SUPPOSITORIES AND PESSARIES Presented By Ms Jyoti Kadam

SUPPOSITORIES AND PESSARIES Presented By: Ms. Jyoti Kadam

DEFINITION A suppository is a medicated solid dosage form generally intended for use in the rectum, vagina and to a lesser extent, the urethra. After insertion they melt or soften at body temperature, whereas vaginal suppositories sometimes called as pessaries, are also made as compressed tablets that disintegrate in body fluids.
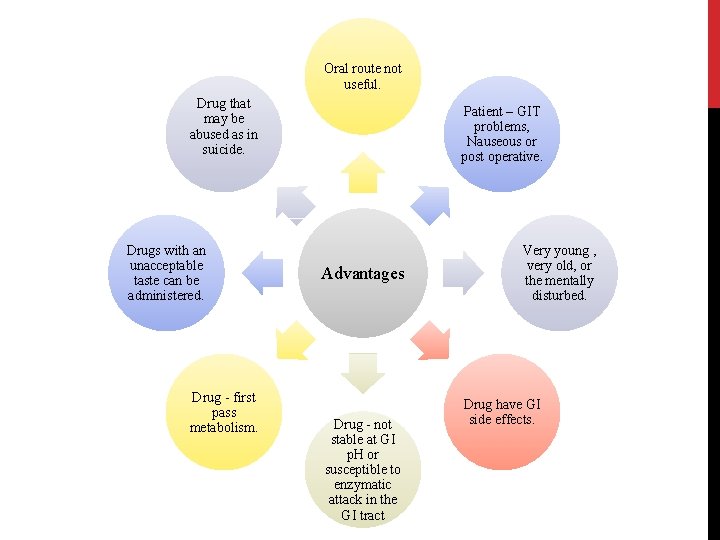
Oral route not useful. Drug that may be abused as in suicide. Drugs with an unacceptable taste can be administered. Drug - first pass metabolism. Patient – GIT problems, Nauseous or post operative. Advantages Drug - not stable at GI p. H or susceptible to enzymatic attack in the GI tract Very young , very old, or the mentally disturbed. Drug have GI side effects.
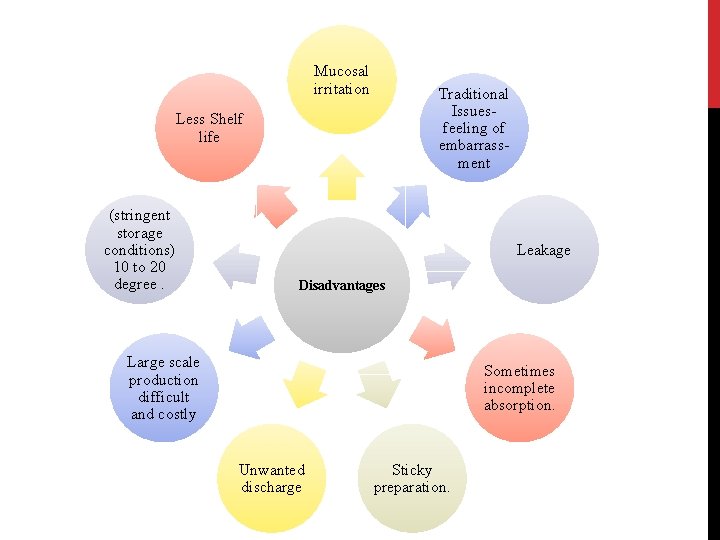
Mucosal irritation Traditional Issuesfeeling of embarrassment Less Shelf life (stringent storage conditions) 10 to 20 degree. Leakage Disadvantages Large scale production difficult and costly Sometimes incomplete absorption. Unwanted discharge Sticky preparation.

TYPES OF SUPPOSITORIES Rectal Vaginal (pessaries) • Cone or torpedo shaped weighing 1 – 2 g • Globular, oviform, cone shaped weighing 2 – 8 g • Made from glycero-gelatin or macrogol base Urethral bougies • Thin pencil shaped pointed at one end • Male bougies – 4 g and 100 -150 mm • Female bougies – 2 g and 60 – 75 mm Nasal bougies • Thin pencil shaped with pointed ends • 1. 2 g and 9 – 10 mm long Ear cones • Pencil shaped

NEW TRENDS OF SUPPOSITORIES 1)TABLET SUPPOSITORIES 2) LAYERED SUPPOSITORIES 3) CAPSULE SUPPSITORIES 4) COATED SUPPOSITERIES 5) DISPOSABLE SUPPOSITORIES
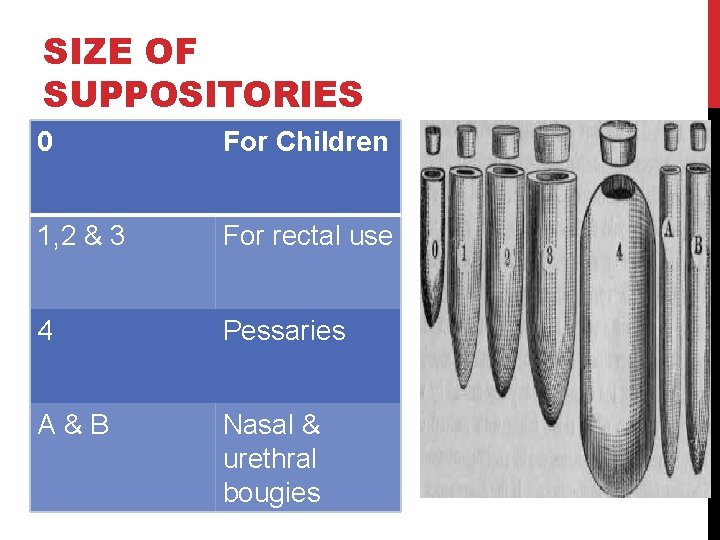
SIZE OF SUPPOSITORIES 0 For Children 1, 2 & 3 For rectal use 4 Pessaries A&B Nasal & urethral bougies
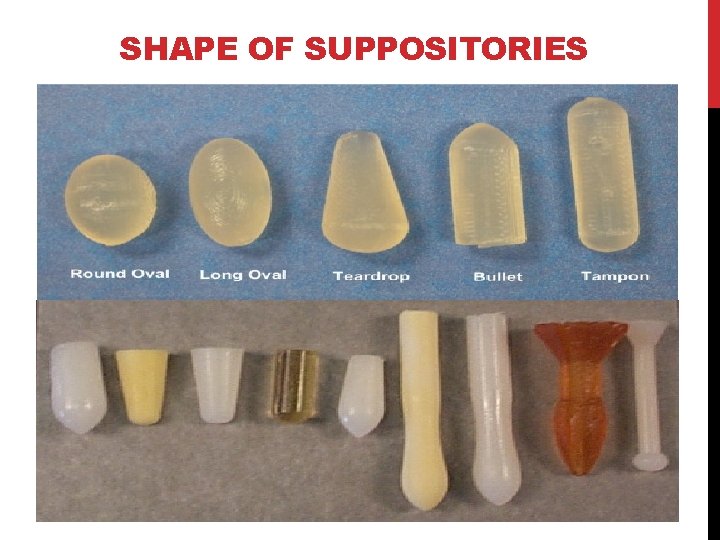
SHAPE OF SUPPOSITORIES

