Suprachoroidal CLSTA Plus Aflibercept Compared with Aflibercept Monotherapy


Suprachoroidal CLS-TA Plus Aflibercept Compared with Aflibercept Monotherapy for DME: Primary and Selected Secondary Results of the Randomized Phase 2 TYBEE Trial Michael S. Ip, MD, Muneeswar Gupta Nittala and Swetha Velaga on behalf of the TYBEE Study Group The Doheny Image Reading Center Doheny Eye Institute University of California - Los Angeles

Disclosures • Financial Disclosures • Consultant: Boehringer Ingelheim, Thrombo. Genics, Genentech, Astellas, Allergan, Novartis, Alimera, Allegro • Study Disclosures • This study includes research conducted on human subjects. Institutional Review Board approval was obtained prior to study initiation

Analyses of Phase 3 clinical trial data have indicated that even what seems to be persistent DME initially, may have good long-term results Ophthalmology Retina 2018 2, 558 -566 DOI: (10. 1016/j. oret. 2017. 10. 014) JAMA Ophthalmol. 2016; 134(3): 278 -285. 4
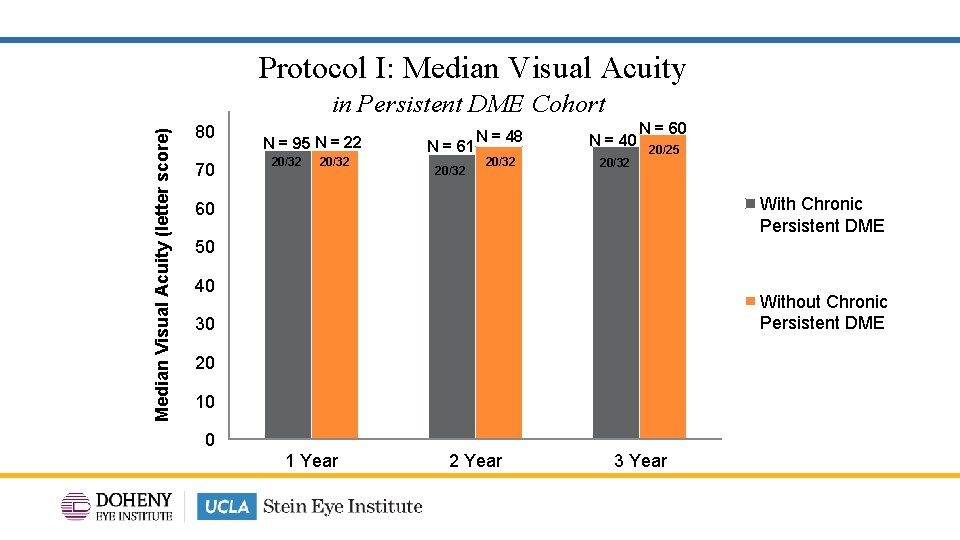
Protocol I: Median Visual Acuity (letter score) in Persistent DME Cohort 80 70 N = 95 N = 22 20/32 N = 61 20/32 N = 48 N = 40 20/32 N = 60 20/25 With Chronic Persistent DME 60 50 40 Without Chronic Persistent DME 30 20 10 0 1 Year 2 Year 3 Year
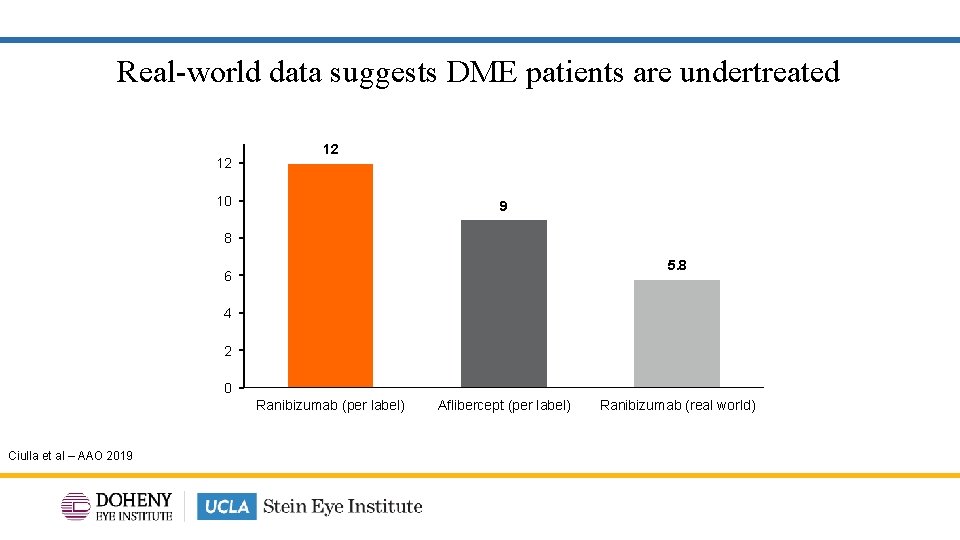
Real-world data suggests DME patients are undertreated 12 12 10 9 8 5. 8 6 4 2 0 Ranibizumab (per label) Aflibercept (per label) Ranibizumab (real world) Ciulla et al – AAO 2019 6
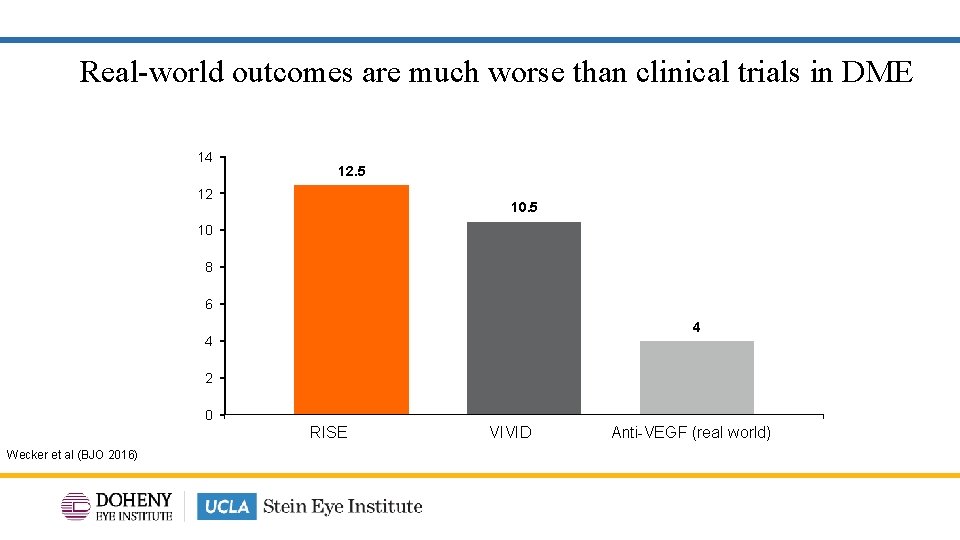
Real-world outcomes are much worse than clinical trials in DME 14 12 12. 5 10 8 6 4 4 2 0 RISE VIVID Anti-VEGF (real world) Wecker et al (BJO 2016) 7
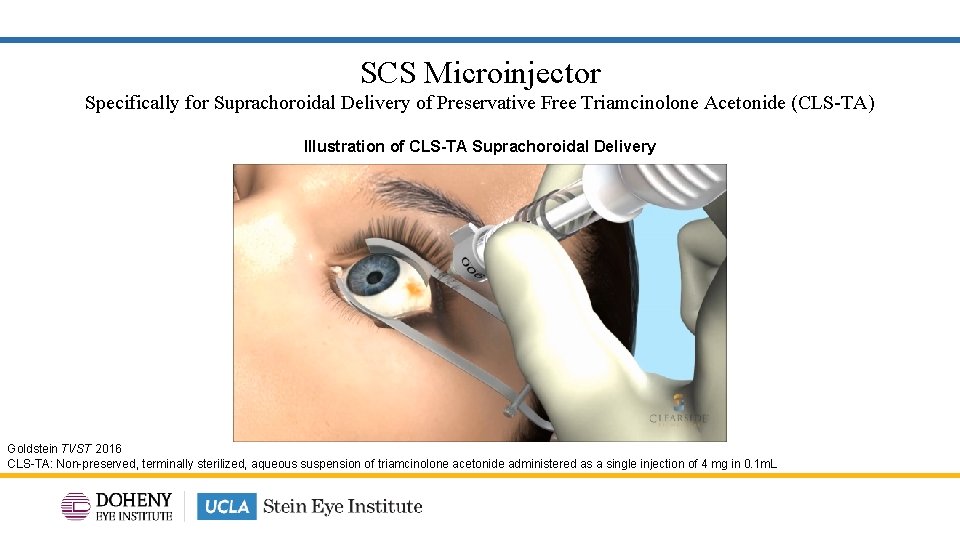
SCS Microinjector Specifically for Suprachoroidal Delivery of Preservative Free Triamcinolone Acetonide (CLS-TA) Illustration of CLS-TA Suprachoroidal Delivery Goldstein TVST 2016 CLS-TA: Non-preserved, terminally sterilized, aqueous suspension of triamcinolone acetonide administered as a single injection of 4 mg in 0. 1 m. L
