Supratherapeutic INR on warfarin Bleeding whatever INR Discontinue

Supratherapeutic INR on warfarin

Bleeding whatever INR • Discontinue warfarin. • ●Vitamin K – Administer 10 mg vitamin K by slow intravenous infusion (eg, over 20 to 60 minutes). For most patients receiving warfarin who have severe, obvious bleeding, vitamin K can be administered without waiting for laboratory tests or imaging studies because the risks associated with vitamin K are low, and if the patient requires reinitiation of anticoagulation while refractory to warfarin, another agent such as heparin can be used. Vitamin K may be repeated at 12 -hour intervals if the INR remains elevated • ●PCC – For patients with serious bleeding and INR >2, we suggest using a 4 -factor prothrombin complex concentrate (PCC, unactivated) rather than a 3 -factor PCC and/or Fresh Frozen Plasma (FFP) for rapid reversal, due to the similar efficacy and lower risk of adverse events with 4 -factor PCC. • PCC can be administered using a fixed dose, or using a regimen that calculates the dose based on the patient's weight and the INR at presentation; a typical dose for INR >6 is 50 units/kg [32]. • Monitoring – Recheck the prothrombin time (PT)/INR at approximately 30 minutes following PCC administration, and periodically thereafter, with the frequency determined by the severity of bleeding. Repeat dosing of PCC is generally not needed for individuals treated with a dose based on the patient's weight and INR; however, if bleeding is truly life-threatening and the INR remains elevated, a second dose may be reasonable. Consultation with an expert in anticoagulant management may be helpful. If a fixed dose regimen of 1000 or 2000 units was used and the INR remains elevated, a second dose may be required for full reversal. (See 'PCC products, efficacy, risks' below. ) • ●Other agents – Antifibrinolytic agents such as tranexamic acid or epsilon-aminocaproic acid may be used in some settings (eg, oral/mucosal bleeding). Desmopressin (DDAVP) may be used for platelet dysfunction. • Recombinant activated factor VII (r. FVIIa) generally is not used to treat warfarin-associated bleeding, because it does not supply the other vitamin K-dependent factors affected by warfarin, although there are reports of its use [33, 34]. Thromboembolic risks of r. FVIIa further have been reported. (See "Recombinant factor VIIa: Clinical uses, dosing, and adverse effects", section on 'Evidence on thromboembolic events'. ) • ●Transfusions • • Platelets – Platelet transfusion may be used if a patient is bleeding in the setting of thrombocytopenia. Some experts use a target platelet count of >50, 000/micro. L and others use a target platelet count of >100, 000/micro. L, especially for life-threatening bleeding. (See "Clinical and laboratory aspects of platelet transfusion therapy", section on 'Actively bleeding patient'. ) • • RBCs – Transfusions of red blood cells (RBCs) are based on hemoglobin levels and/or estimated blood loss for rapid ongoing bleeding. (See "Indications and hemoglobin thresholds for red blood cell transfusion in the adult", section on 'Acute bleeding'. ) • • FFP – Fresh Frozen Plasma (FFP) may be given if no PCC is available (initial dose 15 to 30 m. L/kg), or if the patient is receiving massive transfusions for severe ongoing bleeding (see "Massive blood transfusion"). Doses of FFP required to replace coagulation factors often involve multiple units of FFP, which must be thawed and may take hours to administer, with risks of volume overload and transfusion reactions.

TREATMENT OF SUPRATHERAPEUTIC INR WITHOUT BLEEDING • INR >10 without bleeding — If the INR is >10 and the patient does not have clinically significant bleeding, warfarin should be stopped and vitamin K can be administered as an oral dose of 2. 5 to 5 mg. • Administration of vitamin K should produce a substantial reduction in the INR within 24 to 48 hours. The INR should be monitored closely (eg, daily, every other day), and oral vitamin K may be repeated as necessary. • A reduced maintenance dose of warfarin generally should be restarted when the INR falls into therapeutic range. • Nonbleeding patients should not be given PCC or FFP solely to correct a supratherapeutic INR as these products have associated risks (eg, thrombosis, transfusion reactions) (see 'PCC products, efficacy, risks' below) and there is not clear evidence of benefit. For nonbleeding patients, there is no clear benefit to giving larger doses of vitamin K or to giving vitamin K by the intravenous route (unless the patient cannot take an oral medication). Patients who receive higher doses of vitamin K may have a prolonged period in which the INR cannot be raised, and intravenous administration is associated with a small risk of anaphylaxis, especially when given rapidly [39 -42].