RECTUM PHYSIOLOGY • Terminal 15 -19 cm of large intestine (LI) • Rectal Fluids -> no buffering capacity • p. H 6. 8 • LI function absorb H 2 O and electrolytes • � Low surface area -> poor absorption compare SI
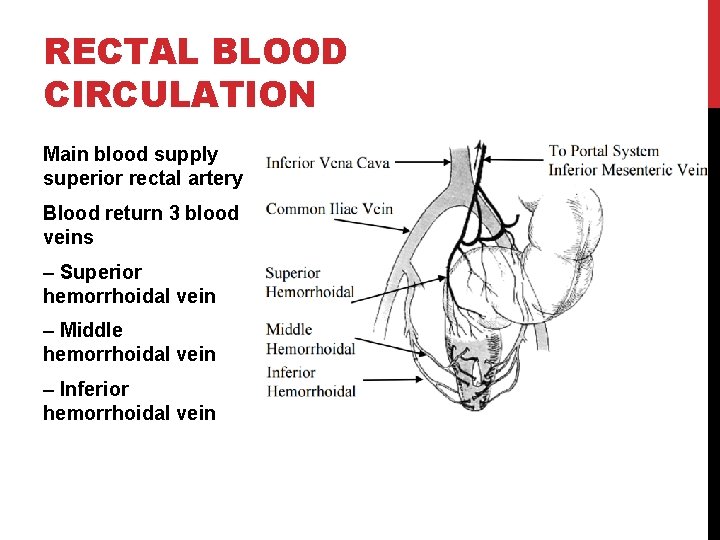
RECTAL BLOOD CIRCULATION Main blood supply superior rectal artery Blood return 3 blood veins – Superior hemorrhoidal vein – Middle hemorrhoidal vein – Inferior hemorrhoidal vein

ADVANTAGE Drug goes directly into systemic circulation No first pass metabolism by liver Drug avoids stomach and digestive enzymes Patient counseling -> don't place too high in rectum
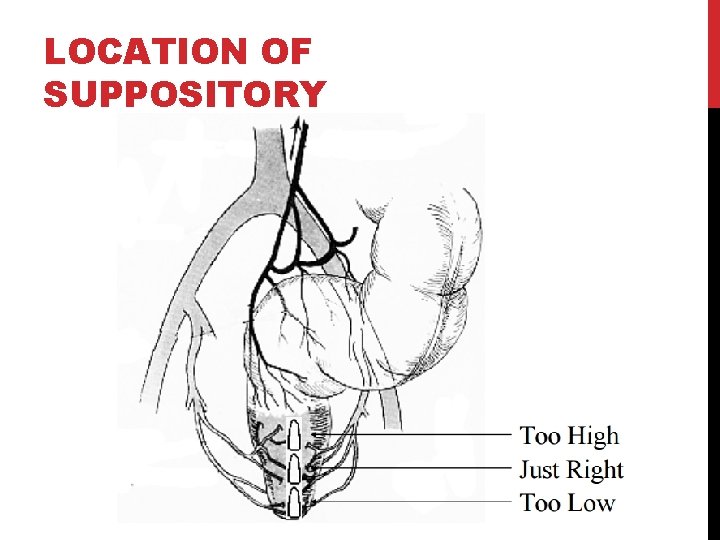
LOCATION OF SUPPOSITORY

FORMULATION

PROBLEMS IN FORMULATION OF SUPPOSITORIES 1. Water in suppositories Formulators do not like to use water for dissolving drugs in suppositories for the following reasons : a. Water causes oxidation of fats. b. If the suppositories are manufactured at a high temperature, the water evaporates, the drugs crystallize out. c. Absorption of water soluble drugs is enhanced only if the base is an oil – in – water emulsion with more than 50% of the water in the external phase. d. Drug excipient interactions are more likely to happen in the presence of water. e. Bacterial contamination may be a problem, so we may be forced to add a preservative.

2. Hygroscopicity Glycerogelatin suppositories lose moisture in dry climates and absorb moisture in humid conditions The hygroscopicity of polyethylene glycol bases depends on the chain length of the molecule As the molecular weight of these ethylene oxide polymers increases the hygroscopicity decreases

3. DRUG-EXCIPIENT INTERACTIONS Incompatibilities exist between polyethylene glycol base and some drugs. Ex: Sodium barbital and salicylic acid crystallize out of polyethylene glycol. High concentrations of salicylic acid soften polyethylene glycol to an ointment like consistency. Penicillin G is stable in cocoa butter and other fatty bases. It decomposes in polyethylene glycol bases.

4. Viscosity When the base has low viscosity, sedimentation of the drug is a problem. 2% aluminium monostearate may be added to increase the viscosity of the base Cetyl and stearyl alcohols or stearic acid are added to improve the consistency of suppositories

5. Brittleness Cocoa butter suppositories are elastic, not brittle Synthetic fat bases are brittle This problem can be overcome by keeping the temperature difference between the melted base and the mold as small as possible Materials that impart plasticity to a fat and make them less brittle are small amounts of Tween 80, castor oil, glycerin or propylene glycol

6. Density of the base, the drug, the volume of the mould and whether the base is having the property of volume contraction are all important. They all determine the weight of the suppository 7. Lubrication of moulds Some widely used lubricating agents are mineral oil, aqueous solution of SLS, alcohol and tincture of green soap. These are applied by wiping, brushing or spraying

8. Volume contraction On solidification the volume of the suppository decreases. The mass of the suppository pulls away from the sides of the mould. This contraction helps the suppository to easily slip away from the mould, preventing the need for a lubricating agent. Sometimes when the suppository mass is contracting, a hole forms at the open end. This gives an inelegant appearance to the suppository. Weight variation among suppositories is also likely to occur. This contraction can be minimized by pouring the suppository mass slightly above its congealing temperature into a mould warmed to about the same temperature. Another way to overcome this problem is to overfill the molds, and scrape off the excess mass which contains the contraction hole.