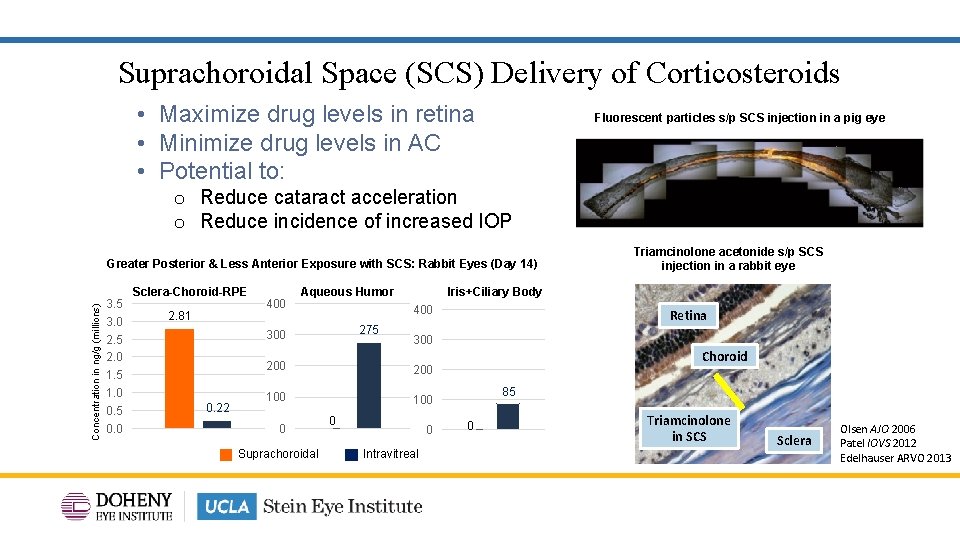
Suprachoroidal Space (SCS) Delivery of Corticosteroids • Maximize drug levels in retina • Minimize drug levels in AC • Potential to: Fluorescent particles s/p SCS injection in a pig eye o Reduce cataract acceleration o Reduce incidence of increased IOP Concentration in ng/g (millions) Greater Posterior & Less Anterior Exposure with SCS: Rabbit Eyes (Day 14) 3. 5 3. 0 2. 5 2. 0 1. 5 1. 0 0. 5 0. 0 Sclera-Choroid-RPE 2. 81 400 0. 22 Iris+Ciliary Body Aqueous Humor 400 275 300 200 100 Suprachoroidal Retina 300 200 0 Triamcinolone acetonide s/p SCS injection in a rabbit eye 00 0 Intravitreal Choroid 85 00 Triamcinolone in SCS Sclera Olsen AJO 2006 Patel IOVS 2012 Edelhauser ARVO 2013
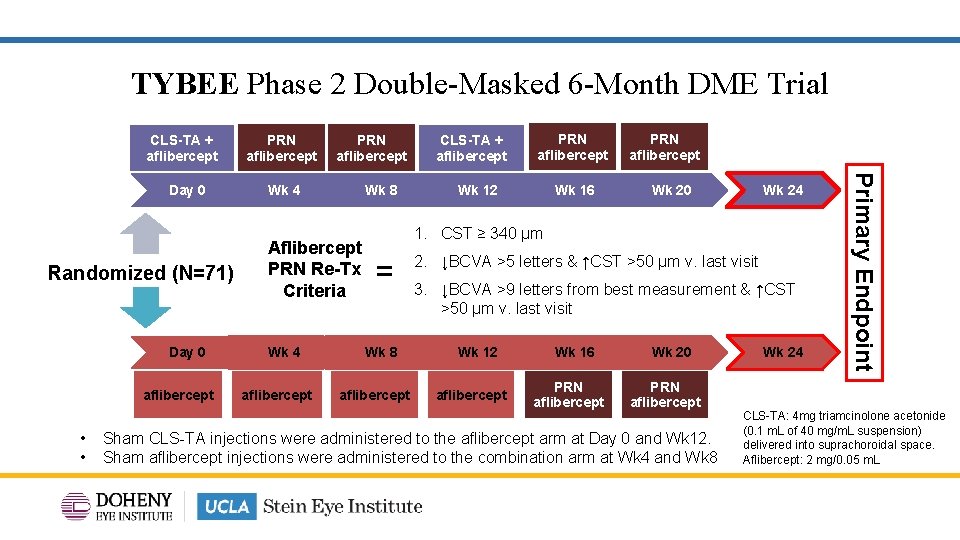
