Surveillance of Antibiotic Resistance Microbiology Department KUMS Dr

Surveillance of Antibiotic Resistance Microbiology Department KUMS Dr. Mohajeri 1

Multidrug Resistant Organisms (MDR) Antibiotic-resistant G-ve rods from 1970 s - Klebsiella spp. - Enterobacter spp. - C. freundii - M. morganii - Providencia spp. - Serratia spp. - P. vulgaris - P. aeroginosa 2

Major antimicrobial resistance problems are typically associated with G+ve nosocomial pathogens include: Ø MRSA → 1980 s - HA-MRSA – Healthcare / Hospital Associated MRSA - CA-MRSA – Community Associated MRSA Glycopeptide (vancomycin) – resistant enterococci (VRE) → 1990 Ø Glycopeptide (vancomycin) – intermediate and resistant S. aureus (VISA- VRSA) → 1990 s & 2002 Ø 3

CA- MRSA v infections most commonly are skin and soft tissue infections outbreaks have been reported among children in child-care settings v has recently been reported among postpartum women and infants in a neonatal intensive-care unit (NICU) v 4
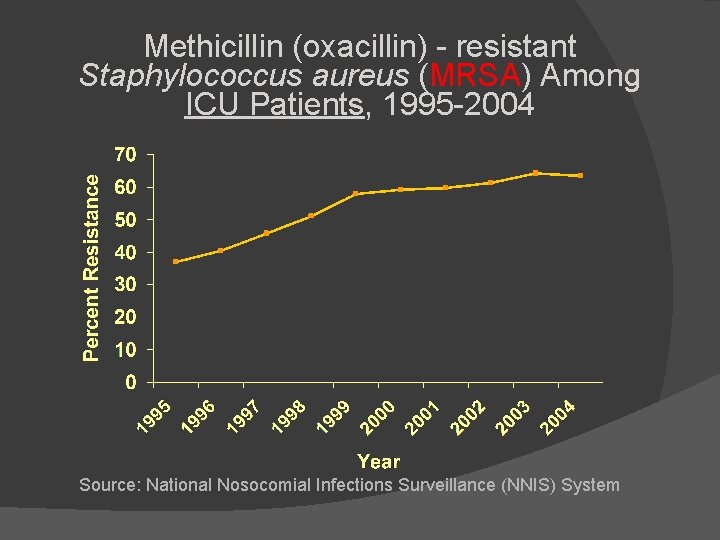
Methicillin (oxacillin) - resistant Staphylococcus aureus (MRSA) Among ICU Patients, 1995 -2004 Source: National Nosocomial Infections Surveillance (NNIS) System

Staphylococcus aureus is important health-care-associated pathogen, is resistant to drying and can persist in the environment and on environmental surfaces for extended periods. ( 26 -27 days) can be shed from heavily colonized persons and discharged into the air People represent the primary reservoir of S. aureus 6

Vancomycin Resistant Enterococci (VRE) E. faecium is the most frequently isolated species of VRE in hospitals Detection of VRE on rectal swabs from colonized patients. High risks: - immunosuppressed - ICU patients - patients previously hospitalized for extended periods - received multi-antimicrobial or vancomycin therapy 7

Many of the environmental surfaces found to be contaminated with VRE In outbreak investigations have been those that are touched frequently by the patient or the health-care worker: - Bed rails - Doorknobs - Bed linens - Gowns - Blood pressure cuffs - Computer table - Bedside tables 8

q Areas where colonized patients are present, but the potential for contamination increases when such patients have diarrhea q Enterococci can persist in a viable state on dry environmental surfaces for extended periods of time (7 days to 4 months) ** The use of stronger solutions of disinfectants for inactivation of either VRE, MRSA or VISA is not recommended because to the organisms are resistance to antibiotics not to disinfectants. 9
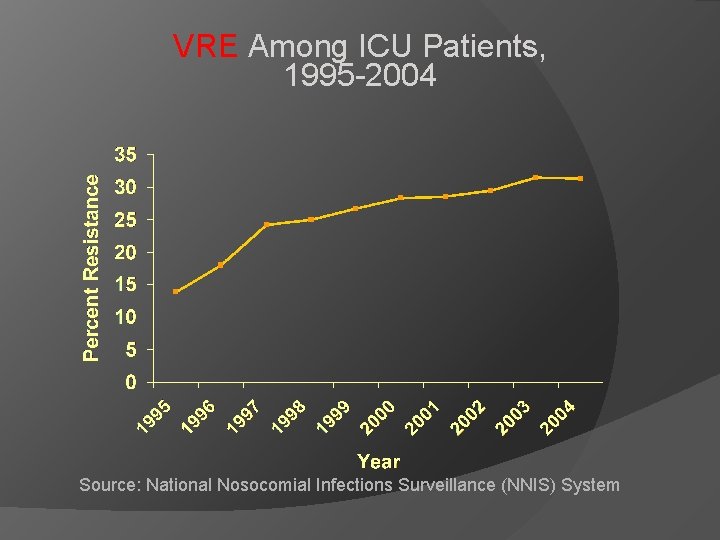
VRE Among ICU Patients, 1995 -2004 Source: National Nosocomial Infections Surveillance (NNIS) System