9. Displacement value The volume of suppositories from a particular mould will be constant but the weight will vary because the densities of the medicaments usually differ from the density of the base, and hence the density of the medicament will affect the amount of the base required for each suppository

10. Weight and volume control Various factors influence the weight of the suppository, the volume of the suppository and the amount of active ingredient in each suppository. They are: 1. Concentration of the drug in the mass 2. Volume of the mould cavity 3. The specific gravity of the base 4. Volume variation between moulds 5. Weight variation between suppositories due to the inconsistencies in the manufacturing process. The limit for the weight variation in suppositories is 5%.

11. Rancidity The unsaturated fatty acids in the suppository bases undergo auto oxidation and decompose into aldehydes, ketones and acids. These products have strong, unpleasant odours The lower the content of unsaturated fatty acids in a base, the higher is its resistance to rancidity

DRUG 1. Should have sufficient absorption from particular body cavity (if for systemic use) 2. Best suited for drugs undergoing first pass metabolism, degrade in GI fluids or irritate the GI mucosa 3. Should be easily dispersible or soluble in the base 4. Should be soluble to achieve homogeneity but should not have more affinity for the base or else it wouldn't get released 5. Particle size should be less to improve bioavailability and decrease irritation 6. Density and solubility of the drug should ensure minimum usage of base and formation of smaller suppositories 7. Drug should be compatible and stable in base and at processing conditions
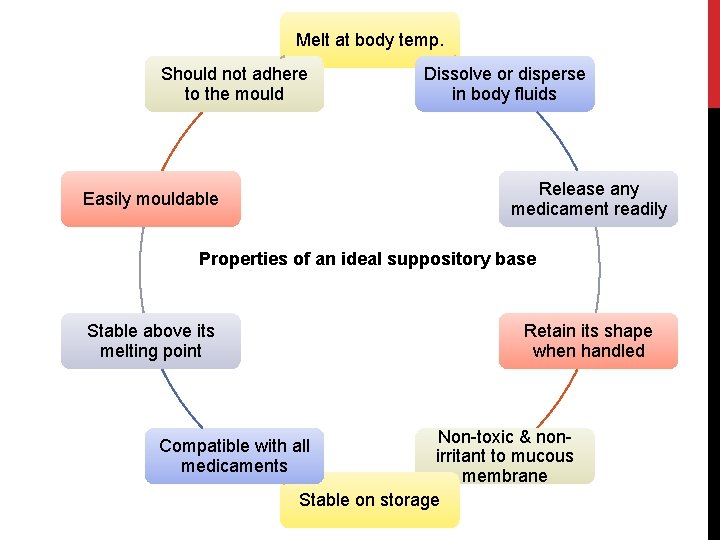
Melt at body temp. Should not adhere to the mould Dissolve or disperse in body fluids Release any medicament readily Easily mouldable Properties of an ideal suppository base Stable above its melting point Retain its shape when handled Non-toxic & nonirritant to mucous membrane Stable on storage Compatible with all medicaments
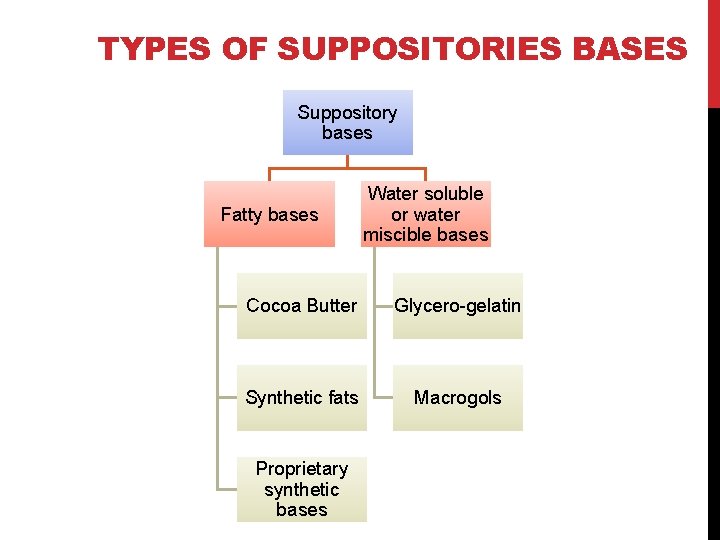
TYPES OF SUPPOSITORIES BASES Suppository bases Fatty bases Water soluble or water miscible bases Cocoa Butter Glycero-gelatin Synthetic fats Macrogols Proprietary synthetic bases

SUPPOSITORY BASES A. Oleaginous bases: 1. Cocoa butter. 2. Emulsified cocoa butter. 3. Hydrogenated oils. B. Hydrophilic bases/ aqueous bases: 1. Glycero-gelatin base. 2. Soap-glycerin base. 3. Polyethylene glycol. C. Emulsifying/Synthetic bases: 1. Witepsol 2. Massa estarinum 3. Massuppol.

FATTY BASES: Cocoa butter: • Source: • Cocoa butter is fat obtained from the roasted seed of Theobroma cocoa. • Properties: • At room temperature it is a yellowish, white solid having a faint, agreeable chocolate like odour. • Chemically, it is a triglyceride (combination of glycerin and one or different fatty acids) primarily of oleopalmitostearin and oleodistearine. • It melts at 30 - 350 C,

• ADVANTAGES: • Melting just below the body temperature. • Maintaining its solidity at usual room temperatures. • Readily liquefy on heating and solidify on cooling. Disadvantages: • Exhibits marked polymorphism. • Rancidity. • Stick to mould. • Leakage from body cavity. • Costly. • Immiscibility with body fluid. • Chloral hydrate or lactic acid liquefies it.

EMULSIFIED THEOBROMA OIL: Emulsified theobroma oil: • It is used as base when large quantities of aqueous solution are to be incorporated.

HYDROGENATED OILS: • These are obtained by hydrogenation of various vegetable oils. • These include hydrogenated vegetable oils, such as coconut, palm kernel, cottonseed, peanut, fractionated palm kernel oil etc.

