the dispersed solid particles the duration and absorption can be controlled. • Vaccines are also formulated as dispersions of killed microorganisms for example in Cholera vaccine or as toxoid adsorbed on to substrate like aluminium hydroxide or phosphate for prolonged antigenic stimulus. For example adsorbed Diphtheria and Tetanus toxoid.

Ophthalmic suspensions • These should also be sterile and should possess very fine particles. • Drugs, which are unstable in aqueous solution, are formulated as stable suspensions using non-aqueous solvents. • For example fractioned coconut oil is used for dispersing tetracycline hydrochloride for ophthalmic use
- Slides: 31