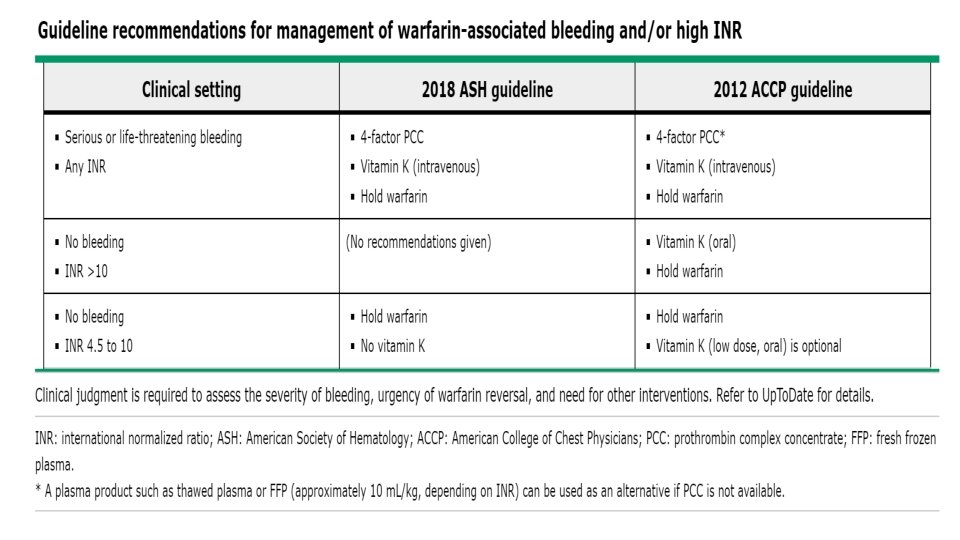
• INR 4. 5 to 10 without bleeding — If the INR is between 4. 5 and 10 and the patient does not have clinically • • • significant bleeding, warfarin is held temporarily (eg, one or two doses), with or without administration of a small dose of oral vitamin K (1 to 2. 5 mg) (table 1). As discussed below, evidence suggests that vitamin K does not reduce bleeding complications in this setting. The 2012 American College of Chest Physicians and the 2018 American Society of Hematology (ASH) guidelines both have a suggestion not to use vitamin K in this setting, although the 2012 ACCP guideline offers low dose oral vitamin K as an option [22, 31, 37]. (See 'ACCP and ASH guidelines' below. ) ●We generally favor the administration of a low dose of oral vitamin K for individuals with a greater risk of bleeding (eg, older, prior bleeding) and a lower risk of thromboembolism. We also have a low threshold for giving vitamin K to patients with conditions known to slow the spontaneous lowering of a supratherapeutic INR such as decompensated heart failure or active malignancy. These risk factors were identified in a retrospective record review of 633 patients with INR >6, in which the following predicted for a supratherapeutic INR two days after stopping warfarin [43]: • Older age (odds ratio [OR] 1. 2 per decade of life) • Higher index INR (OR 1. 25 per unit of elevation) • Lower warfarin maintenance dose (OR 0. 87 per 10 mg increase in total weekly dose) • Decompensated heart failure (OR 2. 79; 95% CI 1. 30 -5. 98) • Active cancer (OR 2. 48; 95% CI 1. 11 -5. 57) Other factors such as lack of rapid access to medical care may also contribute to the decision regarding vitamin K administration. Patients should be instructed to report and/or seek prompt medical attention for any signs of bleeding (eg, headache, melena, cardiovascular symptoms) or thromboembolism. It has been suggested that small oral doses of vitamin K may be given by administering the intravenous preparation (2 mg/m. L) orally, because pills may be difficult to cut ●We generally favor holding the vitamin K antagonist without giving vitamin K for individuals with a low risk of bleeding and those who may have a more rapid return of the INR to therapeutic range

• INR <4. 5 without bleeding — If the INR is above therapeutic range but <4. 5 and no clinically significant bleeding is apparent, the next dose of warfarin should be omitted and/or the maintenance dose of warfarin reduced slightly. • Often, the maintenance dose does not need to be reduced at all, especially if the INR elevation is minimal and/or expected to be transient. Additional therapies such as vitamin K are not indicated in this setting. Increased monitoring (eg, INR testing once or twice a week) during the period of dose adjustment is appropriate.