G-ve bacilli Extended spectrum beta lactamase producing (ESBLs) strains of E. coli and K. pneumoniae → 1990 s ü Resistance to extended-spectrum (third generation) cephalosporins (e. g. , ceftazidime, cefotaxime and ceftriaxone) Fluoroquinolone-resistant strains of P. aeruginosa and E. coli Acinetobacter spp. Candida albicans / Nonalbicans and other Fungi 11

Acinetobacter spp. rarely are associated with episodes of airborne transmission because they generally require moist environments for persistence and growth. The survival periods of Acinetobacter spp. on dry surfaces approximated that for S. aureus (26– 27 days) days have been detected on dry environmental surfaces: - Bed rails - Bed Telephones - Counters - Sinks - Bedding - Floors - Medical charts 12

were cultured from the patients, air and room air conditioners and is prevalent in ICU & Burn Units. Infections caused by Acinetobacter spp. represent a significant clinical problem. Mortality rates associated with Acinetobacteremia are 17– 52%, and rates as high as 71% have been reported for pneumonia. Average infection rates are higher from July through October compared with rates from November through June. 13

Clinically important, opportunistic organisms in tap water include: P. aeruginosa Pseudomonas spp. B. cepacia Stenotrophomonas maltophilia 14
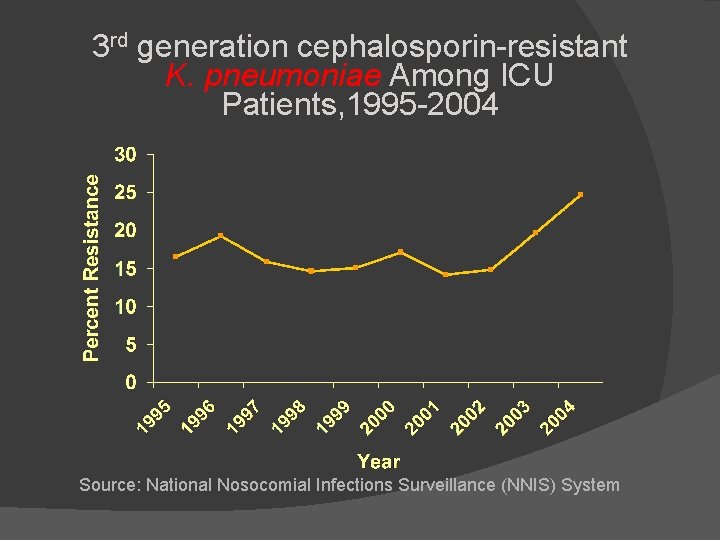
3 rd generation cephalosporin-resistant K. pneumoniae Among ICU Patients, 1995 -2004 Source: National Nosocomial Infections Surveillance (NNIS) System
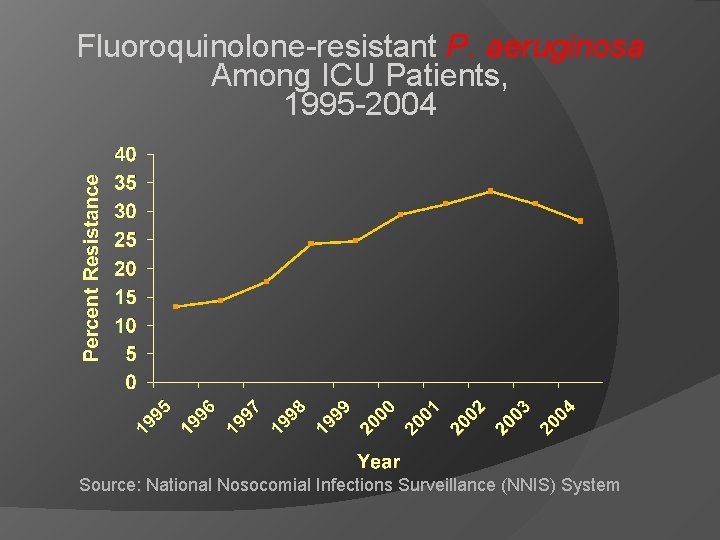
Fluoroquinolone-resistant P. aeruginosa Among ICU Patients, 1995 -2004 Source: National Nosocomial Infections Surveillance (NNIS) System