TYBEE Phase 2 Double-Masked 6 -Month DME Trial CLS-TA + aflibercept Day 0 Randomized (N=71) (N=35) Day 0 aflibercept • • PRN aflibercept Wk 4 Wk 8 Aflibercept PRN Re-Tx Criteria Wk 4 aflibercept CLS-TA + aflibercept PRN aflibercept Wk 12 Wk 16 PRN aflibercept Wk 20 Wk 24 1. CST ≥ 340 µm = Wk 8 aflibercept 2. ↓BCVA >5 letters & ↑CST >50 µm v. last visit 3. ↓BCVA >9 letters from best measurement & ↑CST >50 µm v. last visit Wk 12 aflibercept Wk 16 PRN aflibercept Wk 20 PRN aflibercept Sham CLS-TA injections were administered to the aflibercept arm at Day 0 and Wk 12. Sham aflibercept injections were administered to the combination arm at Wk 4 and Wk 8 Wk 24 Primary Endpoint (N=36) PRN aflibercept CLS-TA: 4 mg triamcinolone acetonide (0. 1 m. L of 40 mg/m. L suspension) delivered into suprachoroidal space. Aflibercept: 2 mg/0. 05 m. L
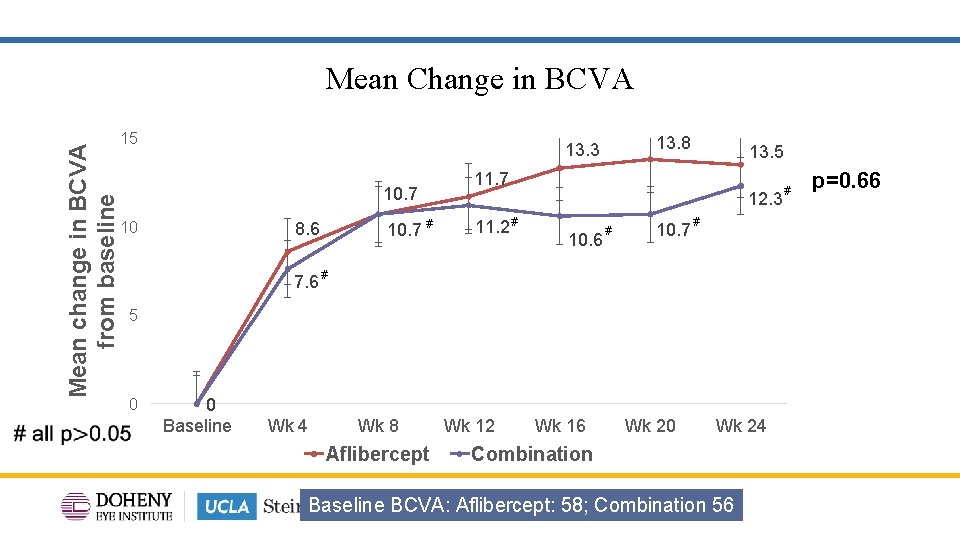
Mean change in BCVA from baseline Mean Change in BCVA 15 13. 8 13. 3 10. 7 10 10. 7 # 8. 6 7. 6 # 13. 5 11. 7 12. 3 11. 2 # 10. 6 # # # 10. 7 # p=0. 66 # # 5 0 0 Baseline Wk 4 Wk 8 Aflibercept Wk 12 Wk 16 Wk 20 Wk 24 Combination Baseline BCVA: Aflibercept: 58; Combination 56 11