Advantages: • Hydrogenation increases resistance to oxidation. • Increases chemical inertness, • Lubrication not required. • Disadvantages: • Become brittle on rapid cooling. • Sedimentation of added substance take place.

HYDROPHILIC BASES/AQUEOUS BASES: Glycerogelatine base; • It is a mixture of glycerin and water which is made stiff by the addition of gelatin. • Type of gelatin bases: to avoid incompatibility. Type A or Pharmagel A: acidic in nature and used for acidic drugs having isoelectric point (7 -9) Type B or Pharmagel B: alkaline in nature and used for basic drugs having isoelectric point (4. 7 -5. 0). • Used for vaginal suppositories The isoelectric point (p. I, p. H(I), IEP), is the p. H at which a particular molecule carries no net electrical charge in the statistical mean.

Advantages: • It melts at body temperature. • It mix with body fluid. • Not rancid. • It can be used to prepare suppositories using boric acid, chloral hydrate bromides, iodoform opium etc. Disadvantages: • Difficult to prepare and handle. • Chance of bacterial growth. • Hygroscopic in nature. (become hard on drying and soft in cont with moisture) • Laxative in action. • Incompatible with tannic acid, gallic acid, ferric chloride etc

Soap-glycerin base: • In this base glycerin is replaced by soap or sodium stearate. • This makes the base hard. Polyethylene glycols: • These are commonly known as carbowax. • These are available in solid, liquid or semi-solid state depending on molecular weight.

EMULSION BASES: Witepsol: • They consist of triglycerides of saturated vegetable fatty acid with varying percentage of partial esters. • A small amount of beeswax is added for use in hot climate. • It should not be cooled rapidly as it become brittle and fracture. • Lubrication is required.

Massa estarinum: • It is a mixture of mono, di and triglycerides of saturated fatty acids having the formula C 11 H 23 COOH to C 17 H 35 COOH. • This is also known as adeps solidus. • It is a white, brittle, almost odourless and tasteless solid. • It has a m. p. 33. 5 to 35. 50 C. • They are available in various grades but grade B is commonly used in dispensing.

Massuppol: • It consists of glyceryl esters mainly of lauric acid to which small amount of glyceryl monostearate has been added to improve its water absorbing capacity.

1)FATTY BASES

COCOA BUTTER (THEOBROMA OIL): Properties It is the most widely used suppository base. And it is used in the prescriptions when no base is specified. It is naturally occuring triglyceride with oleopalmitostearin and oleodistearin glyceride chain and contains 40% of the unsaturated fatty acid. It is yellowish white, solid, brittle fat, which smells and tastes like chocolate. Its melting point lies between 30 -35 o. C (86 -95 o. F) its iodine value is between 34 and 38 and its acid value is no higher than 4. It satisfies the requirements for an ideal base – innocuous, bland, and non-reactive, and melts at body temp.

ADVANTAGES OVER THEOBROMA OIL I. They readily liquefy on worming and solidify rapidly. II. They are most suitable bases III. Melts at body temperature.
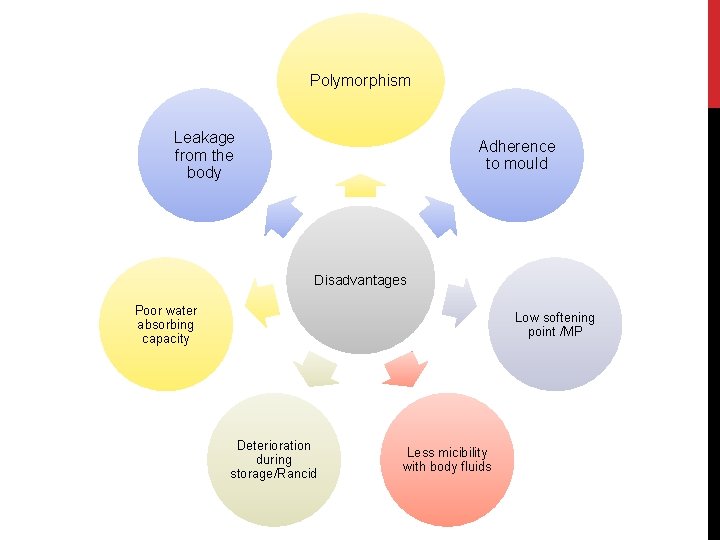
Polymorphism Leakage from the body Adherence to mould Disadvantages Poor water absorbing capacity Low softening point /MP Deterioration during storage/Rancid Less micibility with body fluids

POLYMORPHISM Polymorphism in cocoa butter is observed due to high proportion of unsaturated triglycerides. The formation of various forms of cocoa butter depends on the degree of heating, on the cooling process and on the conditions during this process. Each form of cocoa butter has different melting point and drug release rates.

COCOA BUTTER EXITS IN FOUR CRYSTALLINE STATE α form ß` form γ form • melts at 24 o. C • Obtained by suddenly cooling melted cocoa butter to 0 o. C. • Crystallizes out of the liquefied cocoa butter with stirring at 18 to 23 o. C. • Its melting point lies between 28 and 31 o. C. • changes slowly into the stable ß form. • Melts between 34 and 35 o. C. • Change is accompanied by volume contraction. • melts at 18 o. C • Obtained by pouring a cool cocoa butter, before it solidifies, into a container which is cooled at deep freeze temp.
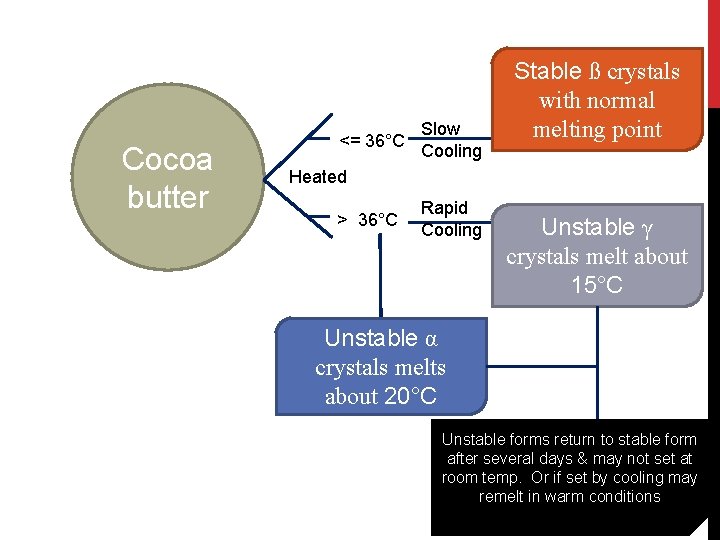
Cocoa butter <= 36°C Slow Cooling Stable ß crystals with normal melting point Heated > 36°C Rapid Cooling Unstable γ crystals melt about 15°C Unstable α crystals melts about 20°C Unstable forms return to stable form after several days & may not set at room temp. Or if set by cooling may remelt in warm conditions

