Surgery Critical Care PSGS Review Bonaventure Plaza Greenhills

Surgery Critical Care PSGS Review Bonaventure Plaza, Greenhills, San Juan 1 -2 PM; April 26, 2013

Objectives • To discuss key events and situations that have a major role in the outcome of critical care patient management • To discuss areas where targeted goal management plays a major role in achieving positive outcomes in critical care

Discussion Points • Conditions resulting to critical care status • Inflammation state of ICU patient • Optimizing nutrition therapy through: – Early enteral nutrition – Pharmaconutrients – Insulin resistance – Adequate intake – Role of the Nutrition Team

Surgical critical care • Critical illness following surgery or trauma • High risk surgical patients • Shock and hemodynamic compromise • Acute lung injury and ARDS following surgery, trauma, or pancreatitis • Sepsis and severe infections • Trauma evaluation and management • Neurologic emergencies • Post-transplantation • Post-operative complications • Peritonitis, perforated viscus, and abdominal sepsis • Enterocutaneous fistulas • Gastrointestinal hemorrhage • Severe acute pancreatitis • Multisystem organ failure

Shock and hemodynamic compromise Critical illness following surgery or trauma Acute lung injury following surgery or trauma ARDS, complication of acute pancreatitis Sepsis and severe infections Peritonitis, perforated viscus, and abdominal sepsis Enterocutaneous fistulas INFLAMMATION IN THE CRITICAL CARE STATE

SHOCK AND HEMODYNAMIC COMPROMISE

• Body compartment percentage of total body fluids: a. 70% b. 60% c. 50% d. 40%
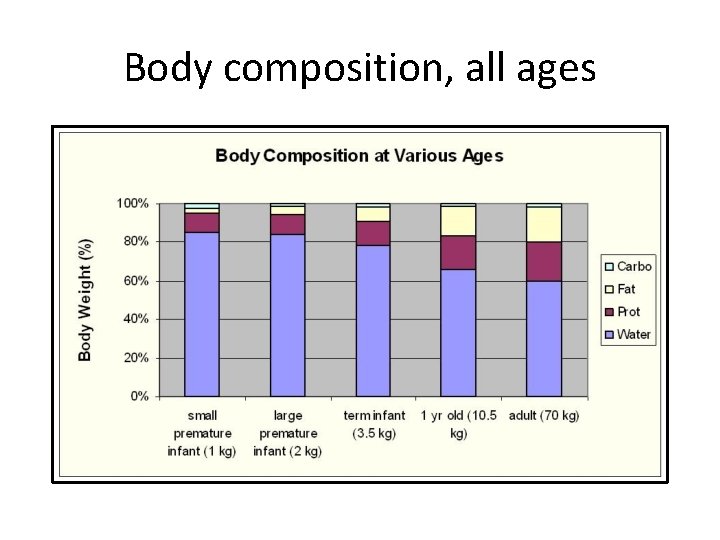
Body composition, all ages
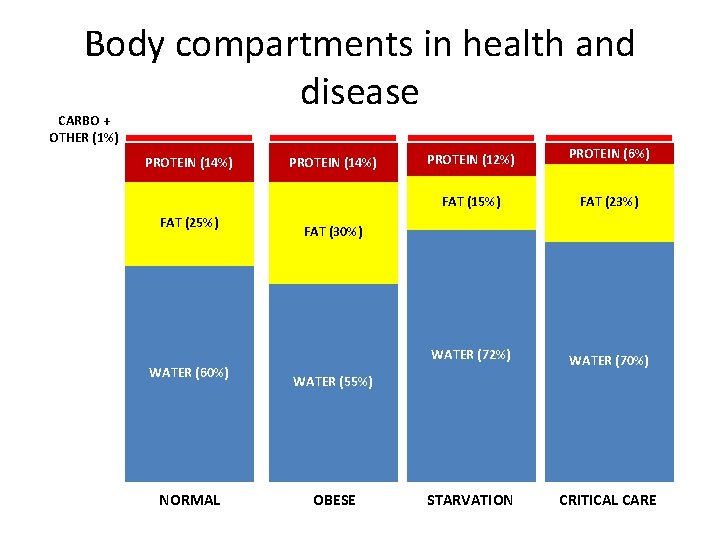
Body compartments in health and disease CARBO + OTHER (1%) PROTEIN (14%) FAT (25%) WATER (60%) NORMAL PROTEIN (14%) PROTEIN (12%) PROTEIN (6%) FAT (15%) FAT (23%) WATER (72%) WATER (70%) STARVATION CRITICAL CARE FAT (30%) WATER (55%) OBESE

• Best solution for volume loss repletion: a. Isotonic saline b. Balanced electrolyte solution c. Colloid d. D 5 LR
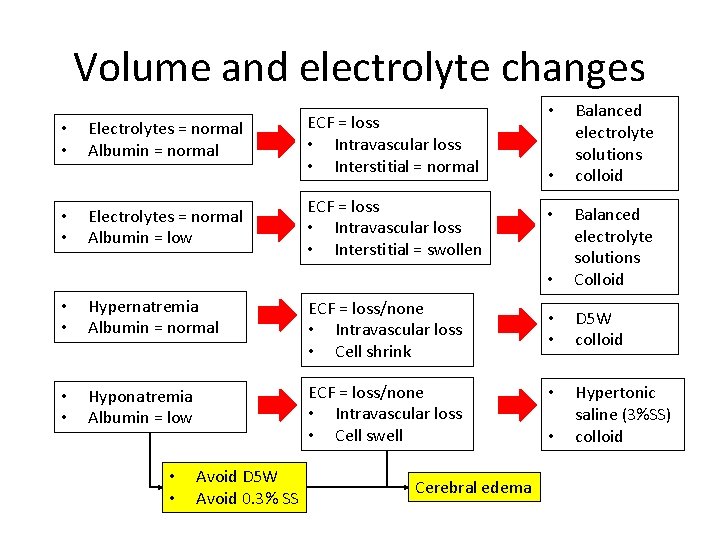
Volume and electrolyte changes • • Electrolytes = normal Albumin = normal Electrolytes = normal Albumin = low ECF = loss • Intravascular loss • Interstitial = normal ECF = loss • Intravascular loss • Interstitial = swollen • • Hypernatremia Albumin = normal ECF = loss/none • Intravascular loss • Cell shrink • • Hyponatremia Albumin = low ECF = loss/none • Intravascular loss • Cell swell • • Avoid D 5 W Avoid 0. 3% SS Cerebral edema • • Balanced electrolyte solutions colloid • Balanced electrolyte solutions Colloid • • D 5 W colloid • Hypertonic saline (3%SS) colloid • •
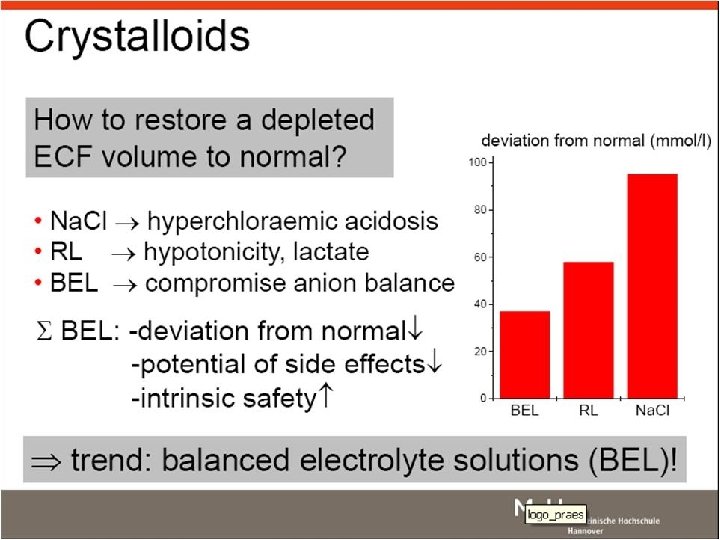
Crystalloids


Resuscitation: fluids Use Compartment Composition Examples Volume Replacement Intravascular fluid volume Iso-oncotic Isotonic Iso-ionic 6% HES 130 in balanced solution Fluid Replacement Extracellular fluid volume Isotonic Iso-ionic Balanced solution: normal saline; ringer’s lactate Electrolyte or osmotherapy (solutions for correction) Total body fluid volume According to need KCL for correction Glucose 5% Mannitol Reference: Zander R, Adams Ha, Boldt J. 2005; 40; 701 -719 18
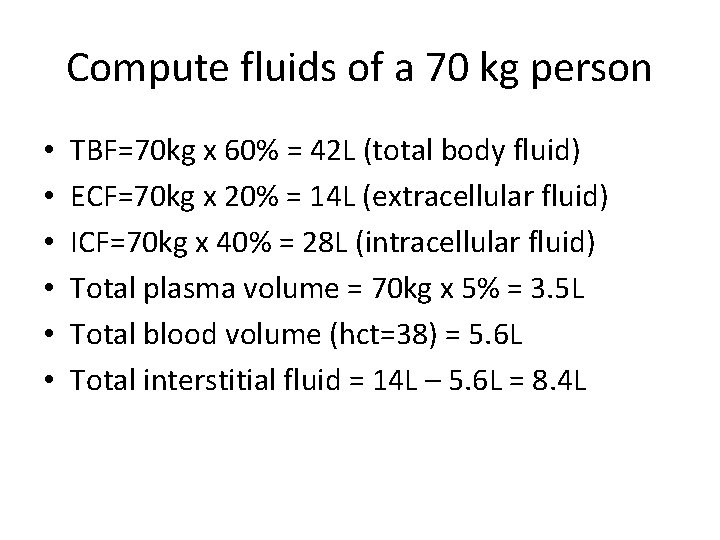
Compute fluids of a 70 kg person • • • TBF=70 kg x 60% = 42 L (total body fluid) ECF=70 kg x 20% = 14 L (extracellular fluid) ICF=70 kg x 40% = 28 L (intracellular fluid) Total plasma volume = 70 kg x 5% = 3. 5 L Total blood volume (hct=38) = 5. 6 L Total interstitial fluid = 14 L – 5. 6 L = 8. 4 L