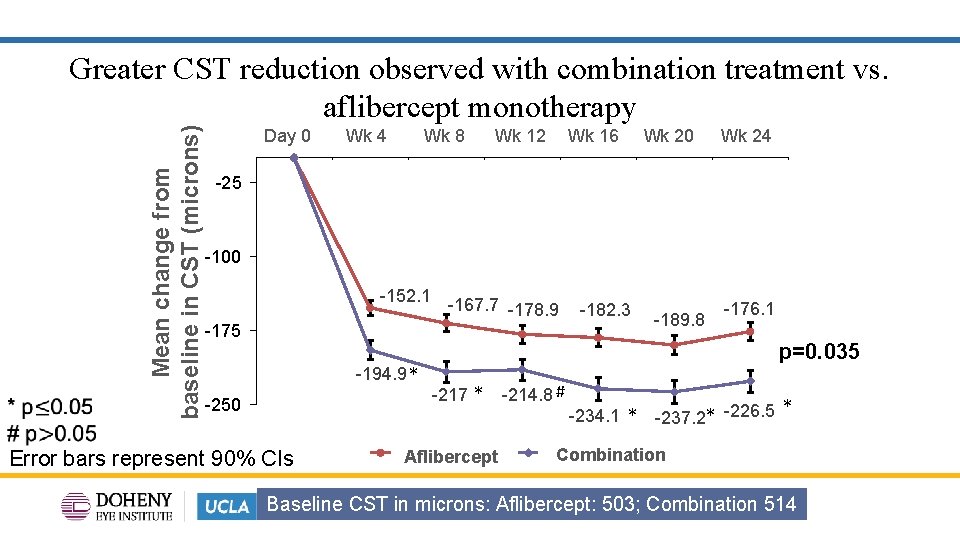
Mean change from baseline in CST (microns) Greater CST reduction observed with combination treatment vs. aflibercept monotherapy Day 0 -25 1 Wk 4 2 Wk 8 Wk 12 3 4 Wk 16 Wk 20 5 6 Wk 24 7 -100 -152. 1 -167. 7 -178. 9 -182. 3 -175 -194. 9 * -250 Error bars represent 90% CIs -189. 8 -176. 1 p=0. 035 -217 * Aflibercept Control -214. 8 # -234. 1 * -237. 2* -226. 5 * Combination Baseline CST in microns: Aflibercept: 503; Combination 514 12
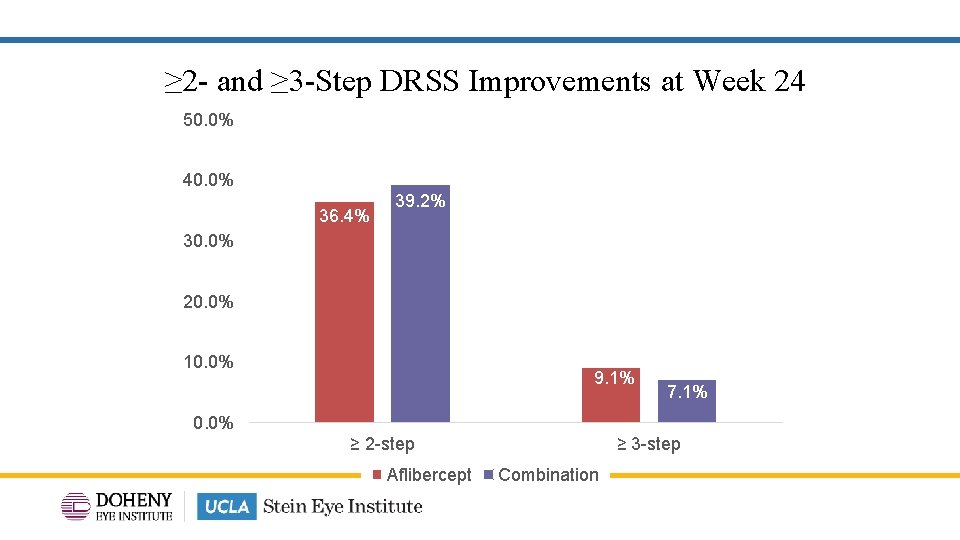
≥ 2 - and ≥ 3 -Step DRSS Improvements at Week 24 50. 0% 40. 0% 36. 4% 39. 2% 30. 0% 20. 0% 10. 0% 9. 1% 7. 1% 0. 0% ≥ 2 -step Aflibercept ≥ 3 -step Combination 13
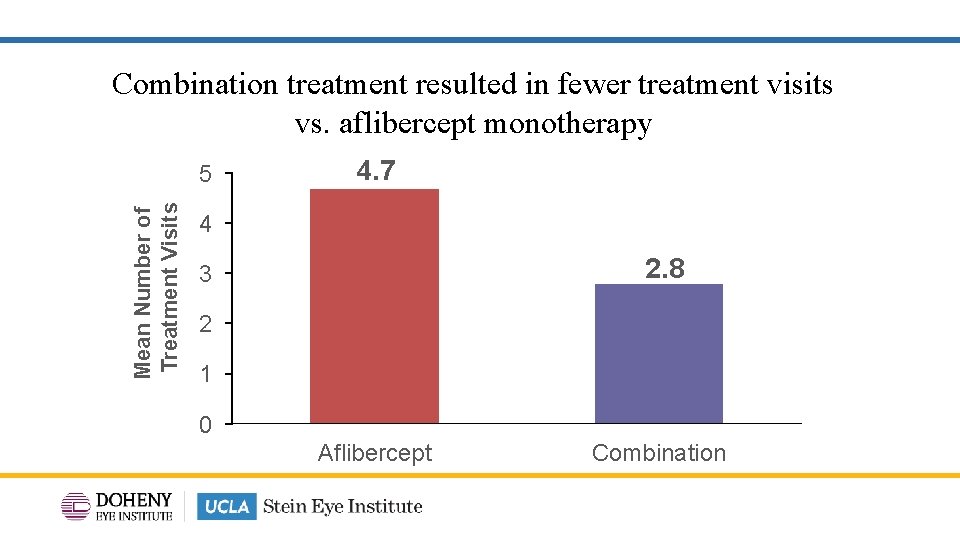
Combination treatment resulted in fewer treatment visits vs. aflibercept monotherapy Mean Number of Treatment Visits 5 4. 7 4 2. 8 3 2 1 0 Aflibercept Combination 14