Adherence to the mould Cocoa butter does not contract sufficiently on cooling to loosen the suppositories in the mould. Sticking may be overcome by adequate lubrication. Softening point too low for hot climates To raise the softening point, white bees wax may be added to theobroma oil suppositories intended for use in tropical and subtropical countries. Melting point reduced by soluble ingredients Phenol and chloral hydrate have a tendency to lower the melting point of cocoa butter. So, solidifying agents like beeswax (4%) may be incorporated to compensate for the softening effect of the added substance.

Rancidity on storage: Due to the oxidation of unsaturated glycerides. Poor water Improved by the addition of emulsifying agents. -absorbing ability: Leakage from the body: Sometimes the melted base escapes from the rectum or vagina, so, it is rarely used as a pessary base. Expensive Relatively high cost

SYNTHETIC FATS(EMULSIFIED THEOBROMA OIL AND HYDROGENATED OILS To overcome the disadvantages of theobroma oil synthetic substitutes were searched. Obtained from hydrogenation and heat treatment to vegetable oils such as palm kernel and arachis. Hydrogenation saturates unsaturated glycerides and heat treatment splits some of the triglycerides into fatty acid and partial esters (mono and di glycerides). Most synthetic fat bases are made by first hydrolysing the vegetable oil, then hydrogenating the resulting fatty acids and finally esterifying the acids by heating with glycerol.

Advantages Their solidifying points are unaffected by over heating. They have good resistance to oxidation because their unsaturated fatty acids have been reduced. The difference between melting and setting points is small; generally only 1. 5 to 2 o. C and seldom over 3 o. C. Hence, they set quickly, the risk of sedimentation is low and they are easier to administer. The melting point depression caused by fat soluble drugs can be counteracted by choosing a high melting point grade, while the hardness and brittleness that sometimes results from a high content of insoluble powder can be prevented by using a low melting point grade.

Advantages High softening point grades are advantageous for tropical and sub tropical formulations. They usually contain a proportion of partial glycerides some of which e. g. glyceryl monostearate, are w/o emulsifying agents and therefore their emulsifying and water absorbing capacities are good. No mould lubricant is needed because they contract significantly on cooling. They produce suppositories that are white and almost odourless and have very attractive, clean and polished appearance.

Disadvantages They should not be cooled in a refrigerator or ice because they become brittle if cooled quickly. Additives such as polysorbate 80 correct this fault. They are more fluid than theobroma oil when melted and at this stage sedimentation is greater. Thickeners such as magnesium stearate, bentonite reduce this problem. The release and absorption of drugs in the body may differ for theobroma oil and synthetic bases.

3) EMULSIFYING BASES

These are synthetic bases and a number of propriatery synthetic bases are available in the market. Some of these are described as under 1) WITEPSOL They consist of triglycerides of saturated vegetable acid with varying percentage pf partial esters. A small amount of beeswax is added for use in hot climates. The suppositories prepared with witepsol bases should not be cooled rapidly , in order to prevent them from becoming brittle and fracture. The mould should be also properly lubricated to get good qualities of suppositories.

2)MASSA ESTARINUM It is mixture of mono , di- and –triglycerides of saturated fatty acids having the formula C 11 H 23 COOH to C 17 H 35 COOH. These is also known as adeps solidus. It is a white , brittle , almost odurless and tasteless solid. It has a m. p 33. 5 to 35. 5 C. They are available in various grades but grades B is commonly used in dispensing. 3) MASSUPPOL It consist of glycerol esters mainly of lauric acid to which small amount of glycerol monosterate has been added to improve its water absorbing capacity.

PROPRIETARY SYNTHETIC BASES Massuppol Whitepsol Massa Estarinum • It consists of triglycerides of saturated vegetable acids (C 12 to C 18) with varying proportions of partial esters. • The W 45 grade is used for general dispensing. • It consists of mixture of tri, di and monoglycerides of saturated fatty acids with chain lengths of C 11 to C 17. Grade B is recommended for general dispensing. • This differs from the previous materials in being single general purpose base with only one modification, for cold moulding. • It consists of glyceryl esters, mainly of lauric acid to which a small amount of glyceryl monostearate has been added to improve its water absorbing capacity. • The B. P. C allows the use of hydrogenated vegetable oils provided the melting point of the suppositories is not above 37 o. C.

2)WATER SOLUBLE OR WATER MISCIBLE BASES

GLYCERO-GELATIN This is a mixture of glycerol and water into a stiff jelly by adding gelatin. It is used for making jellies, suppositories and pessaries and its proportion is changed according to its intended purpose. Glycero-gelatin dissolves in body secretions and therefore is preferable to a fatty base for administering antiseptics. Since, solution is slow, drug release is more prolonged than from fatty base. At present the B. P allows a maximum disintegration time of 1 hr. for Glycerol Suppositories B. P made with gelatin of B. P standard.

Disadvantages of glycerogelatin base They have a physiological action (used as laxative) They are more difficult to prepare and handle. Their solution time depends on the content and quality and gelatin and the age of the base. They are hygroscopic. So a careful storage is required. It also leads to dehydration of the rectal mucosa with consequent irritation; this is an advantage where a laxative effect is required. Gelatin is incompatible with protein precipitants such as tannic acid.

SOAP –GLYCERIN SUPPOSITORIES In glycero – gelatin bases , the gelatin is replaced with either curd soap or sodium stearate which makes base sufficiently hard to prepare good quality of suppositories. Soap also helps in evacuation action of glycerin. POLYETHYLINE GLYCOLS These are polymers are commonly known as carbowaxes or polyglycoals or macrogols. The physical charater of these carbowaxes varies accorrding to the molecular weight. The macrogols having molecular weight less than 1000 are liquids and those with Molecular weight higher than 1000 are wax like solids.