How much is the total blood volume? • How to compute: – Plasma volume is 5% of actual body weight – Weight=70 kg; hematocrit = 38 – Total plasma volume = 5% x 70 kg = 3500 ml – Total blood volume = 3500 ml x (100/[100 -38]) – TBV = 3500 ml x (100/62) = 3500 ml x 1. 61 – Total blood volume = 5645 ml or 5. 6 liters

Body composition and water Human body composition (% TBF = ICF + ECF = 42 liters of weight): (60% of weight) • Water: 60% • ECF = 14 liters – ECF (extracellular fluid): 20% • Intravascular fluid • Extravascular interstital fluid – ICF (intracellular fluid): 40% • Mass: 40% – Lean body mass – Fat mass – Plasma – Interstitial Fluid • ICF = 28 liters • Computation of usual fluid requirement per day: – 30 ml/kg – or 1. 5 to 2. 5 L/day


Osmolality • Normal cellular function requires normal serum osmolality • Water homeostasis maintains serum osmolality • The contributing factors to serum osmolality are: Na, glucose, and BUN • Sodium is the major contributor (accounts for 90% of extracellular osmolality) • Acute changes in serum osmolality will cause rapid changes in cell volume
![How to compute for plasma osmolality Osmolality = 2 x [Na] + [glucose]/18 + How to compute for plasma osmolality Osmolality = 2 x [Na] + [glucose]/18 +](http://slidetodoc.com/presentation_image_h2/a228142dbce362fa19bbc9237032a535/image-24.jpg)
How to compute for plasma osmolality Osmolality = 2 x [Na] + [glucose]/18 + [BUN]/2. 8 Na = 140 mmol/L Glucose = 110 mg/d. L BUN = 20 mg/d. L Division of glucose and BUN by 18 and 2. 8 converts these to mmol/L Osmolality = (2 x 140) + (110/18) + (20/2. 8) Osmolality = 280 + 6. 1 + 7. 1 Osmolality = 293. 2 mmol/L
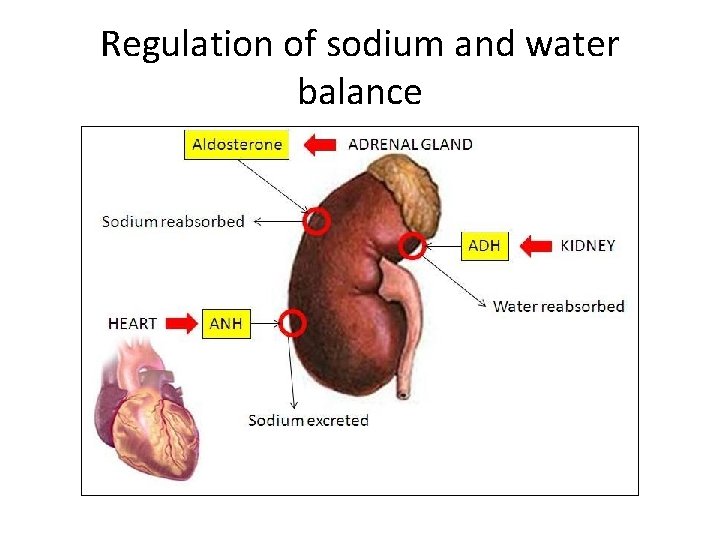
Regulation of sodium and water balance

• Antidiuretic hormone a. Leads to water retention b. Synthesized by the kidney c. Leads to water loss through the urine d. Stimulated by low plasma sodium level

Anti-diuretic hormone
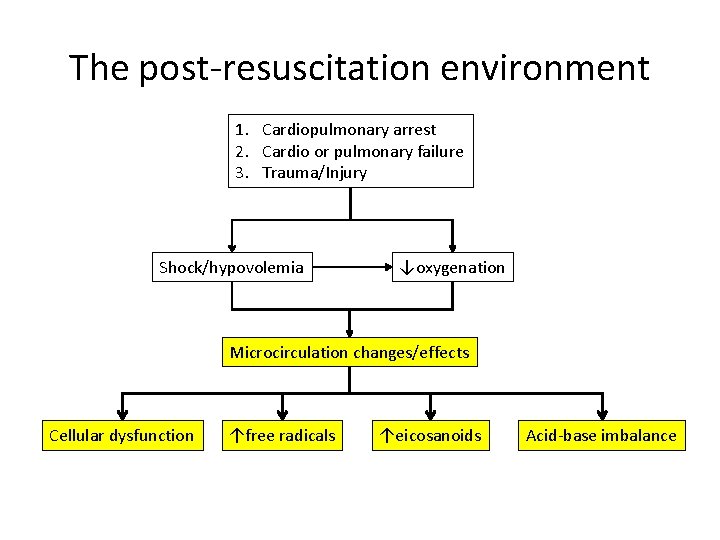
The post-resuscitation environment 1. Cardiopulmonary arrest 2. Cardio or pulmonary failure 3. Trauma/Injury Shock/hypovolemia ↓oxygenation Microcirculation changes/effects Cellular dysfunction ↑free radicals ↑eicosanoids Acid-base imbalance
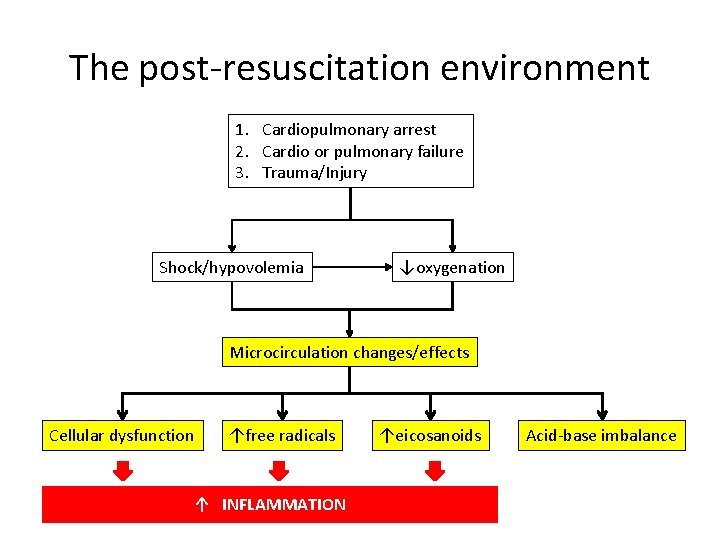
The post-resuscitation environment 1. Cardiopulmonary arrest 2. Cardio or pulmonary failure 3. Trauma/Injury Shock/hypovolemia ↓oxygenation Microcirculation changes/effects Cellular dysfunction ↑free radicals ↑ INFLAMMATION ↑eicosanoids Acid-base imbalance
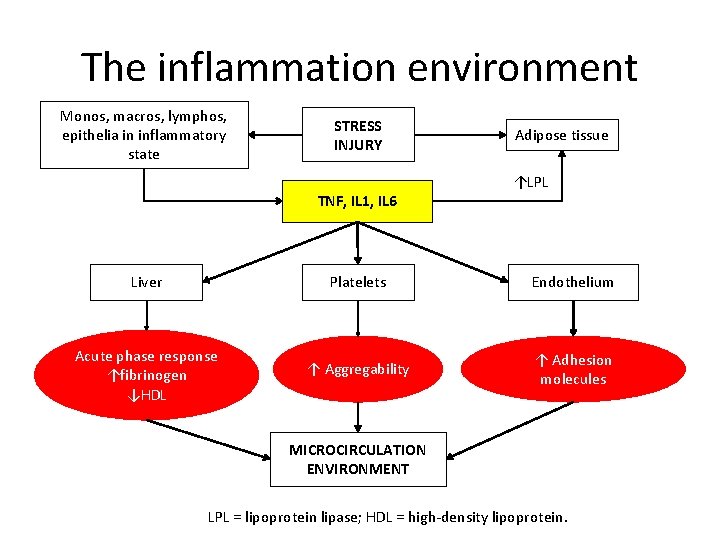
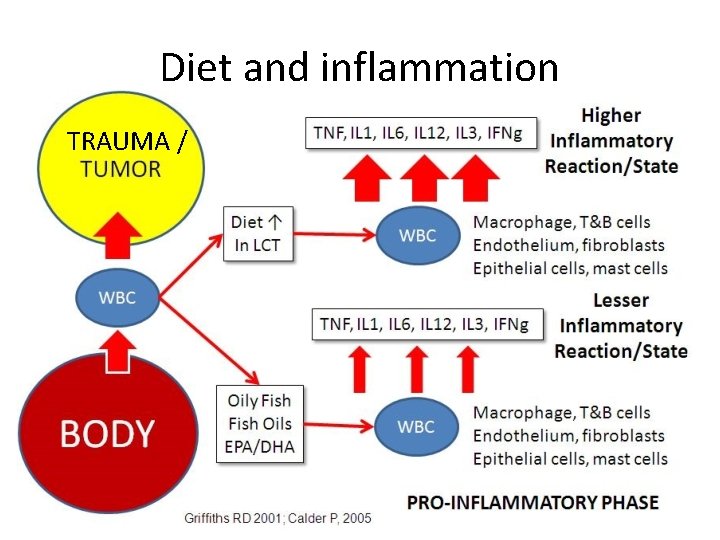
The inflammation environment Monos, macros, lymphos, epithelia in inflammatory state STRESS INJURY TNF, IL 1, IL 6 Adipose tissue ↑LPL Liver Platelets Endothelium Acute phase response ↑fibrinogen ↓HDL ↑ Aggregability ↑ Adhesion molecules MICROCIRCULATION ENVIRONMENT LPL = lipoprotein lipase; HDL = high-density lipoprotein.