Clostridium difficile Causes antibiotic-associated diarrhoea and pseudomembranous colitis (PMC) life threatening illnesses Normally affects only the elderly, especially those on long-term broad-spectrum antibiotics is a spore-former, difficult to eradicate, resistant to alcohol. Nosocomial disease spread primarily by hands of staff and “outbreaks” are common. 17

18

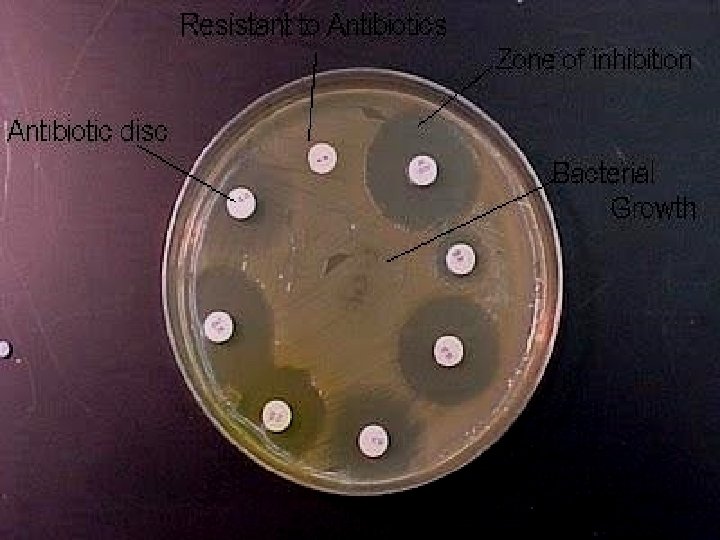
Antibiotic Susceptibility: Antibiotic sensitivity test (AST) is a laboratory method for determining the susceptibility of organisms to therapy with antibiotics. Antibiotic susceptibility testing is usually carried out to determine which antibiotic will be most successful in treating a bacterial infection in vivo. Methods Used: 1. Kirby-Bauer Method (Disc diffusion method). 2. Dilution method. 3. Epsilometer test or simply E-Test.

Preparation of the inoculum: The routinely used method is the turbidity standard (0. 5 Mcfarland). Emulsify 2 -3 colonies in sterile saline matching the turbidity that standard.

1 - Kirby-Bauer Method (Disc Diffusion Test): Commonly used method for determining the antibiotic susceptibility of a bacteria. The test can be done by using : 1. Pure cultures of the organism isolated from the pathologic specimen. 2. Directly on the pathologic specimen (pus, urine, sputum, ect. ) prior to isolation of each organism. Although the former method may preferred, the latter has certain advantages such as: 1. More rapid results. 2. The results will be more relevant to the case , since the inoculums represent more correctly the condition of the patient.

Materials: 1. Muller Hinton Agar. 2. Antibiotic Disks. 3. Turbidity Standard. 4. Swabs.

Procedure: 1. Mostly Muller Hinton agar is used in this antibiotic susceptibility test. 2. Take 24 -48 hours old broth (Liquid) culture of bacteria to be tested. 3. Place a sterile cotton swab in the bacterial suspension and remove the excess fluid by pressing and rotating the cotton against the inside of the tube above the fluid level. 4. The swab is streaked in three directions over the surface of the Mueller-Hinton agar to obtain uniform growth. A final sweep is made around the rim of the agar.