ADVANTAGES 1)They are chemically stable. 2)They are non irritant. 3)They do not allow the bacterial or mould growth to take place. 4)They are physiologically inert substances. 5)Physical properties of the base can be change according to the need by combining the different proportion of high and low and high melting point polymers. 6)They provide the prolonged action because they do not melt in the body cavity but dissolves slowly for a long time. 7)They do not stick to the site of the mould. 8) The suppositories made from microgols have clean and smooth apperance.

DISADVANTAGES I. They are hygroscopic and hence require special storage conditions to store them. II. They are incompatible with certain drugs like tannins , phenols , etc. III. Its goods solvent properties can results retention of the drug in liquified base in body , thereby redusing there therapeuic activity. IV. High solubility of microgols leads to supersaturation which in turn make crystals and fracture the product on storage.

MACROGOLS (PEG) Properties long chain polymers of ethylene oxide with general formula HOCH 2(CH 2 OCH 2)8 CH 2 OH Exist as liquid if their average molecular range from 200 to 600 and they exist as wax like solid it is above 1000. Their water solubility, hygroscopicity and vapour pressure with increase in average molecular weight. They do not hydrolyse or deteriorate and are physiologically inert and do not support mold growth. The PEG suppositories can be prepared by both moulding and cold compression methods.

ADVANTAGES OF MACROGOLS 1. The mixtures have melting point above 42 o. C. Hence, cool storage is not required, they are satisfactory for use in hot climates, and administration is easy because they are not slippery to handle. 2. Because of this high melting point they do not melt in the body but gradually dissolve and disperse, freeing their medication slowly and providing longer action than fatty bases. 3. Their physical properties can be varied by suitable admixture of high and low polymers. High polymers give hard products that disintegrate and release their drug slowly. • Softer, less brittle preparations that disperse and liberate their drug more quickly are obtained by mixing high with either medium or medium and low polymers or by adding plasticizers.

ADVANTAGES OF MACROGOLS 4. They do not stick to the mould since they contract on cooling. 5. Because of their high molecular weight solution of high viscosity are produced when they disperse in the body. 6. They absorb water well and have excellent solvent properties. 7. Products have clean smooth appearance.

DISADVANTAGES OF MACROGOLS 1. They are hygroscopic so careful storage is required. Irritancy can be reduced by incorporating about 20% of water in the mass or by instructing the patient to dip the preparation in water just before insertion. This type of base is suitable for systemically active drugs. 2. Its good solvent properties can result in retention of the drug in the liquefied base in the body with consequent reduction in therapeutic activity. 3. Products sometimes fracture on storage, particularly if they contain. High solubility of macrogols which can lead to a super saturated solution in the water and subsequent crystallisation and this the mass granular and brittle.

DISADVANTAGES OF MACROGOLS 4. Crystal growth of certain medicaments may occur particularly if they are partly in solution and partly in suspension in the base. This makes the product brittle and crystals may be irritating because they are large and takes longer time to dissolve. 5. They are incompatible with bismuth salts, tannins and phenol. They lower the activity of some antibacterial agents and dissolve certain plastics necessitating care in choosing containers.

PREPARATION OF SUPPOSITORIES AND PESSARIES

3 METHODS 1. Moulding 2. Hand rolling 3. Compression

PREPARATION OF SUPPOSITORIES MOULDS
CLEANING OF MOULDS Mould Interior – Don’t scrap or rub with abrasive Immerse in hot detergent water Wipe with soft cloth Rinse thoroughly
NORMAL CAPACITIES OF MOULDS 1 g (15 gr) 2 g (30 gr) 4 g (60 gr) 8 g (120 gr)

WAYS OF PREPARING PROPRIETARY SUPPOSITORIES – CONVENTIONAL MOULDS Individual packing in laminated foil Strip packing in hermatically sealed tight fitting laminated foil or plastic film

PLASTIC MOULDS Heat sealable laminated foil Press on plastic caps Plastic Moulds – polythene, polyvinyl chloride or a complex film with polyvinyl chloride. Choice depends on stability of contents to air and moisture.

CALIBRATION OF MOULD Calibration of mould is necessary before preparing suppositories and pessaries. The capacity of the mould varies with the different bases. Each mould should be calibrated using the base alone, weighing the products and taking the mean weight as true capacity.

DISPLACEMENT VALUES The volume of the suppositories is uniform but the weight differs because there is difference in densities of the medicaments and base. So the allowance must be made for the change in the density of the mass due to added medicaments. Number of parts by weight of medicament that displaces one part by weight of the base.

DISPLACEMENT VALUE: It is the amount of medicament required to displace one part of suppository base. Significance: (2 Marks) The volume of suppositories from a particular mould will be constant but the weight will vary because the densities of the medicaments usually differ from the density of the base, and hence the density of the medicament will affect the amount of the base required for each suppository.
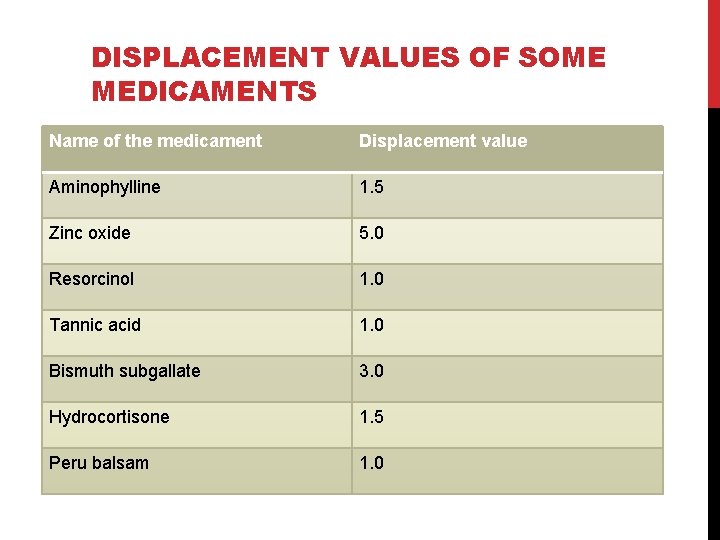
DISPLACEMENT VALUES OF SOME MEDICAMENTS Name of the medicament Displacement value Aminophylline 1. 5 Zinc oxide 5. 0 Resorcinol 1. 0 Tannic acid 1. 0 Bismuth subgallate 3. 0 Hydrocortisone 1. 5 Peru balsam 1. 0

