Surface Tension Below surface forces act equal in

Surface Tension • • Below surface, forces act equal in all directions At surface, some forces are missing, pulls molecules down and together, like membrane exerting tension on the surface • Pressure increase is balanced by surface tension, s • surface tension = magnitude of tension/length • s = 0. 073 N/m (water @ 20 o. C) Interface water air Net force inward No net force

Surface Tension • Liquids have cohesion and adhesion, both involving molecular interactions – Cohesion: enables liquid to resist tensile stress – Adhesion: enables liquid to adhere to other bodies • Capillarity = property of exerting forces on fluids by fine tubes or porous media – – – due to cohesion and adhesion If adhesion > cohesion, liquid wets solid surfaces at rises If adhesion < cohesion, liquid surface depresses at pt of contact water rises in glass tube (angle = 0 o) mercury depresses in glass tube (angle = 130 -140 o)
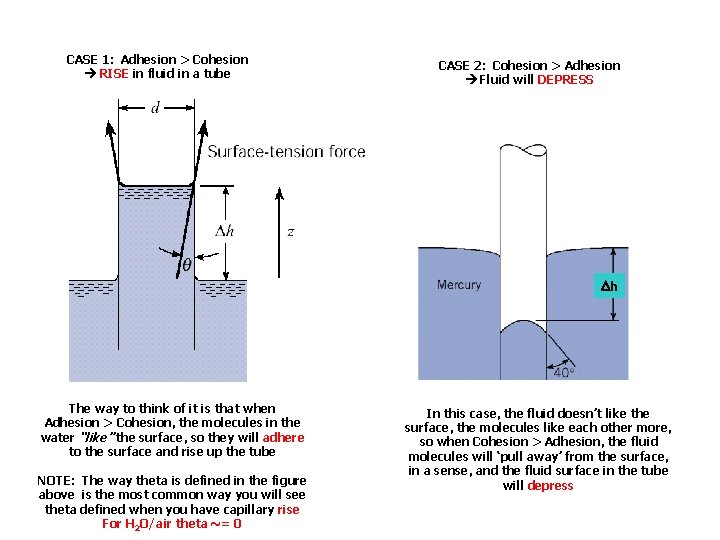
CASE 1: Adhesion > Cohesion RISE in fluid in a tube CASE 2: Cohesion > Adhesion Fluid will DEPRESS Dh The way to think of it is that when Adhesion > Cohesion, the molecules in the water “like” the surface, so they will adhere to the surface and rise up the tube NOTE: The way theta is defined in the figure above is the most common way you will see theta defined when you have capillary rise For H 2 O/air theta ~= 0 In this case, the fluid doesn’t like the surface, the molecules like each other more, so when Cohesion > Adhesion, the fluid molecules will ‘pull away’ from the surface, in a sense, and the fluid surface in the tube will depress
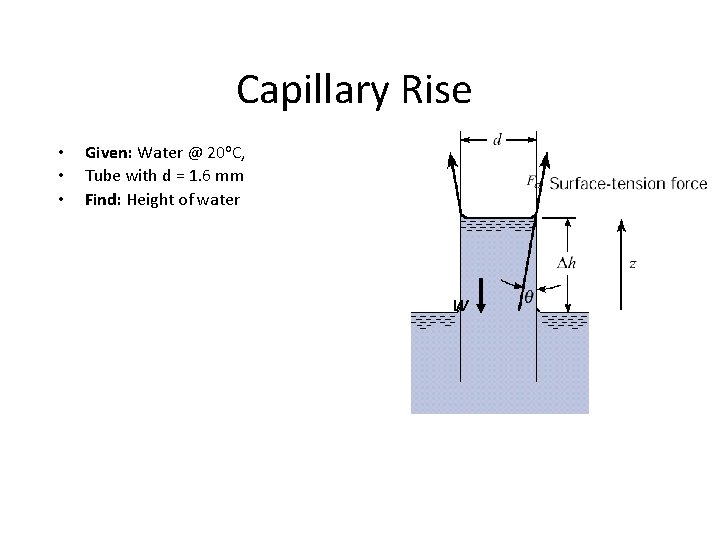
Capillary Rise • • • Given: Water @ 20 o. C, Tube with d = 1. 6 mm Find: Height of water W
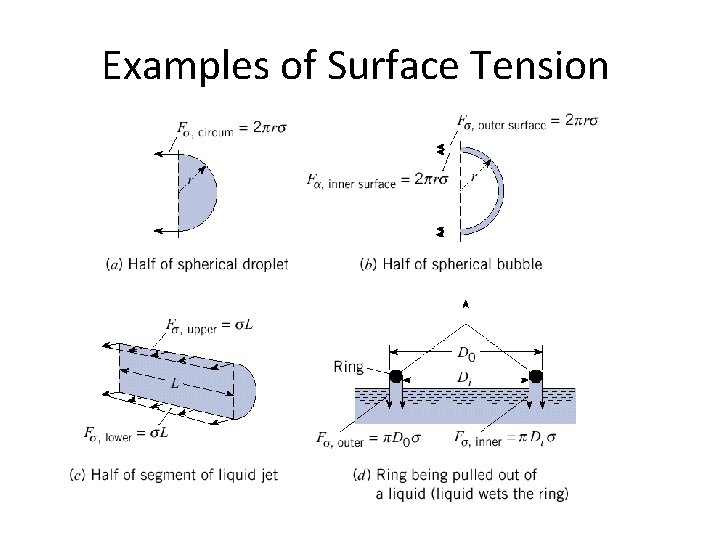
Examples of Surface Tension