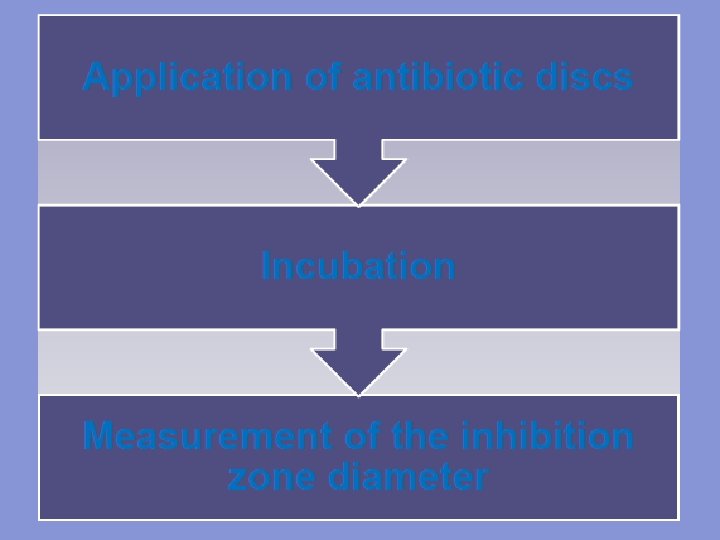
5. Allow the plates to dry for five minutes. 6. Using sterile forceps or a suitable disc dispenser, place paper disks impregnated with a fixed concentration of an antibiotic, on the surface agar plates at equal distance. 7. Incubate the plates at 37 o. C for 24 hours. 8. Following overnight incubation, measure the diameter of the zone of inhibition in millimeter (mm) around each disk.
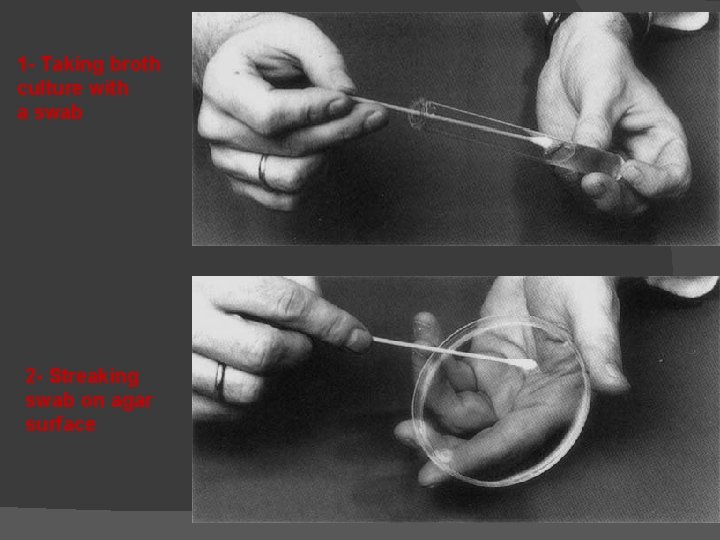
1 - Taking broth culture with a swab 2 - Streaking swab on agar surface
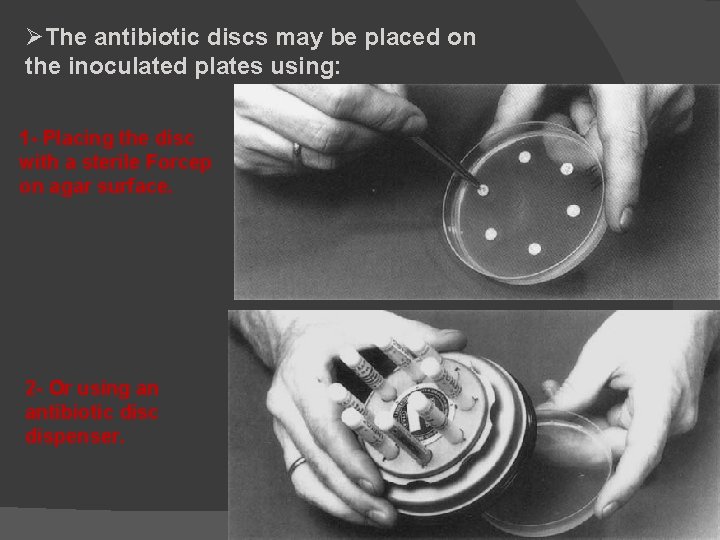
ØThe antibiotic discs may be placed on the inoculated plates using: 1 - Placing the disc with a sterile Forcep on agar surface. 2 - Or using an antibiotic dispenser.
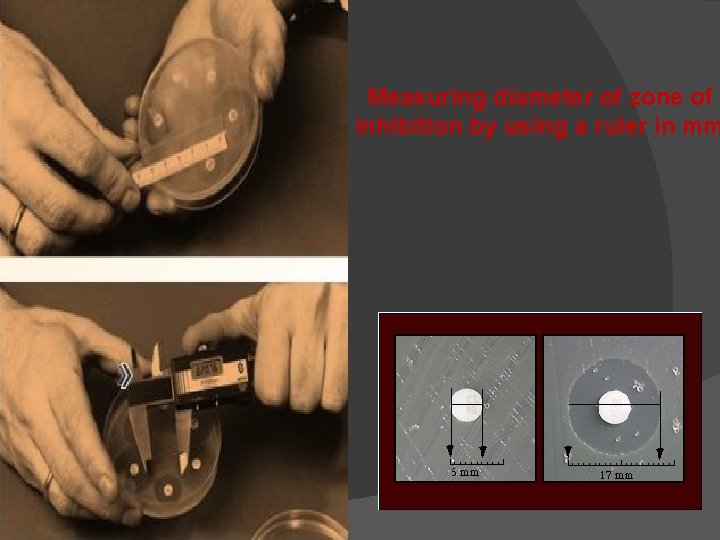
Measuring diameter of zone of inhibition by using a ruler in mm
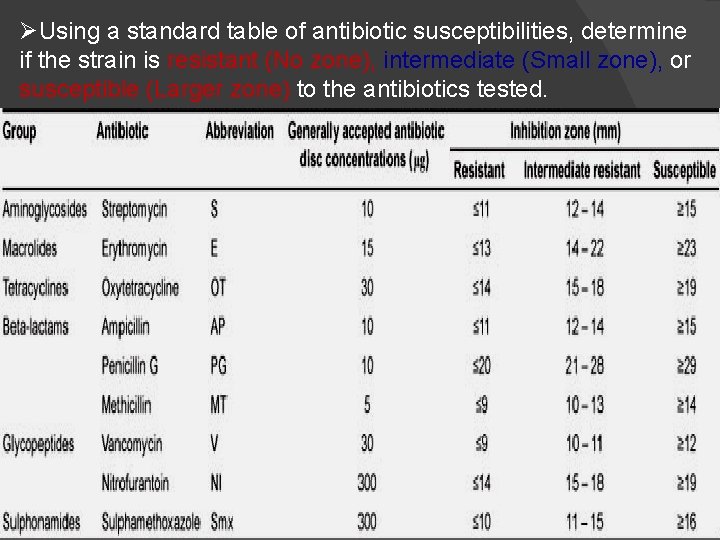
ØUsing a standard table of antibiotic susceptibilities, determine if the strain is resistant (No zone), intermediate (Small zone), or susceptible (Larger zone) to the antibiotics tested.