Organ status post-resuscitation Cardiac status • electrolyte status • neural activity • muscle activity Lung status • gas exchange • mucosal immunity GUT ISCHEMIA 1. ↑free radicals 2. ↑eicosanoids 3. Accumulation of inflammatory mediators ü Cytokines ü complement 4. ↓digestion and absorption “FIRST HIT”

Organ status post-resuscitation Cardiac status • electrolyte status • neural activity • muscle activity Lung status • gas exchange • mucosal immunity GUT ISCHEMIA 1. ↑free radicals 2. ↑eicosanoids 3. Accumulation of inflammatory mediators ü Cytokines ü complement 4. ↓digestion and absorption “FIRST HIT” RESUSCITATION EFFORTS 1. Reperfusion 2. Laparotomy 3. ICU therapies 4. Gut disuse
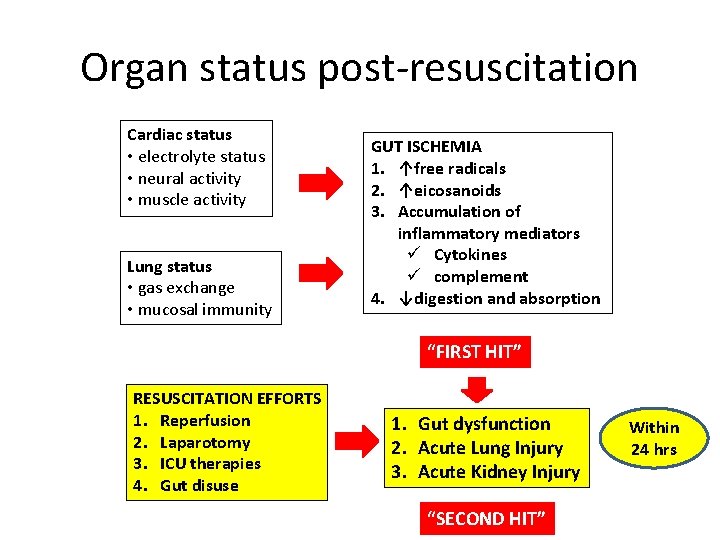
Organ status post-resuscitation Cardiac status • electrolyte status • neural activity • muscle activity Lung status • gas exchange • mucosal immunity GUT ISCHEMIA 1. ↑free radicals 2. ↑eicosanoids 3. Accumulation of inflammatory mediators ü Cytokines ü complement 4. ↓digestion and absorption “FIRST HIT” RESUSCITATION EFFORTS 1. Reperfusion 2. Laparotomy 3. ICU therapies 4. Gut disuse 1. Gut dysfunction 2. Acute Lung Injury 3. Acute Kidney Injury “SECOND HIT” Within 24 hrs
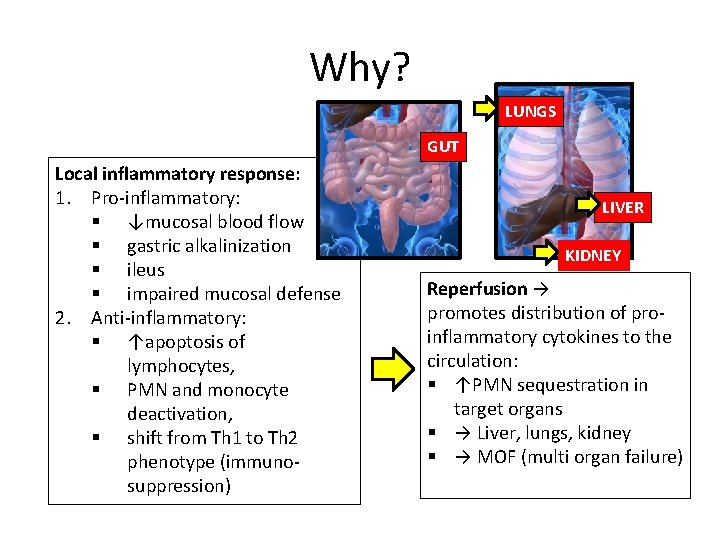
Why? LUNGS GUT Local inflammatory response: 1. Pro-inflammatory: § ↓mucosal blood flow § gastric alkalinization § ileus § impaired mucosal defense 2. Anti-inflammatory: § ↑apoptosis of lymphocytes, § PMN and monocyte deactivation, § shift from Th 1 to Th 2 phenotype (immunosuppression) LIVER KIDNEY Reperfusion → promotes distribution of proinflammatory cytokines to the circulation: § ↑PMN sequestration in target organs § → Liver, lungs, kidney § → MOF (multi organ failure)
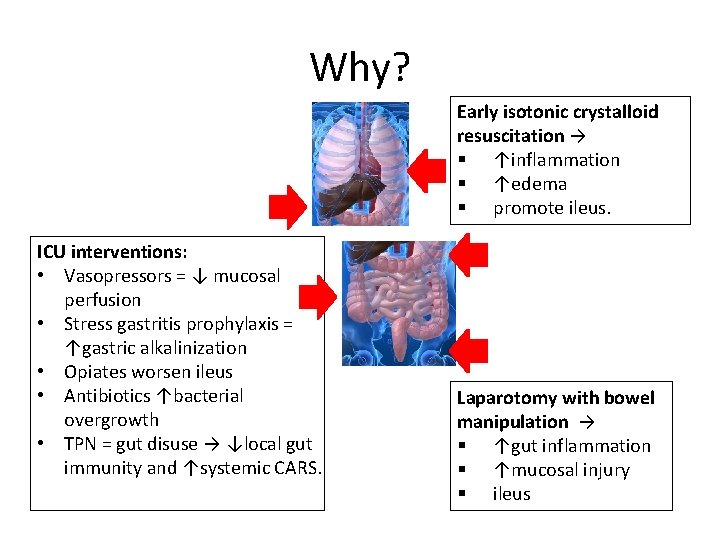
Why? Early isotonic crystalloid resuscitation → § ↑inflammation § ↑edema § promote ileus. ICU interventions: • Vasopressors = ↓ mucosal perfusion • Stress gastritis prophylaxis = ↑gastric alkalinization • Opiates worsen ileus • Antibiotics ↑bacterial overgrowth • TPN = gut disuse → ↓local gut immunity and ↑systemic CARS. Laparotomy with bowel manipulation → § ↑gut inflammation § ↑mucosal injury § ileus
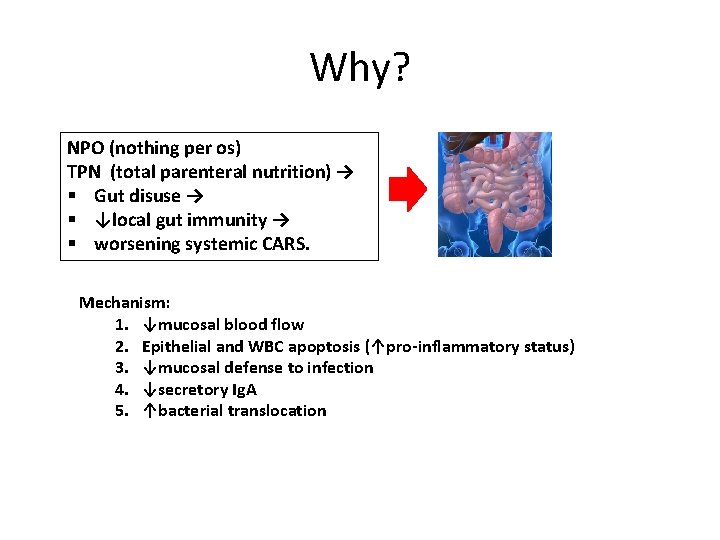
Why? NPO (nothing per os) TPN (total parenteral nutrition) → § Gut disuse → § ↓local gut immunity → § worsening systemic CARS. Mechanism: 1. ↓mucosal blood flow 2. Epithelial and WBC apoptosis (↑pro-inflammatory status) 3. ↓mucosal defense to infection 4. ↓secretory Ig. A 5. ↑bacterial translocation
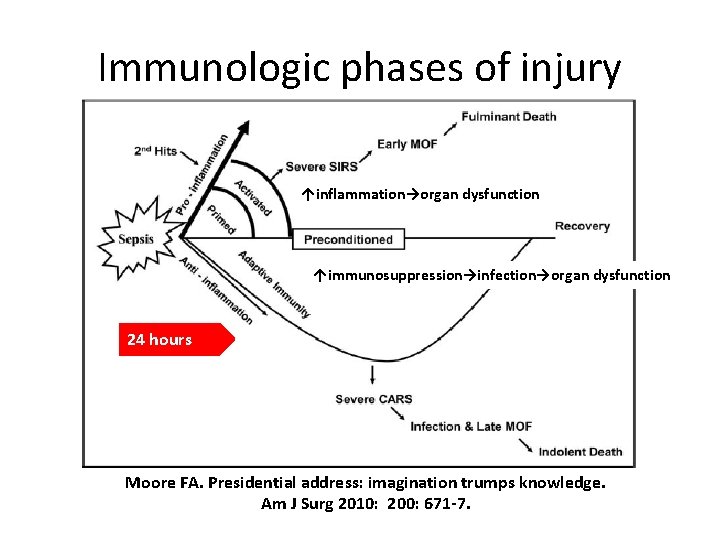
Immunologic phases of injury ↑inflammation→organ dysfunction ↑immunosuppression→infection→organ dysfunction 24 hours Moore FA. Presidential address: imagination trumps knowledge. Am J Surg 2010: 200: 671 -7.
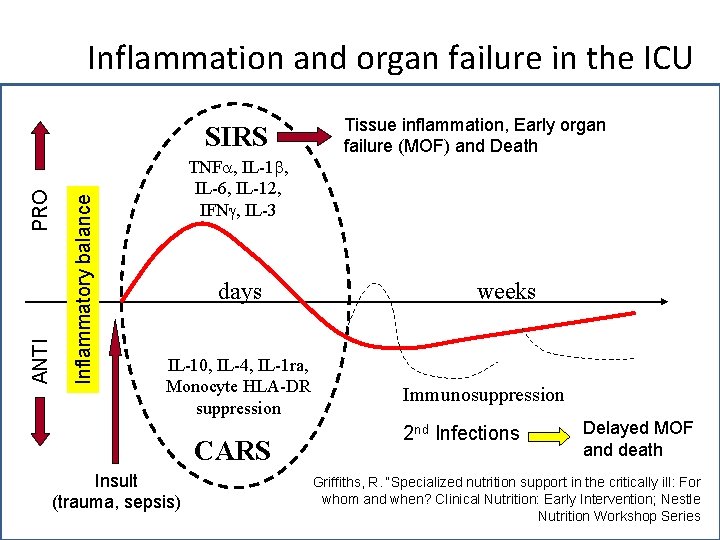
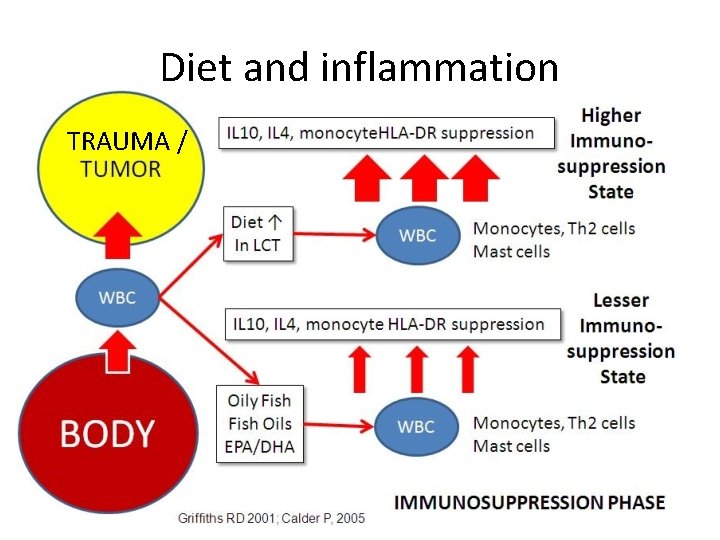
Inflammation and organ failure in the ICU Inflammatory balance ANTI PRO SIRS Tissue inflammation, Early organ failure (MOF) and Death TNF , IL-1 , IL-6, IL-12, IFN , IL-3 days IL-10, IL-4, IL-1 ra, Monocyte HLA-DR suppression CARS Insult (trauma, sepsis) weeks Immunosuppression 2 nd Infections Delayed MOF and death Griffiths, R. “Specialized nutrition support in the critically ill: For whom and when? Clinical Nutrition: Early Intervention; Nestle Nutrition Workshop Series