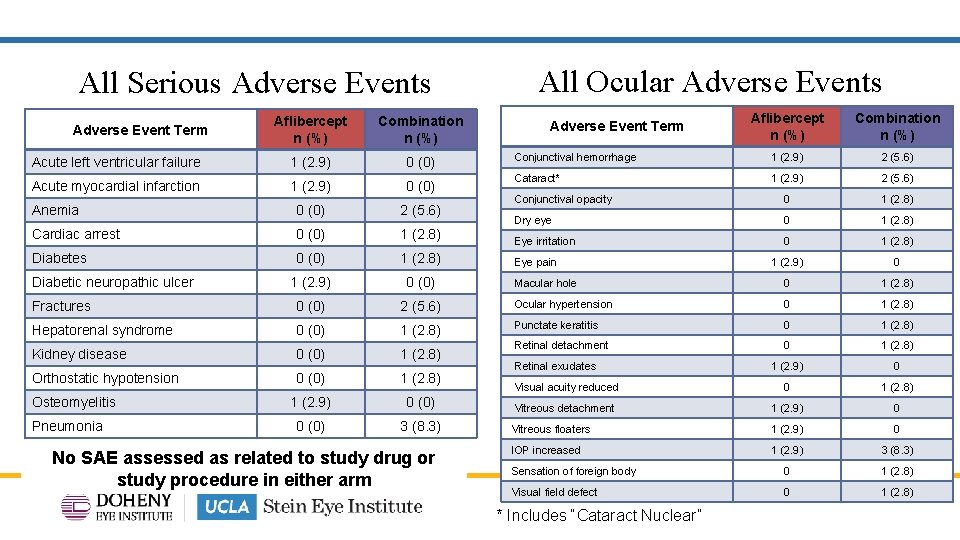
All Serious Adverse Events All Ocular Adverse Events Aflibercept n (%) Combination n (%) Conjunctival hemorrhage 1 (2. 9) 2 (5. 6) Cataract* 1 (2. 9) 2 (5. 6) Conjunctival opacity 0 1 (2. 8) Dry eye 0 1 (2. 8) Eye irritation 0 1 (2. 8) 1 (2. 9) 0 Macular hole 0 1 (2. 8) 2 (5. 6) Ocular hypertension 0 1 (2. 8) 0 (0) 1 (2. 8) Punctate keratitis 0 1 (2. 8) Kidney disease 0 (0) 1 (2. 8) Retinal detachment 0 1 (2. 8) Orthostatic hypotension 0 (0) 1 (2. 8) 1 (2. 9) 0 Visual acuity reduced 0 1 (2. 8) 1 (2. 9) 0 (0) Vitreous detachment 1 (2. 9) 0 0 (0) 3 (8. 3) Vitreous floaters 1 (2. 9) 0 IOP increased 1 (2. 9) 3 (8. 3) Sensation of foreign body 0 1 (2. 8) Visual field defect 0 1 (2. 8) Aflibercept n (%) Combination n (%) Acute left ventricular failure 1 (2. 9) 0 (0) Acute myocardial infarction 1 (2. 9) 0 (0) Anemia 0 (0) 2 (5. 6) Cardiac arrest 0 (0) 1 (2. 