2 Dilution method: Used to determine the minimal concentration of the antibiotic to inhibit or kill micro organisms. Achieved by: 1. Tube dilution methods. 2. Agar dilution method. Minimum inhibitory concentration (MIC): The lowest concentration of antibiotic that inhibit growth of bacteria. Minimum bactericidal concentration (MBC): Lowest concentration of antibiotic that kills bacteria isolated from patient.

1. Tube dilution methods: In this method we use sterile Muller Hinton broth. We make 2 -folds dilution of antibiotics in the broth i. e. 2µg/ml, 4µg/ml, 8µg/ml, 16µg/ml and so on. Then we add broth culture (0. 1 ml) of test organism to the prepared dilutions. Two types of dilution methods can be used: a) Micro-dilution method: It is performed in 96 well microtiter plate. We use about 0. 1 ml total broth volume.

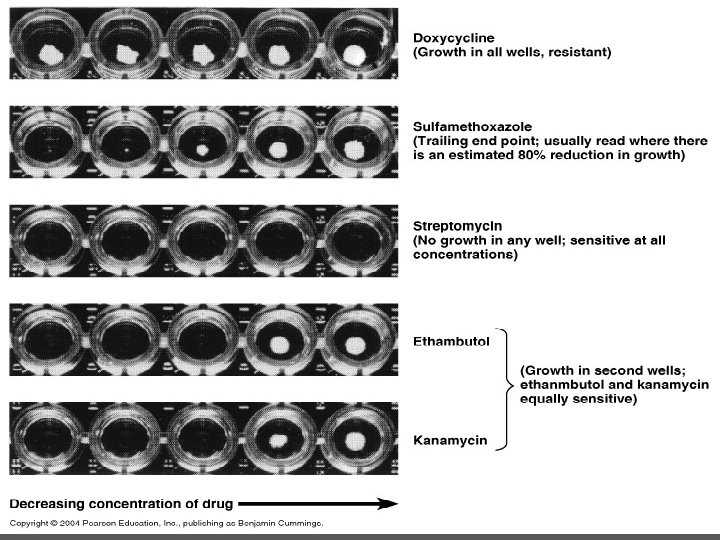

b) Macro-dilution method We use test tubes for this test. We use about 1 ml total broth volume in which we make 2 -folds dilution of antibiotic. Incubation: In both dilution methods, after adding test organism we incubate the tubes at 37 o. C for 24 hours. Then we determine minimum inhibitory concentration (MIC) of antibiotic in µg/ml. Minimum concentration of antibiotic that inhibits the growth of bacteria i. e. when we see a clear broth.