INSULIN RESISTANCE
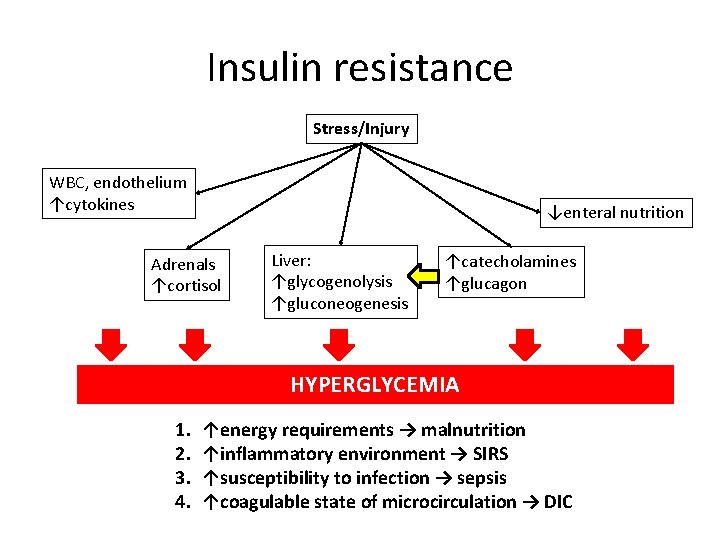
Insulin resistance Stress/Injury WBC, endothelium ↑cytokines ↓enteral nutrition Adrenals ↑cortisol Liver: ↑glycogenolysis ↑gluconeogenesis ↑catecholamines ↑glucagon HYPERGLYCEMIA 1. 2. 3. 4. ↑energy requirements → malnutrition ↑inflammatory environment → SIRS ↑susceptibility to infection → sepsis ↑coagulable state of microcirculation → DIC
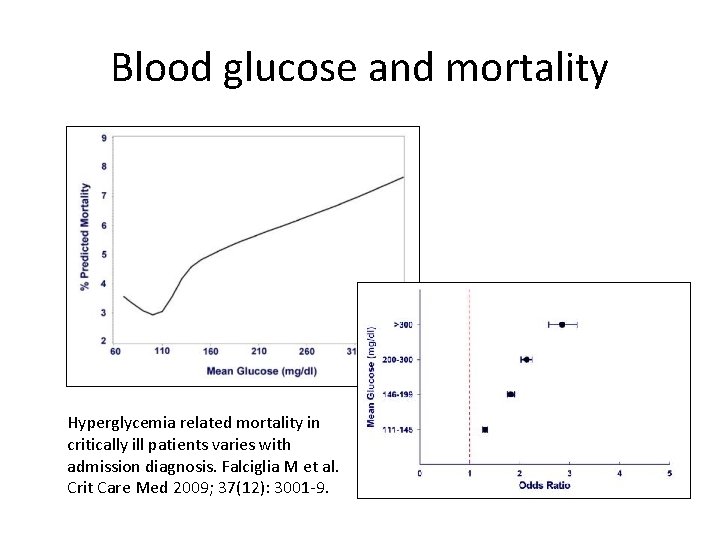
Blood glucose and mortality Hyperglycemia related mortality in critically ill patients varies with admission diagnosis. Falciglia M et al. Crit Care Med 2009; 37(12): 3001 -9.
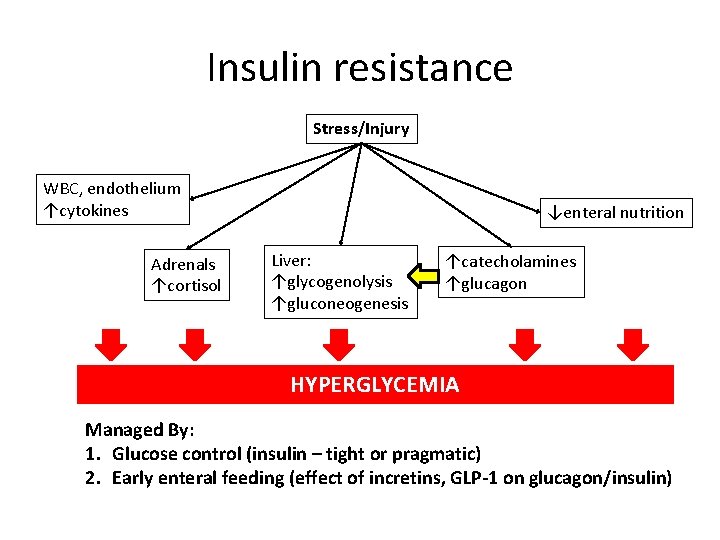
Insulin resistance Stress/Injury WBC, endothelium ↑cytokines Adrenals ↑cortisol ↓enteral nutrition Liver: ↑glycogenolysis ↑gluconeogenesis ↑catecholamines ↑glucagon HYPERGLYCEMIA Managed By: 1. Glucose control (insulin – tight or pragmatic) 2. Early enteral feeding (effect of incretins, GLP-1 on glucagon/insulin)
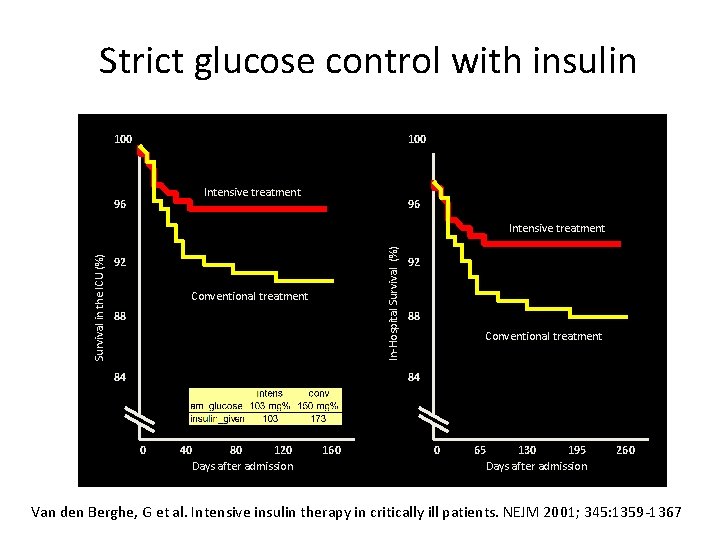
Strict glucose control with insulin 100 Intensive treatment 96 96 In-Hospital Survival (%) Survival in the ICU (%) Intensive treatment 92 Conventional treatment 88 84 92 88 Conventional treatment 84 0 40 80 120 Days after admission 160 0 65 130 195 Days after admission 260 Van den Berghe, G et al. Intensive insulin therapy in critically ill patients. NEJM 2001; 345: 1359 -1367