8) Diabetes 0 (0) 1 (2. 8) 1 (2. 9) 0 (0) Fractures 0 (0) Hepatorenal syndrome Adverse Event Term Diabetic neuropathic ulcer Osteomyelitis Pneumonia No SAE assessed as related to study drug or study procedure in either arm Adverse Event Term Eye pain Retinal exudates * Includes “Cataract Nuclear”

Analysis: Disorganization of the Inner Retinal layers (DRIL) Disorganization of the inner retinal layers was defined as the horizontal extent (μm) for which 1 or more boundaries between the inner retinal layers (ganglion cell layer and inner plexiform layer complex, inner nuclear layer, and outer plexiform layer) were not separately identifiable
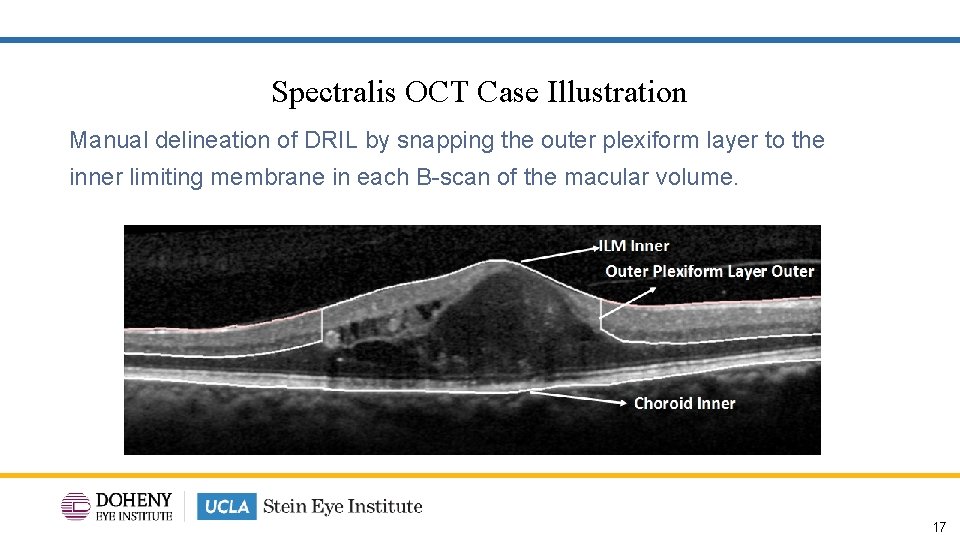
Spectralis OCT Case Illustration Manual delineation of DRIL by snapping the outer plexiform layer to the inner limiting membrane in each B-scan of the macular volume. 17
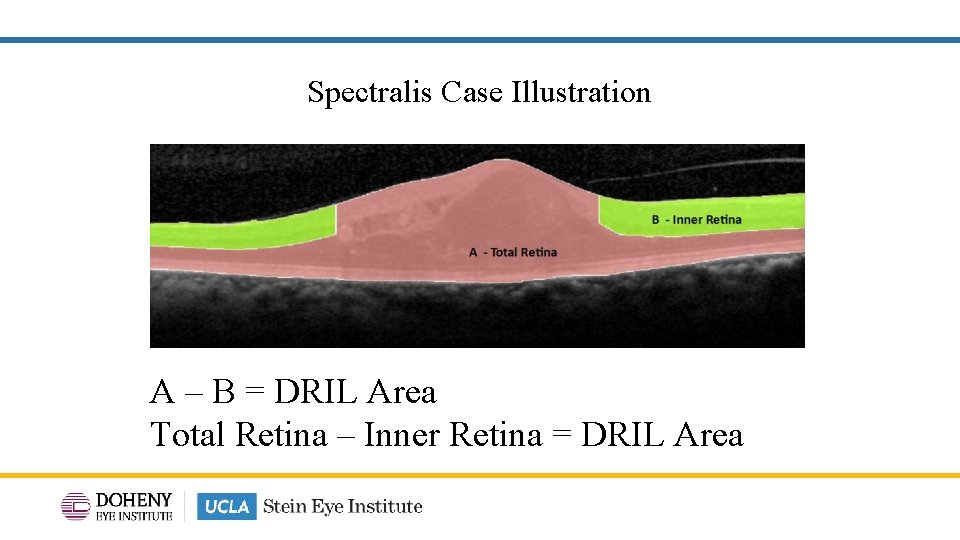
Spectralis Case Illustration A – B = DRIL Area Total Retina – Inner Retina = DRIL Area