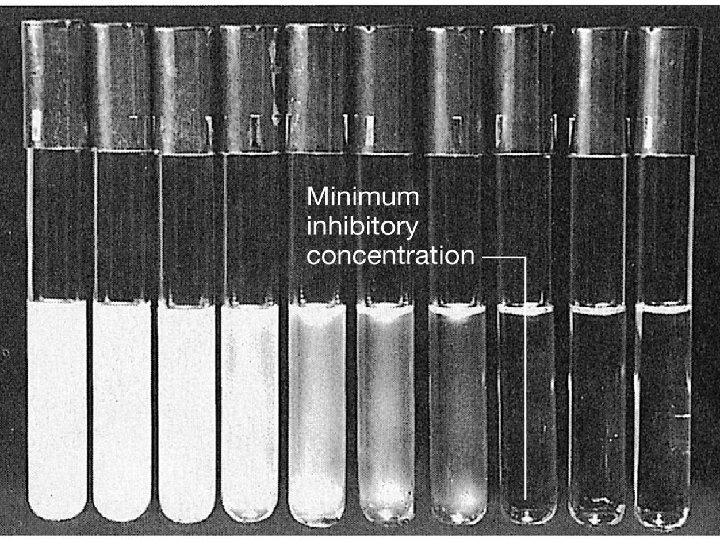
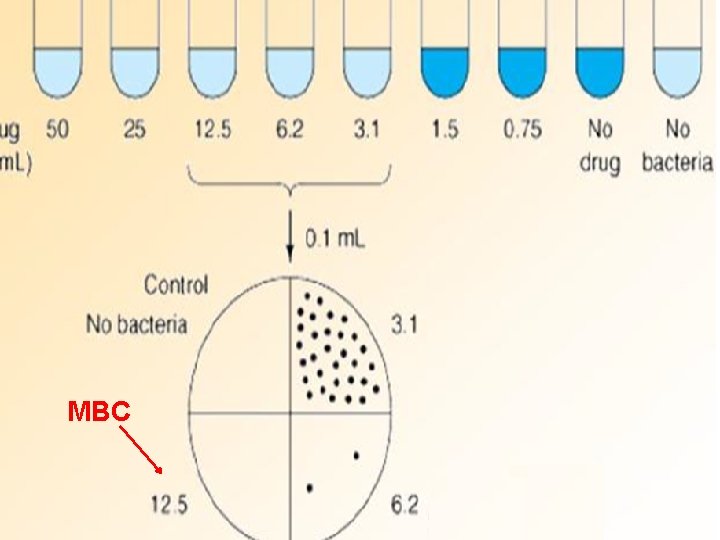
MBC

The minimal bactericidal concentration (MBC) can be determined by sub culturing all tubes showing no visible turbidity. The tube with the highest dilution that fails to yield growth on the subculture plate contains the MBC of antibiotic for the test strain.

2. Agar dilution method: Serial dilution of antibiotics are prepared in agar and poured into plates.

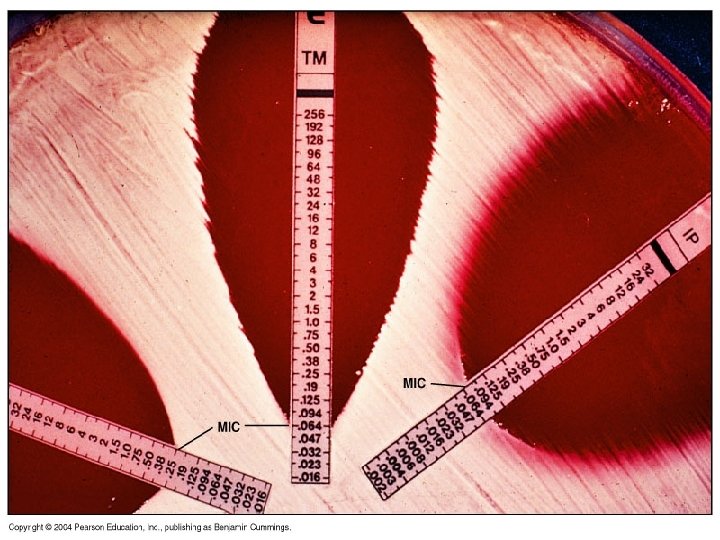
3 - Epsilometer test or E test: It is a quantitative assay for determining the Minimum Inhibitory Concentration (MIC) of antimicrobial agents against microorganisms and for detecting the resistance mechanisms. MIC Test Strip are paper strips with special features that are impregnated with a predefined concentration gradient of antibiotic. On one side of the strip is indicated a MIC scale in μg/ml and a code that identify the antimicrobial agent.