EARLY ENTERAL NUTRITION

“ NPO until further orders”
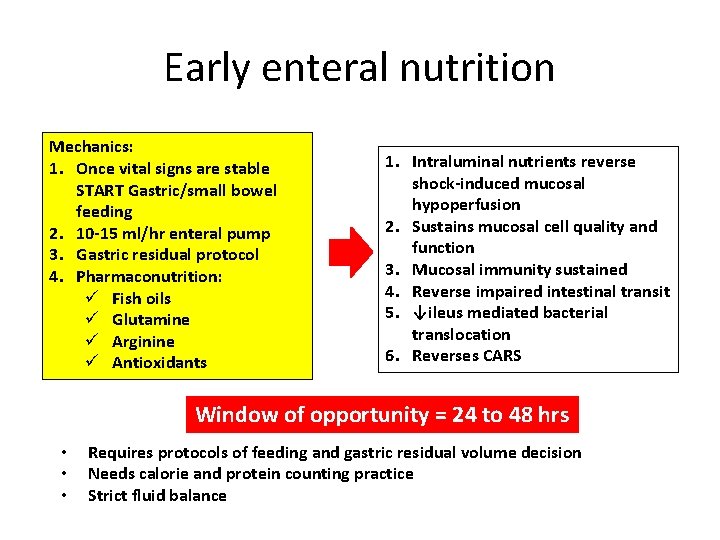
Early enteral nutrition Mechanics: 1. Once vital signs are stable START Gastric/small bowel feeding 2. 10 -15 ml/hr enteral pump 3. Gastric residual protocol 4. Pharmaconutrition: ü Fish oils ü Glutamine ü Arginine ü Antioxidants 1. Intraluminal nutrients reverse shock-induced mucosal hypoperfusion 2. Sustains mucosal cell quality and function 3. Mucosal immunity sustained 4. Reverse impaired intestinal transit 5. ↓ileus mediated bacterial translocation 6. Reverses CARS Window of opportunity = 24 to 48 hrs • • • Requires protocols of feeding and gastric residual volume decision Needs calorie and protein counting practice Strict fluid balance
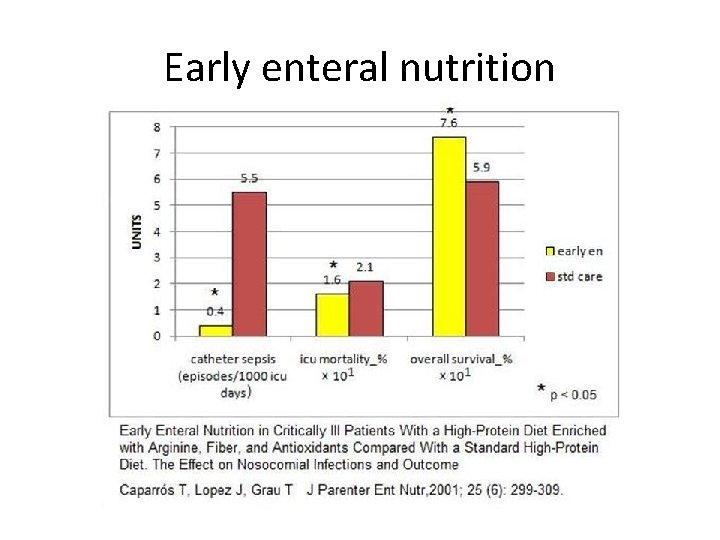
Early enteral nutrition
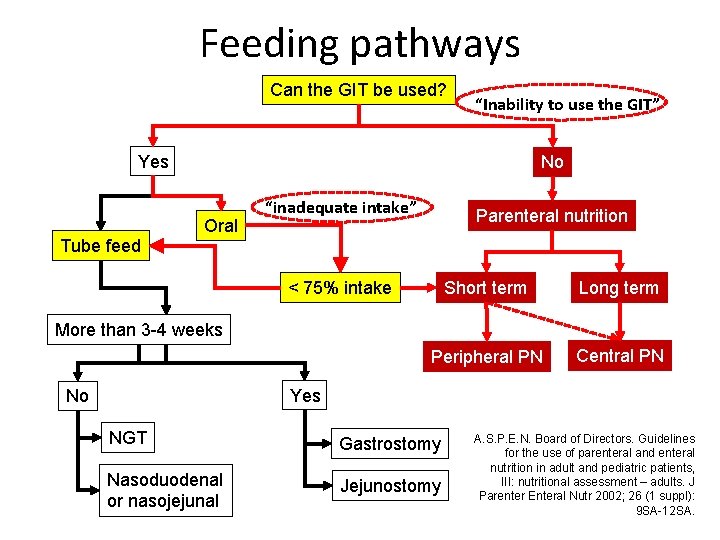
Feeding pathways Can the GIT be used? “Inability to use the GIT” Yes Tube feed No Oral “inadequate intake” Parenteral nutrition < 75% intake Short term Long term Peripheral PN Central PN More than 3 -4 weeks Yes No NGT Gastrostomy Nasoduodenal or nasojejunal Jejunostomy A. S. P. E. N. Board of Directors. Guidelines for the use of parenteral and enteral nutrition in adult and pediatric patients, III: nutritional assessment – adults. J Parenter Enteral Nutr 2002; 26 (1 suppl): 9 SA-12 SA.
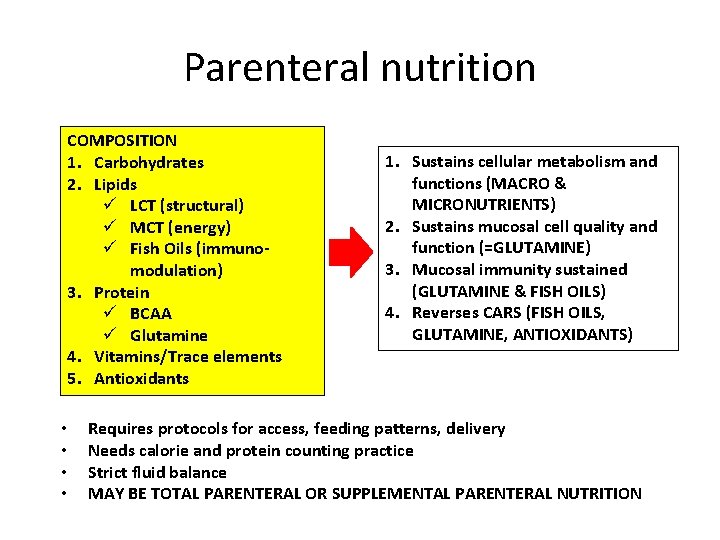
Parenteral nutrition COMPOSITION 1. Carbohydrates 2. Lipids ü LCT (structural) ü MCT (energy) ü Fish Oils (immunomodulation) 3. Protein ü BCAA ü Glutamine 4. Vitamins/Trace elements 5. Antioxidants • • 1. Sustains cellular metabolism and functions (MACRO & MICRONUTRIENTS) 2. Sustains mucosal cell quality and function (=GLUTAMINE) 3. Mucosal immunity sustained (GLUTAMINE & FISH OILS) 4. Reverses CARS (FISH OILS, GLUTAMINE, ANTIOXIDANTS) Requires protocols for access, feeding patterns, delivery Needs calorie and protein counting practice Strict fluid balance MAY BE TOTAL PARENTERAL OR SUPPLEMENTAL PARENTERAL NUTRITION
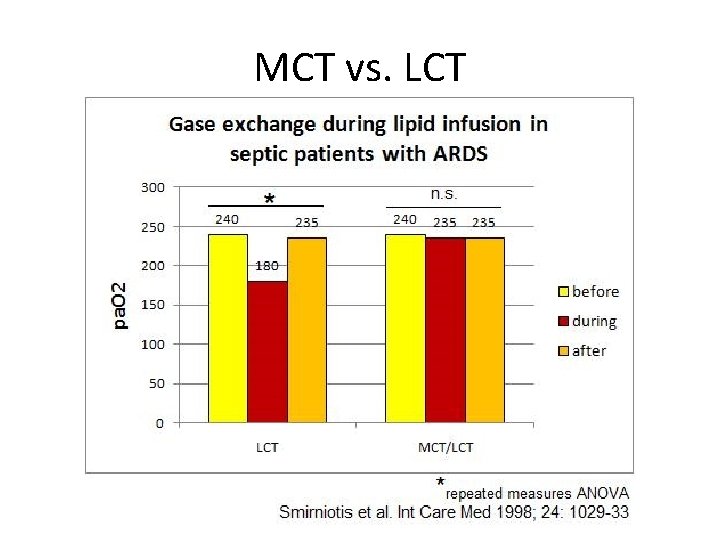
MCT vs. LCT