LUBRICATION OF MOULD – MUST PROVIDE A BUFFER FILM BETWEEN THE SUPPOSITORY AND THE METAL Synthetic fat or macrogol base Glycerogelatin base Theobroma oil Contract significantly on cooling Sticky nature Oily lubricant cannot be used Oily lubricant – liquid paraffin or arachis oil Soft soap -10 g Glycerol – 10 ml Alcohol(90%) – 50 ml Lubrication is not needed
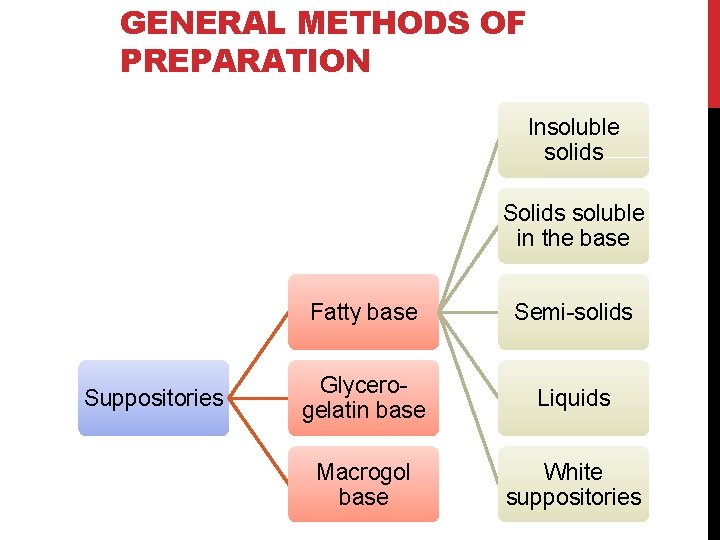
GENERAL METHODS OF PREPARATION Insoluble solids Solids soluble in the base Suppositories Fatty base Semi-solids Glycerogelatin base Liquids Macrogol base White suppositories

FATTY BASE – SUPPOSITORIES CONTAINING INSOLUBLE SOLIDS – THEOBROMA OIL AND SYNTHETIC BASES 1. Check the displacement values of medicaments 2. Calculate the quantity of medicaments 3. Excess quantity is weighed – unavoidable wastage Major loss due to sticking of the material to the sides of the dish So the smallest dish should be selected.
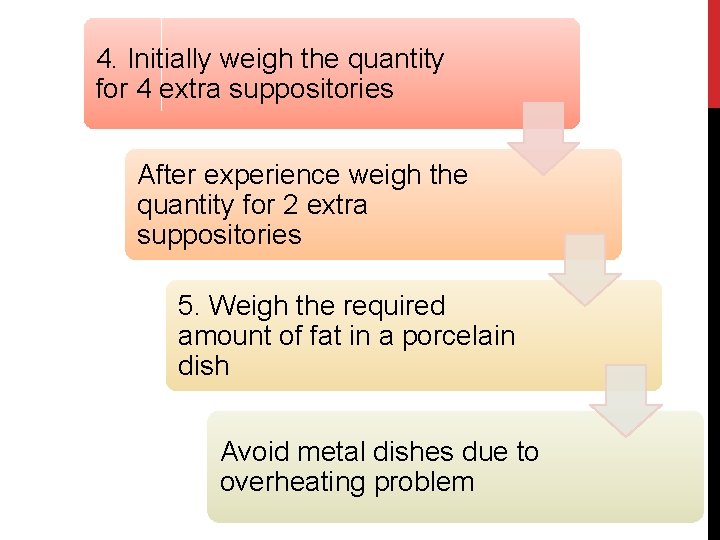
4. Initially weigh the quantity for 4 extra suppositories After experience weigh the quantity for 2 extra suppositories 5. Weigh the required amount of fat in a porcelain dish Avoid metal dishes due to overheating problem

6. Finely powder the medicaments and pass it through sieve no. 180 and weigh the required quantities 7. Heat a tile on water bath and mix the powder on a tile with a flexible spatula 8. Place the base on water bath until 2/3 rd contents are melted and then remove it. Overheating may occur if the base is allowed to melt completely. 9. Rest of the base is melted with continuous stirring with spatula and avoid glass rod for stirring

10. Pour about half of the base on to the mixed medicaments 11. Prepare a smooth dispersion as quickly as possible by levigating with spatula 12. To prevent excessive cooling use small tile and this will also avoid loss on the edge of the tile 13. If the base is solidified hold the tile on water bath for few seconds 14. Transfer the dispersion to the dish and stir to form homogenous mixture
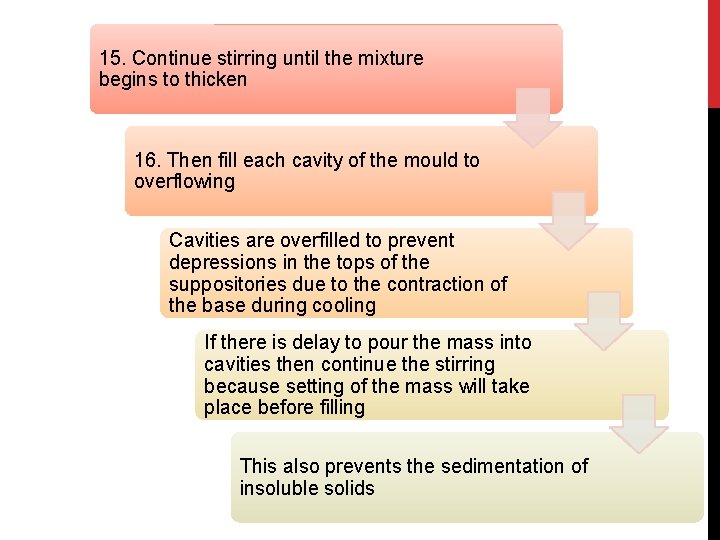
15. Continue stirring until the mixture begins to thicken 16. Then fill each cavity of the mould to overflowing Cavities are overfilled to prevent depressions in the tops of the suppositories due to the contraction of the base during cooling If there is delay to pour the mass into cavities then continue the stirring because setting of the mass will take place before filling This also prevents the sedimentation of insoluble solids

17. There may be solidification of mass during pouring so reheat the mass and make it pourable The base of the dish should be wiped to prevent contamination of mass with water 18. Leave in a cool place until the mass is set and remove the excess with a sharp knife or razor blade or warm spatula 19. Put the knife or blade or spatula at an angle of about 30°C and draw it from left to right pressing down at the same time 20. Open the mould and remove the suppositories. If difficult to open tap the mould and remove it.