The exponential gradient of antimicrobial agent is immediately transferred to the agar matrix. After 18 hours incubation or longer, a symmetrical inhibition ellipse centered along the strip is formed. The MIC is read directly from the scale in terms of μg/ml at the point where the edge of the inhibition ellipse intersects the MIC Test Strip. Advantages: 1. Simple. 2. Active. 3. Reliable.

Procedure: 1. Take 24 -48 hours old broth (Liquid) culture of bacteria to be tested. 2. Place a sterile cotton swab in the bacterial suspension and remove the excess fluid by pressing and rotating the cotton against the inside of the tube above the fluid level. 3. The swab is streaked in three directions over the surface of the Mueller-Hinton agar to obtain uniform growth. A final sweep is made around the rim of the agar. 4. Allow the plates to dry for five minutes.

5. With the help of sterile forcep apply E-test strips at equal distance on inoculated Muller Hilton agar plate. 6. Incubate the plates at 37 o. C for 24 hours. 7. Following overnight incubation, an inhibition ellipse is produced. 8. Edge of the ellipse corresponding to the antibiotic concentration on the scale indicates the MIC.


Ideal antibiotic therapy is based on determination of the etiological agent and its relevant antibiotic sensitivity. Empiric treatment is often started before laboratory microbiological reports are available when treatment should not be delayed due to the seriousness of the
















ﺳﻮﻳﻪ ﻫﺎﻱ ﻣﺮﺟﻊ ﺑﺮﺍﻱ ﻛﻨﺘﺮﻝ ﻛﻴﻔﻲ Reference Strains for Quality Control § Enterococcus faecalis ATCC 29212; § Escherichia coli ATCC 25922; § Escherichia coli ATCC 35218; § Haemophilus influenzae ATCC 49247; § Haemophilus influenzae ATCC 49766; § Klebsiella pneumoniae ATCC 700603; § Neisseria gonorrhoeae ATCC 49226; § Pseudomonas aeruginosa ATCC 27853; § Staphylococcus aureus ATCC 25923; § Streptococcus pneumoniae ATCC 49619





Enterobacteriaceae Antibiotic Class Ampicillin 10 Cefazolin 30 Gentamicin Amikacin 30 Cefepime 30 Cefotaxime 30 Cefoxitin 30 Ciprofloxacin 5 Imipenem 10 SXT Cefixime 5 Nalidixic acid 30 Nitrofurantoin 300 A A A B B B B O U U Cefazolin 30 Norfloxacin 10 U U 69

Staphylococi Antibiotic Class Cefoxitin A Azithromycin 15 ﺑﺠﺰ ﺍﺩﺭﺍﺭ A Clindamycin 2 A SXT A Doxycycline 30 B Linezolid 30 B Rifampin 5 B Norfloxacin 10 U Nitrofurantoin 300 U Gentamicin 10 C Ciprofloxacin 5 C Amikacin 30 O 70