PHARMACONUTRITION
Diet and inflammation TRAUMA /
Diet and inflammation TRAUMA /

Immune modulating nutrients • Another name = pharmaconutrients – Fish oils – Glutamine – Antioxidants – Arginine Jones NE and Heyland DK. Pharmaconutrition: a new paradigm. Curr Opinion Gastroenterology 2008; 24: 215 -22) Pharmaconutrition: a new paradigm Curr Opinion Gastroenterology 2008; 24: 215 -22
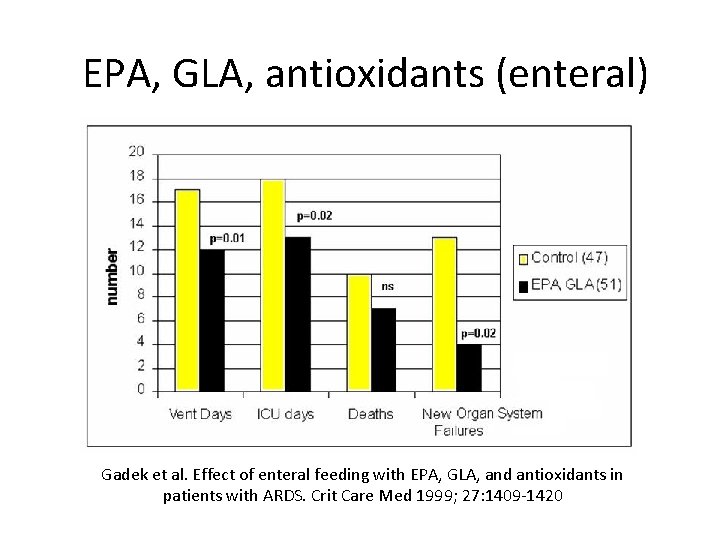
EPA, GLA, antioxidants (enteral) Gadek et al. Effect of enteral feeding with EPA, GLA, and antioxidants in patients with ARDS. Crit Care Med 1999; 27: 1409 -1420
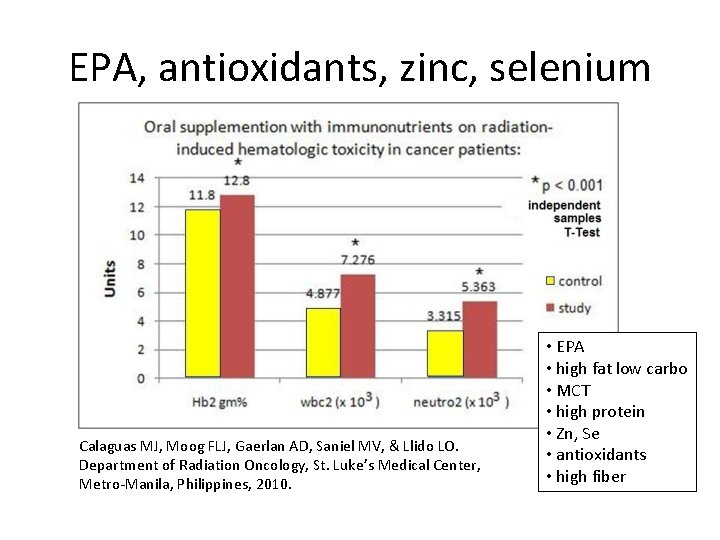
EPA, antioxidants, zinc, selenium Calaguas MJ, Moog FLJ, Gaerlan AD, Saniel MV, & Llido LO. Department of Radiation Oncology, St. Luke’s Medical Center, Metro-Manila, Philippines, 2010. • EPA • high fat low carbo • MCT • high protein • Zn, Se • antioxidants • high fiber
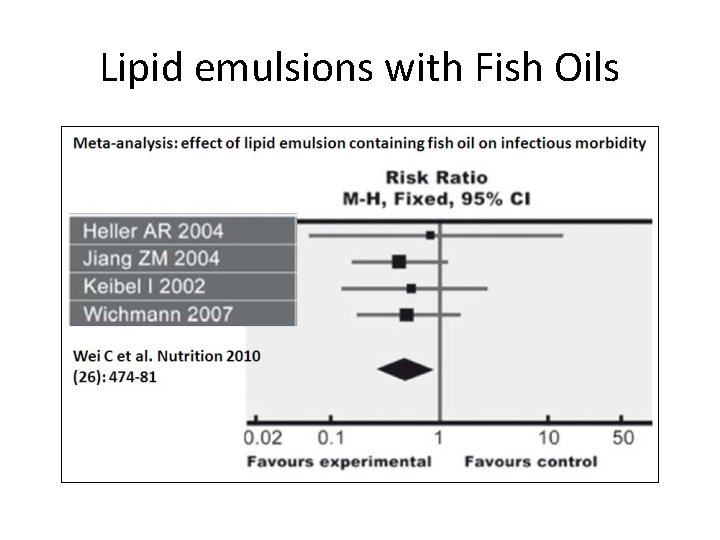
Lipid emulsions with Fish Oils
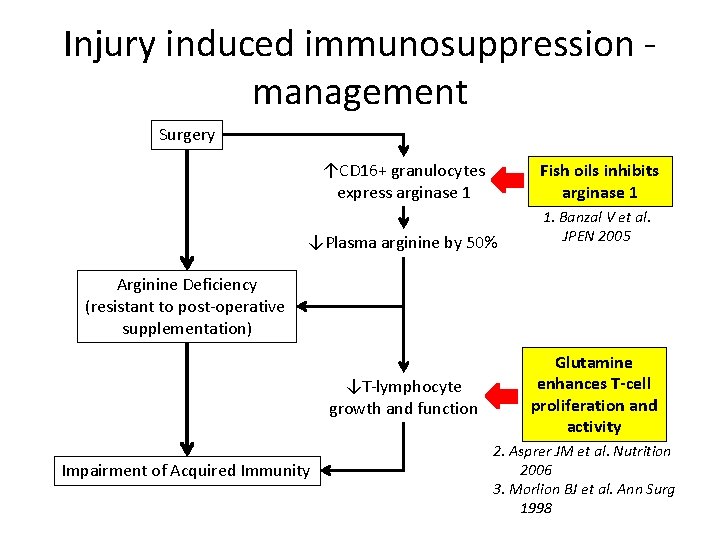
Injury induced immunosuppression management Surgery ↑CD 16+ granulocytes express arginase 1 Fish oils inhibits arginase 1 ↓Plasma arginine by 50% 1. Banzal V et al. JPEN 2005 Arginine Deficiency (resistant to post-operative supplementation) ↓T-lymphocyte growth and function Impairment of Acquired Immunity Glutamine enhances T-cell proliferation and activity 2. Asprer JM et al. Nutrition 2006 3. Morlion BJ et al. Ann Surg 1998
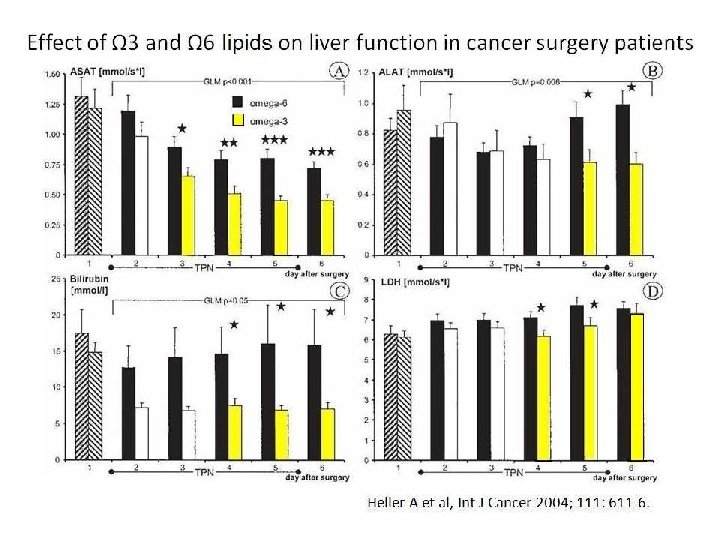
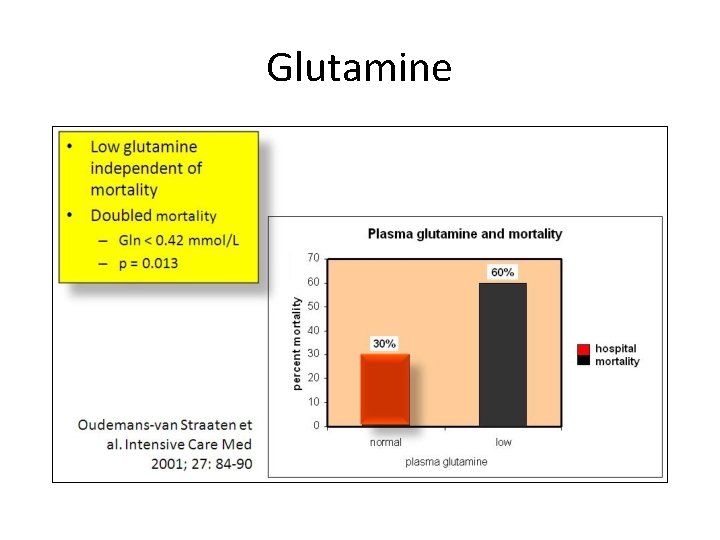
Glutamine
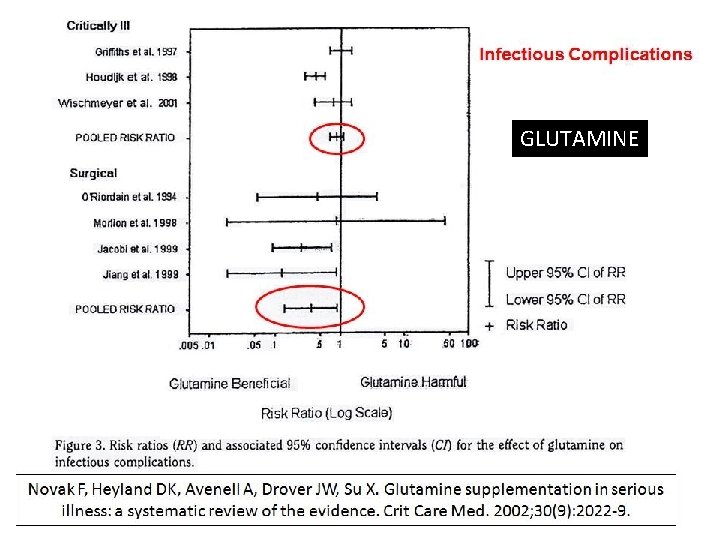
GLUTAMINE
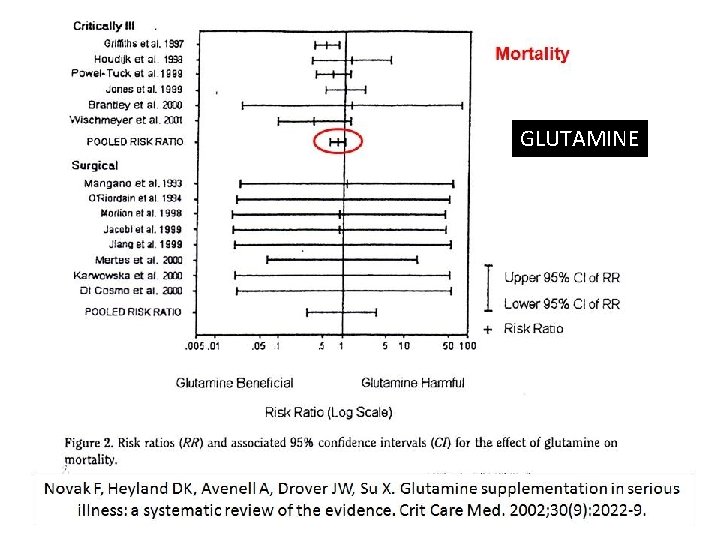
GLUTAMINE
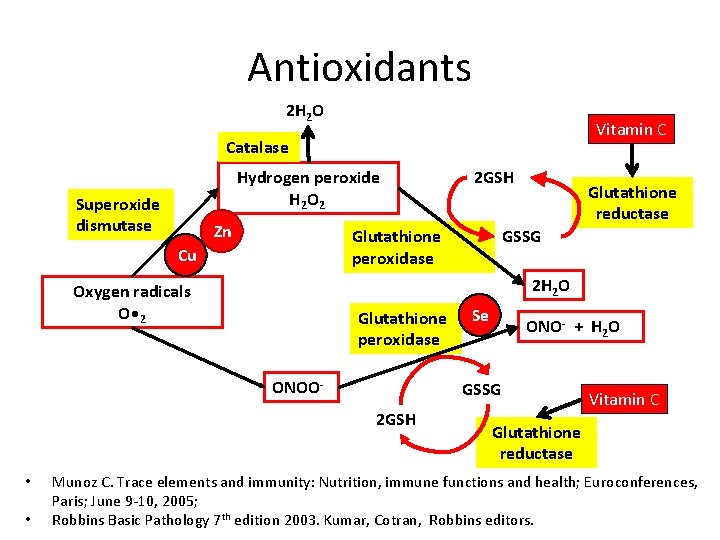
Antioxidants 2 H 2 O Vitamin C Catalase Hydrogen peroxide H 2 O 2 Superoxide dismutase Zn Glutathione peroxidase Cu Glutathione peroxidase ONOO- GSSG Se ONO- + H 2 O GSSG 2 GSH • Glutathione reductase 2 H 2 O Oxygen radicals O • 2 • 2 GSH Vitamin C Glutathione reductase Munoz C. Trace elements and immunity: Nutrition, immune functions and health; Euroconferences, Paris; June 9 -10, 2005; Robbins Basic Pathology 7 th edition 2003. Kumar, Cotran, Robbins editors.
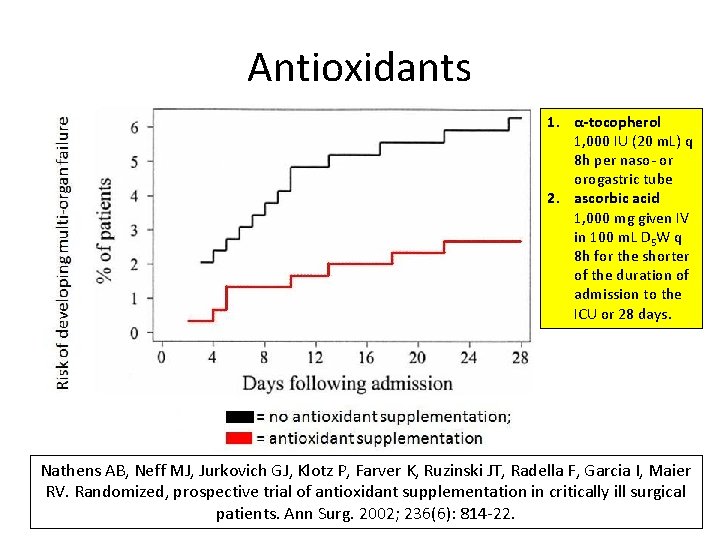
Antioxidants 1. α-tocopherol 1, 000 IU (20 m. L) q 8 h per naso- or orogastric tube 2. ascorbic acid 1, 000 mg given IV in 100 m. L D 5 W q 8 h for the shorter of the duration of admission to the ICU or 28 days. Nathens AB, Neff MJ, Jurkovich GJ, Klotz P, Farver K, Ruzinski JT, Radella F, Garcia I, Maier RV. Randomized, prospective trial of antioxidant supplementation in critically ill surgical patients. Ann Surg. 2002; 236(6): 814 -22.
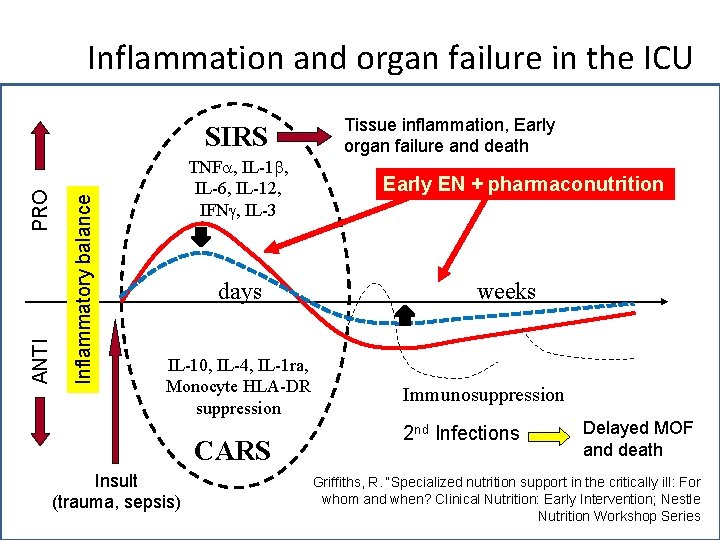
Inflammation and organ failure in the ICU Inflammatory balance ANTI PRO SIRS TNF , IL-1 , IL-6, IL-12, IFN , IL-3 days IL-10, IL-4, IL-1 ra, Monocyte HLA-DR suppression CARS Insult (trauma, sepsis) Tissue inflammation, Early organ failure and death Early EN + pharmaconutrition weeks Immunosuppression 2 nd Infections Delayed MOF and death Griffiths, R. “Specialized nutrition support in the critically ill: For whom and when? Clinical Nutrition: Early Intervention; Nestle Nutrition Workshop Series