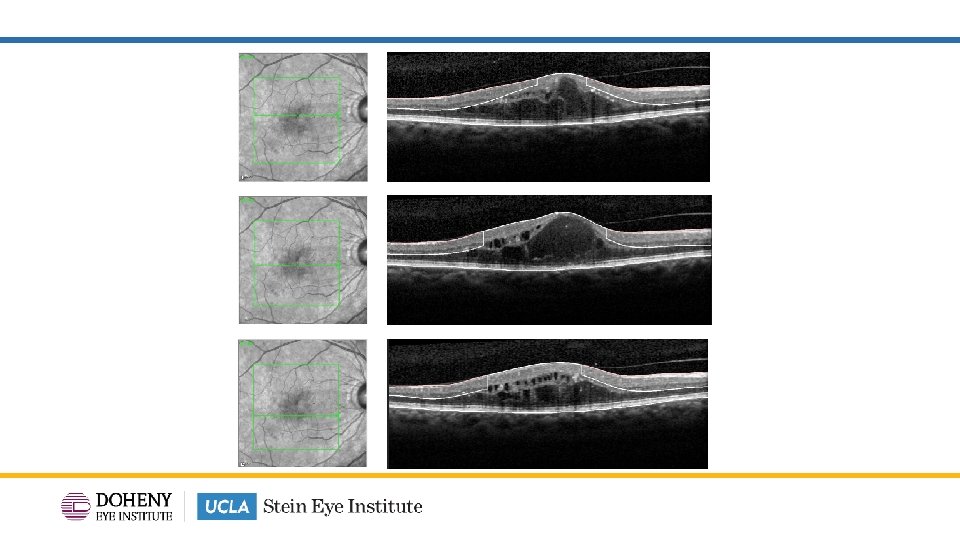

DRIL area map from volume scans 20
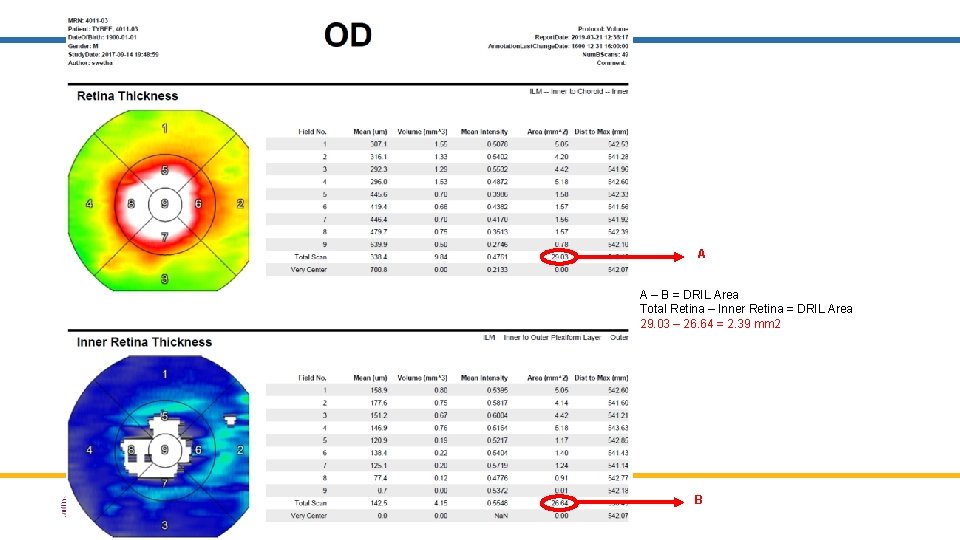
A A – B = DRIL Area Total Retina – Inner Retina = DRIL Area 29. 03 – 26. 64 = 2. 39 mm 2 B
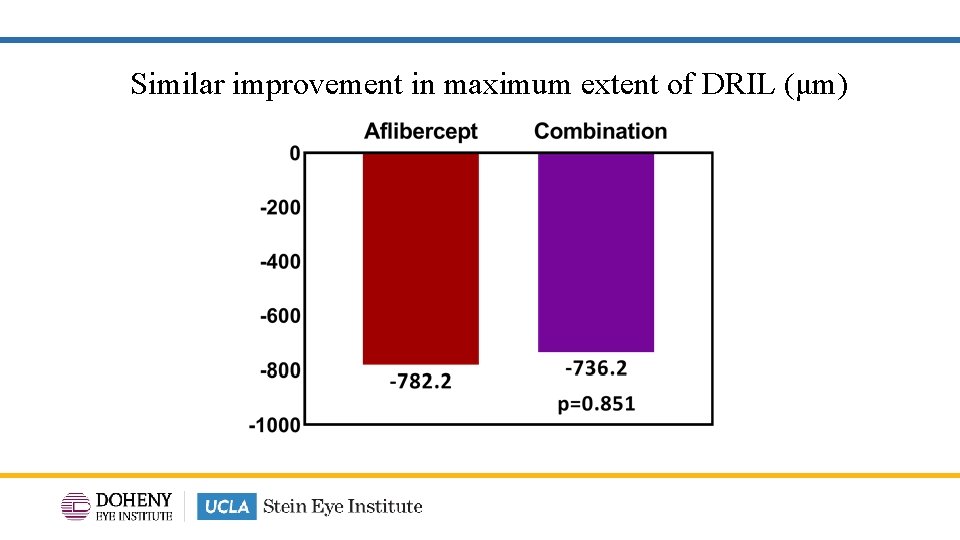
Similar improvement in maximum extent of DRIL (μm)
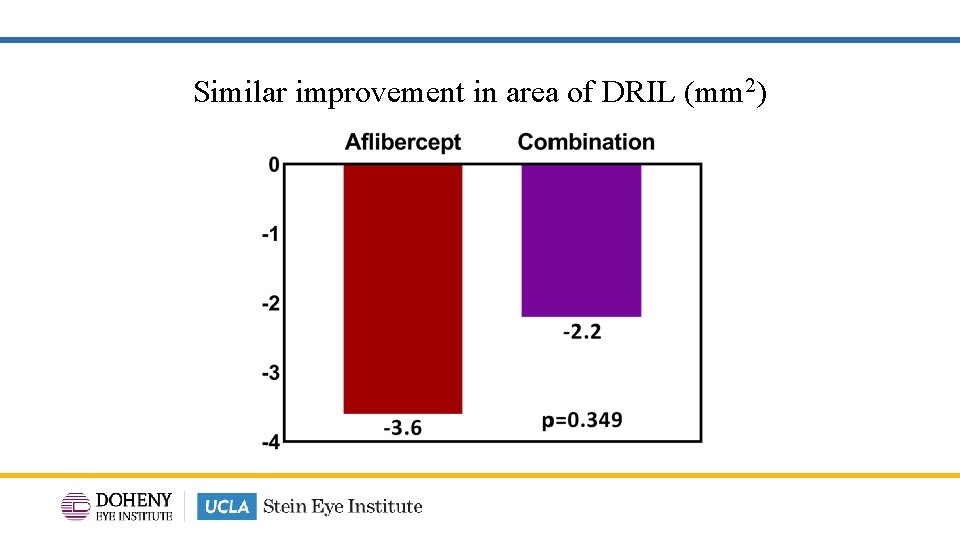
Similar improvement in area of DRIL (mm 2)

TYBEE: Conclusion • Similar BCVA improvements with combination aflibercept & suprachoroidal CLS -TA treatment vs aflibercept monotherapy. • CST improvement was significantly greater with combination treatment vs aflibercept monotherapy. • Other anatomic outcomes such as DRSS and changes in DRIL (maximum extent and area) were similar when comparing combination aflibercept & suprachoroidal CLS-TA treatment vs aflibercept monotherapy. • Fewer treatments in the combination arm compared to aflibercept monotherapy: 4. 7 vs 2. 8 mean treatment visits. 24

- Slides: 25