Pseudomonas Antibiotic Class Pipercillin-Tazobactam 100/10 Ceftazidime 30 Gentamicin 10 A Piperacillin 100 O Tobramycin 10 A Amikacin 30 B Aztreonam 30 B Cefepime 30 B Ciprofloxacin 5 Imipenem 10 B B Colistin O Polymyxin B O Norfloxacin 10 U A A 71
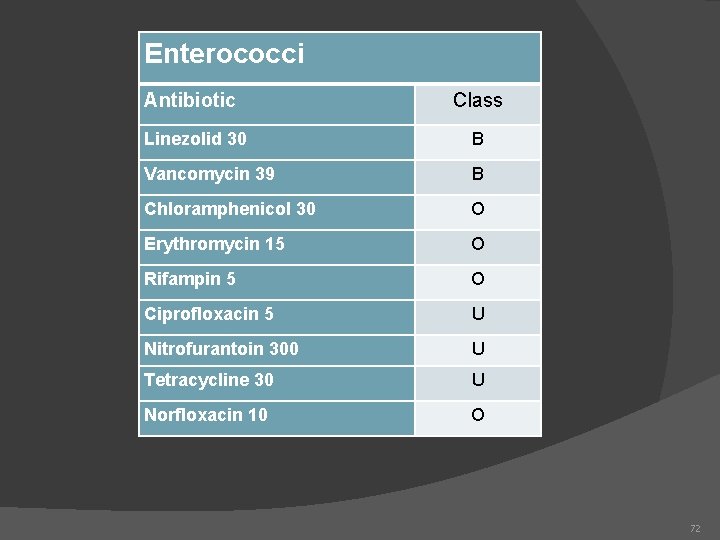
Enterococci Antibiotic Class Linezolid 30 B Vancomycin 39 B Chloramphenicol 30 O Erythromycin 15 O Rifampin 5 O Ciprofloxacin 5 U Nitrofurantoin 300 U Tetracycline 30 U Norfloxacin 10 O 72
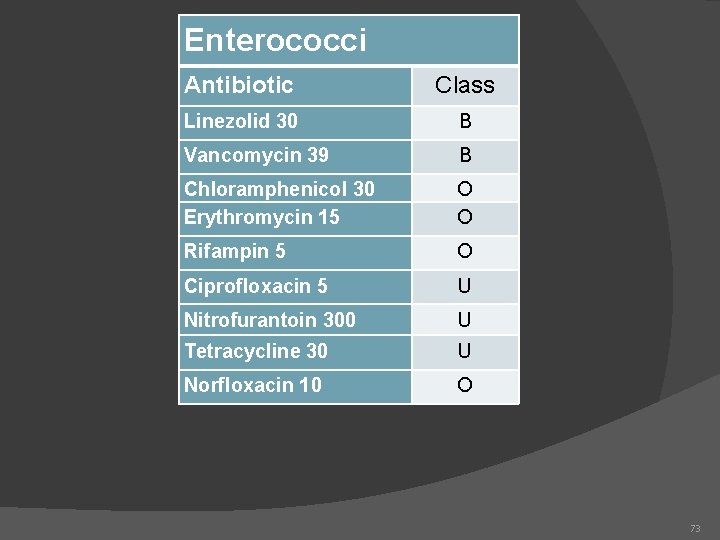
Enterococci Antibiotic Class Linezolid 30 B Vancomycin 39 B Chloramphenicol 30 Erythromycin 15 O O Rifampin 5 O Ciprofloxacin 5 U Nitrofurantoin 300 U Tetracycline 30 U Norfloxacin 10 O 73
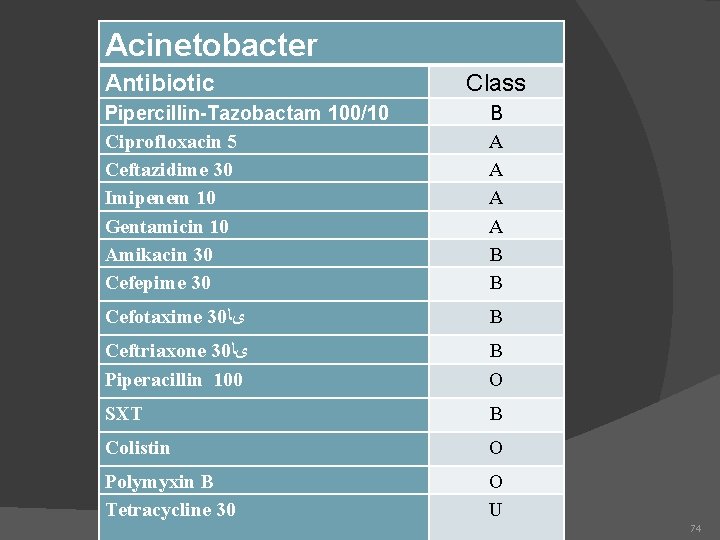
Acinetobacter Antibiotic Class Pipercillin-Tazobactam 100/10 Ciprofloxacin 5 Ceftazidime 30 Imipenem 10 Gentamicin 10 Amikacin 30 Cefepime 30 B A A B B Cefotaxime 30 یﺎ B Ceftriaxone 30 یﺎ Piperacillin 100 B O SXT B Colistin O Polymyxin B Tetracycline 30 O U 74

Stenotrophomonas maltophilia Antibiotic Class SXT Ceftazidime Levofloxacin A B B Chloramphenicol Minocycline B B 75

76
- Slides: 76