ANTIBIOTICS AND PRO/PRE-BIOTICS

Guidelines • Prophylaxis: single pre-operative dose; one hour before incision • Patients on antibiotics: continue • Prolonged surgery: repeat antibiotics within 4 hours of procedure • Per 1500 ml of blood loss: give antibiotics • Type of antibiotics: broad spectrum recommended by infection committee EBM guidelines: ACS

Antibiotics and gut microflora • Antibiotics → alterations in gastrointestinal microbiota composition → ↑disease risk → by ↑susceptibility to gastrointestinal infections. – Antibiotic-associated diarrhea and colitis → Clostridium difficile or Clostridium perfringens (human study) – Increased susceptibility to invasive salmonellosis after streptomycin and vancomycin (animal study) – Antibiotic therapy for children infected with E. coli strain O 157: H 7 → ↑risk of hemolytic-uremic syndrome (human study) – Antimicrobial treatment for Helicobacter pylori induces marked disturbances in the intestinal microbiota. (human study) Preidis GA, Versalovic J. Targeting the Human Microbiome With Antibiotics, Probiotics, and Prebiotics: Gastroenterology Enters the Metagenomics Era. Gastroenterology 2009; 136: 2015– 2031
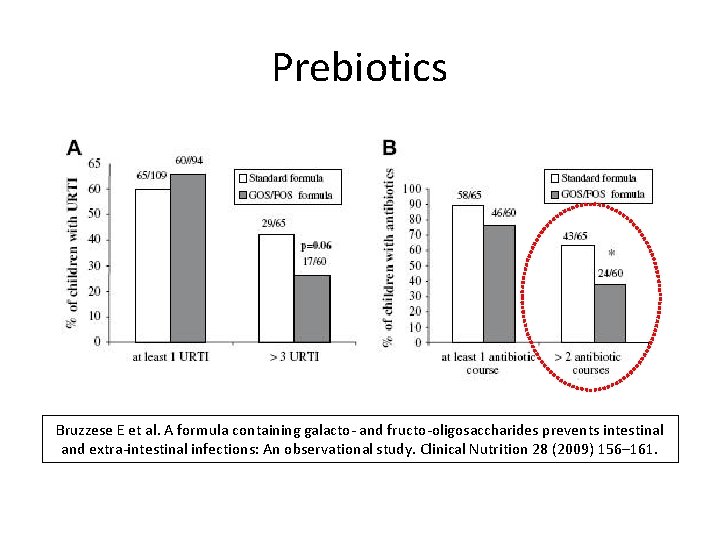
Prebiotics Bruzzese E et al. A formula containing galacto- and fructo-oligosaccharides prevents intestinal and extra-intestinal infections: An observational study. Clinical Nutrition 28 (2009) 156– 161.
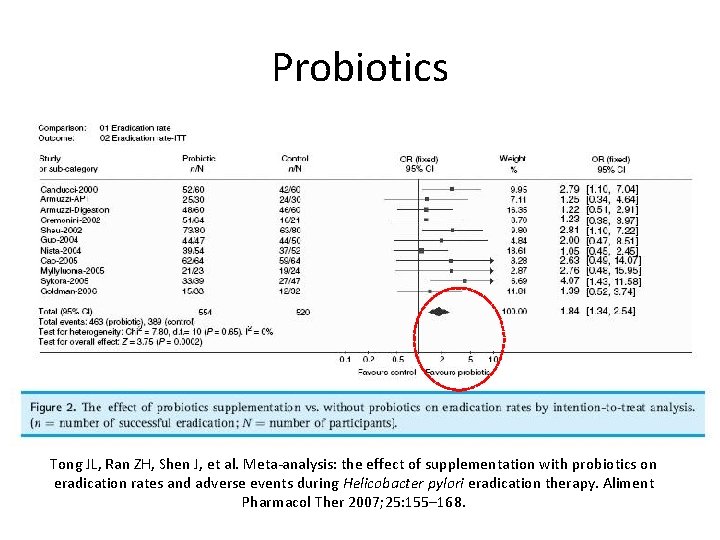
Probiotics Tong JL, Ran ZH, Shen J, et al. Meta-analysis: the effect of supplementation with probiotics on eradication rates and adverse events during Helicobacter pylori eradication therapy. Aliment Pharmacol Ther 2007; 25: 155– 168.