Example: Textbook Problem 2. 49 • Find: The formula for the gage pressure within a spherical droplet of water?

Example: Textbook Problem 2. 50 • Given: Spherical bubble, inside radius r, film thickness t, and surface tension s. • Find: Formula for pressure in the bubble relative to that outside.

Elasticity (Compressibility) • If pressure acting on mass of fluid increases: fluid contracts • If pressure acting on mass of fluid decreases: fluid expands • Elasticity relates to amount of deformation for a given change in pressure Ev = bulk modulus of elasticity How does second part of equation come about? Small d. V/V = large modulus of elasticity

Vapor Pressure (Pvp) • Vapor pressure of a pure liquid = equilibrium partial pressure of the gas molecules of that species above a flat surface of the pure liquid – Concept on board – Very strong function of temperature (Pvp up as T up) – Very important parameter of liquids (highly variable – see attached page) • When vapor pressure exceeds total air pressure applied at surface, the liquid will boil. • Pressure at which a liquid will boil for a given temperature – At 10 o. C, vapor pressure of water = 0. 012 atm = 1200 Pa – If reduce pressure to this value can get boiling of water (can lead to “cavitation”) • If Pvp > 1 atm compound = gas • If Pvp < 1 atm compound = liquid or solid
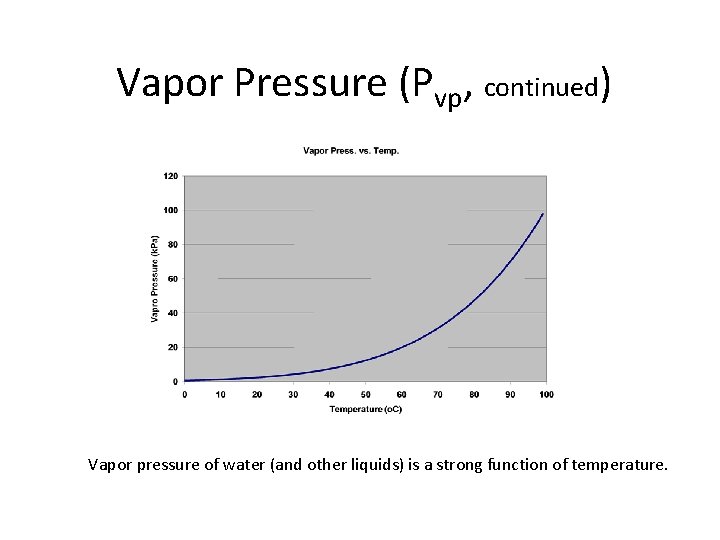
Vapor Pressure (Pvp, continued) Vapor pressure of water (and other liquids) is a strong function of temperature.
- Slides: 10