Vit K • Vitamin K dosing can be repeated at approximately 12 -hour intervals if needed based on the criteria outlined above (see 'Treatment of bleeding' above). A requirement for more than one or two days should raise the possibility of superwarfarin poisoning or impaired absorption of an oral preparation. (See 'Superwarfarin poisoning' above. ) • A meta-analysis of 10 randomized and 11 prospective trials evaluated the relative effectiveness of the various routes of vitamin K administration in patients with an INR of 4 to 10 without bleeding. At 24 hours after stopping warfarin, the percentages of patients with an INR in the range of 1. 8 to 4. 0 were as follows [74]: • ●Placebo – 20 percent (95% CI 0 -47) • ●Subcutaneous vitamin K – 31 percent (95% CI 7 -55) • ●Intravenous vitamin K – 77 percent (95% CI 60 -95) • ●Oral vitamin K – 82 percent (95% CI 70 -93) • The authors concluded that the subcutaneous route for vitamin K was no better for this purpose than placebo and should not be employed. This analysis was unable to determine the optimal dose of vitamin K; effective doses ranged from 1 to 2. 5 and 0. 5 to 3 mg for the oral and intravenous routes, respectively. Given the size of vitamin K tablets available in the United States (5 mg, scored to allow administration of 2. 5 mg), it has been suggested that a 1 mg oral dose is most reliably given by administering 1 mg of the intravenous preparation (2 mg/m. L) by mouth [ 44]. • Vitamin K represents a family of structurally similar compounds required for the synthesis of vitamin K-dependent clotting factors. Commercially available preparations include vitamin K 1 (phytonadione, the form produced by plants), K 2 (menaquinone, the form synthesized by bacteria and mitochondria, and the main storage form in humans), and other synthetic forms [ 75, 76]. Most of the clinical trials that have studied vitamin K administration for treatment of coagulopathy have used vitamin K 1 and this is the most commonly available formulation in the United States. Since both vitamin K 1 and K 2 are effective in reversing coagulopathy, we favor whichever formulation is most readily available. (See "Vitamin K and the synthesis and function of gamma-carboxyglutamic acid" and "Overview of vitamin K". )


• vitamin k • Vitamin K 2 is inactive. Vitamin K 1 is active. Warfarin inhibits the K 2 –> K 1 conversion by inhibiting vitamin K epoxide reductase. You already know that. • However, don’t confuse that enzyme with the one that vitamin K is a cofactor for, which is gamma glutamyl carboxylase. That enzyme name isn’t written in FA, although it’s more just implied, but I’ve seen it come up in practice questions, and they’ll throw the epoxide reductase in there as a distractor. • With regard to what I’ve encountered in practice questions, if there’s bleeding in a newborn, immediately think vitamin K deficiency or factor XIII deficiency. Neonates have sterile intestines, so they require vitamin K immediately after birth, so that deficiency is the most common, particularly if there is “bleeding from the umbilical stump. ” I had encountered factor XIII deficiency in a Kaplan QBook question. Apparently that’s fairly common too, and it presents like vitamin K deficiency in a neonate. • In contrast, factor XII, not XIII, deficiency results in pro-coagulation because Hageman factor (factor XII) converts prokallikrein to kallikrein, and kallikrein converts plasminogen to plasmin. So without factor XII, there’s pro-coagulation (or anti-coagulation). • I’ve also seen vitamin K as an answer choice to an easy question, but it was listed as phytonadione, so be aware that’s the medical name for vitamin K 1. It’s also called phylloquinone, but I’ve only seen the former show up so far, not this latter one. • And the other thing to know is that factor VII has the shortest half-life of any of the factors, so that’s why vitamin K deficiency prolongs PT, not a. PTT, first. • Last thing, I had remembered from MS 1 that parsley is the best known source of vitamin K, in addition to the cruciferous vegetables (which you can remember by saying “BBCC”), which include broccoli, Brussell sprouts, cauliflower and cabbage, however, in terms of practice questions, green leafy vegetables almost always points to folate, not vitamin K. The USMLE is overly obsessed with intestinal flora as being the vitamin K association.
- Slides: 9