ADEQUATE INTAKE
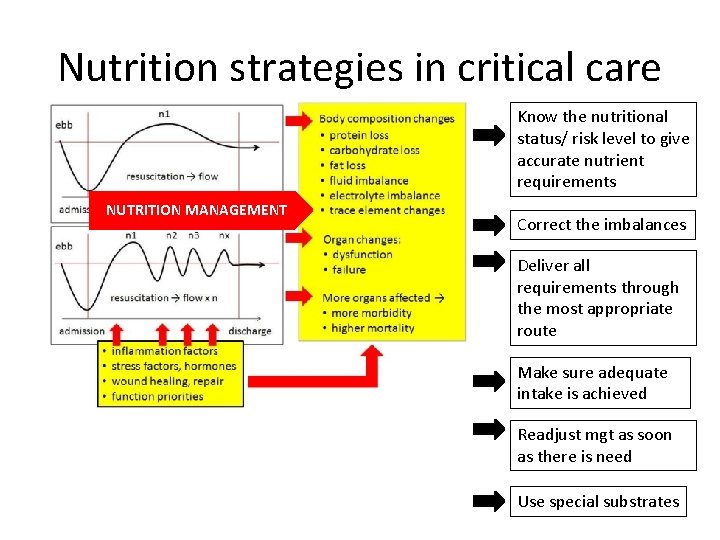
Nutrition strategies in critical care Know the nutritional status/ risk level to give accurate nutrient requirements NUTRITION MANAGEMENT Correct the imbalances Deliver all requirements through the most appropriate route Make sure adequate intake is achieved Readjust mgt as soon as there is need Use special substrates
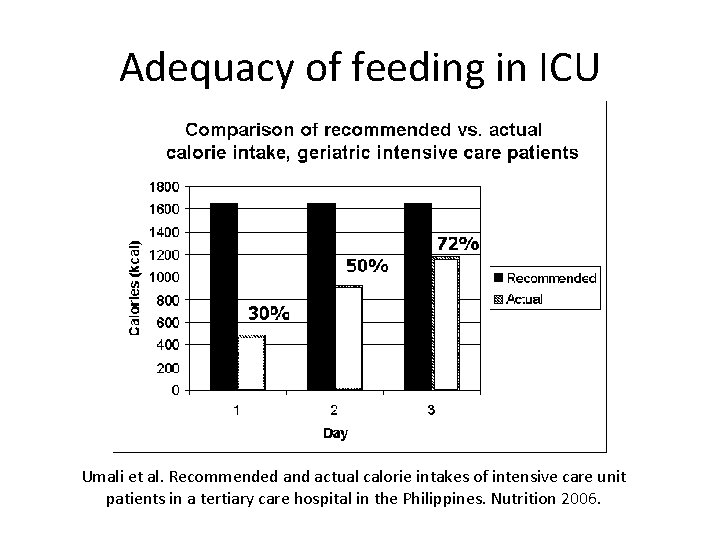
Adequacy of feeding in ICU Umali et al. Recommended and actual calorie intakes of intensive care unit patients in a tertiary care hospital in the Philippines. Nutrition 2006.
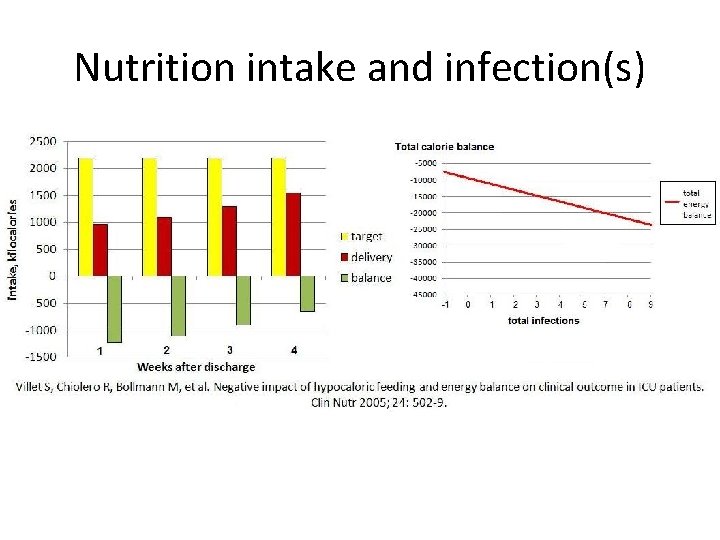
Nutrition intake and infection(s)
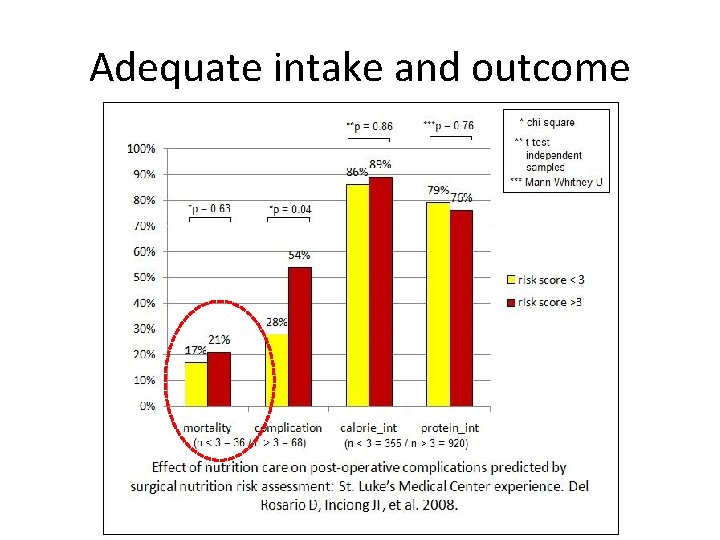
Adequate intake and outcome
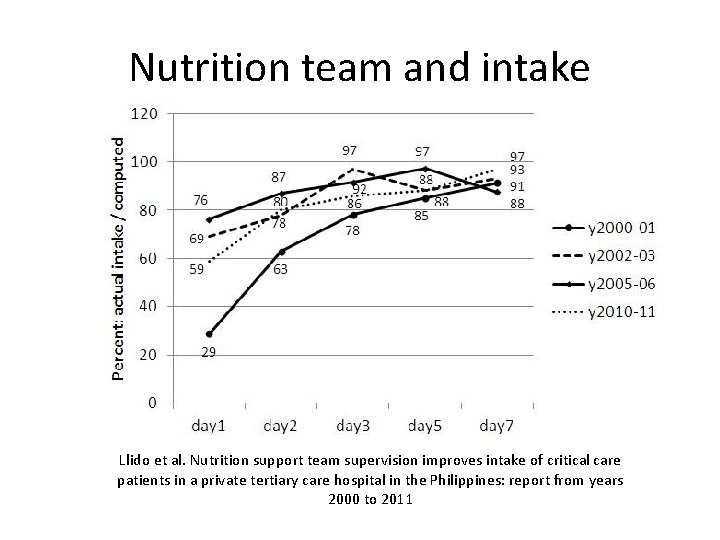
Nutrition team and intake Llido et al. Nutrition support team supervision improves intake of critical care patients in a private tertiary care hospital in the Philippines: report from years 2000 to 2011
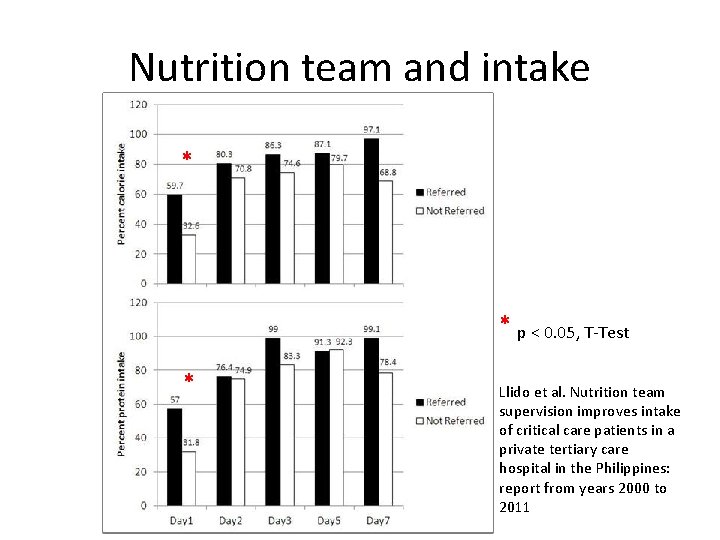
Nutrition team and intake * * p < 0. 05, T-Test * Llido et al. Nutrition team supervision improves intake of critical care patients in a private tertiary care hospital in the Philippines: report from years 2000 to 2011
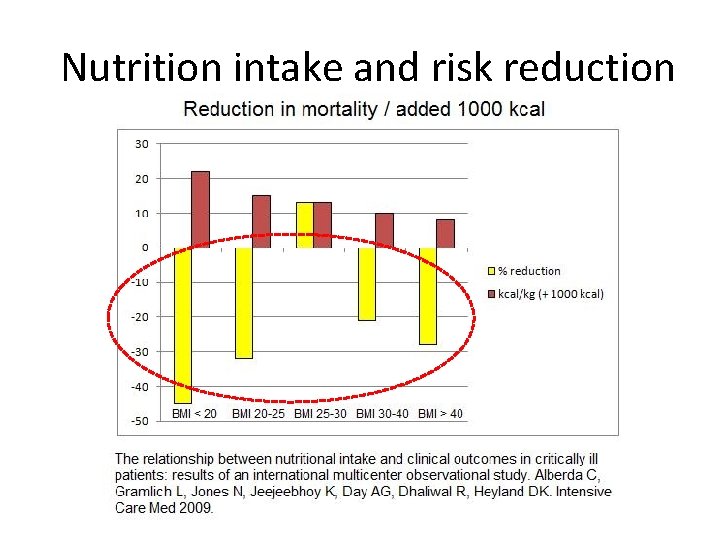
Nutrition intake and risk reduction

Refeeding syndrome • Severely malnourished, geriatric, low electrolyte values, artificial nutrition 1. Sodium and water retention → fluid overload, edema, heart failure 2. Hypophosphatemia → ventilatory failure, rhabdomyolysis 3. Hypokalemia → cardiac arrhythmia, ventilatory failure, rhabdomyolysis, ileus 4. Hypomagnesemia → cardiac arrhythmia, rhabdomyolysis 5. Vitamin deficits (thiamine) → encephalopathy, lactic acidosis E. Fiaccadori. Fluids and electrolytes. PN Workshop 2009, Kuala Lumpur, Malaysia

Targeted nutrition • Burns • Surgical site infections • Pressure ulcers

MANAGEMENT STRATEGIES

Surgical critical care • Critical illness following surgery or trauma • High risk surgical patients • Shock and hemodynamic compromise • Acute lung injury and ARDS following surgery, trauma, or pancreatitis • Sepsis and severeinfections • Trauma evaluation and management • Neurologic emergencies • Post-transplantation • Post-operative complications • Peritonitis, perforated viscus, and abdominal sepsis • Enterocutaneous fistulas • Gastrointestinal hemorrhage • Severe acute pancreatitis • Multisystem organ failure

Management strategies • Shock and hemodynamic compromise / Acute lung injury following surgery, trauma, pancreatitis – Resuscitation • Choose appropriate solutions – Stabilize microcirculation • Maintain oxygen delivery and perfusion • Renal support – Early feeding within 24 hrs – pharmaconutrition • Increasing daily vitamin, trace elements, glutamine, fish oils • Pre/probiotics with antibiotics – Strict glucose control

Management strategies • High risk surgical patients / critical illness following surgery or trauma – Severely malnourished – Immunosuppressed – Management: • • • Pre-operative build up Zero fluid balance Post-op early feeding Adequate intake + pharmaconutrition Targeted nutrition

Management strategies • Sepsis and severe infections/postoperative complications/peritonitis, ruptured viscus – Prevention: • • Pre-op - severely malnourished build up Intra-op: zero-fluid balance Post op: early enteral nutrition and zero-fluid balance Adequate nutrient delivery – Macronutrients – Daily vitamins and trace elements • Pharmaconutrition • Antibiotics, culture& sensitivity, pre/probiotics

Management strategies • Enterocutaneous fistulas – severely malnourished: build up – Post op: • High output: parenteral nutrition • Medium to low: combined enteral and parenteral nutrition – Adequate nutrient delivery • Macronutrients • Daily vitamins and trace elements – Pharmaconutrition – New substrates
- Slides: 86