ADDITIONAL PRECAUTIONS FOR THEOBROMA OIL SUPPOSITORIES Thoroughly lubricate the mould and cool and drain the excess in refrigerator or ice box Cooling hasten setting of base and reduces sedimentation of suspended solids Take care not to over heat the base If the base is too thick to pour then cool the base to excess and again reheat it and then pour the base in mould.

SUPPOSITORIES CONTAINING SOLIDS SOLUBLE IN BASE When soluble substances are used there is depression in melting point. Product is satisfactory and quite firm. Occasionally the suppositories become too soft to use. To avoid this a synthetic base with higher melting should be used. E. g Massa Estranium Type C or Witepsol E 75 BPC – White beeswax (M. P: 62 to 64 °C) – complicates the preparation when used with theobroma oil – affects the solidifying point of the base and delays setting. These problems can be avoided if following procedure is followed
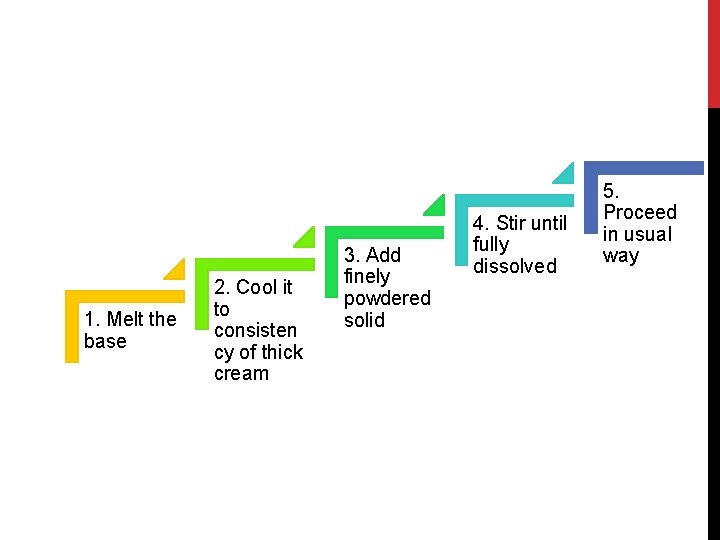
1. Melt the base 2. Cool it to consisten cy of thick cream 3. Add finely powdered solid 4. Stir until fully dissolved 5. Proceed in usual way

SUPPOSITORIES CONTAINING SEMISOLIDS 1. Reduce the viscosity of drug by trituration on warm tile with enough water 2. Homogenous liquid of consistency of glycerol or somewhat thinner is formed 3. Added water is allowed for and transfer the dilution to the tared container 4. Add sufficient melted base to produce a weight equal to the product of the nominal capacity of the mould and the number of suppositories in excess 5. Mass is stirred vigorously to form a mechanical emulsion and the preparation is completed in normal manner

Care should be taken during stirring the mass to form mechanical emulsion because it should not be continued before the liquid becomes somewhat unpourable. Satisfactory dispersion can be ensured by – • The mixture must stirred vigorously to produce a perfect dispersion. • Pouring must be delayed to the last possible moment so that the globules of aqueous liquid are enclosed in a solid film fat. In this method synthetic bases are preferred because they contain emulsifying agents. Water containing suppositories are susceptible to microbial growth so addition of preservative agent is necessary. Water may evaporate leaving cavities or creating high local concentration of drug that cause irritation. Failure to establish and maintain a stable emulsion during preparation may lead to moisture seeping of the product.

SUPPOSITORIES CONTAINING LIQUIDS Liquids used in suppositories fall into 2 categories 1. Liquids that are volatile (e. g. volatile oils) or owe their medicinal use to volatile constituents (e. g. peru balsam). These cannot be evaporated. They lower the melting point of fatty base. 2. Liquids that are aqueous or hydro-alcoholic that owe their medicinal value to non-volatile constituents. Removal of part of solvent is permissible.
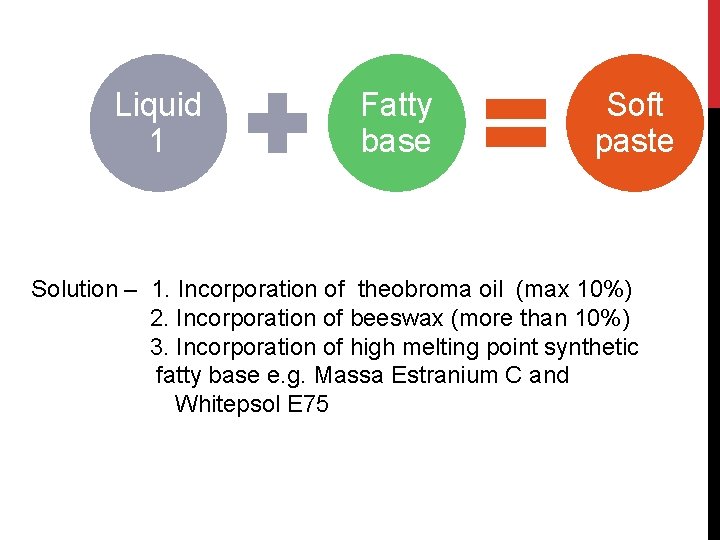
Liquid 1 Fatty base Soft paste Solution – 1. Incorporation of theobroma oil (max 10%) 2. Incorporation of beeswax (more than 10%) 3. Incorporation of high melting point synthetic fatty base e. g. Massa Estranium C and Whitepsol E 75

For liquids of group 2 the method for semi-solids is suitable. 20% of aqueous or hydro-alcoholic liquid can be mechanically emulsified in theobroma oil. Liquids are concentrated by evaporation (if permissible) or a larger size of suppository is used.

WHITE SUPPOSITORIES Suppositories containing no coloured ingredient. Care is taken to ensure that the product is looking white and not a dirty looking grey. Mould, tile, spatula, mortar, sieve, and hands must be clean. Porcelain dish is preferred over metal dish because their smooth surface is easy to wash and their white colour displays dirt clearly. E. g. Aminophylline, suppositories. Cinchona and morphine white

SUPPOSITORIES WITH A GLYCERO-GELATIN BASE Dispensed as Unmedicated Glycerol suppositories BP Medicated BPC pessaries of crystal violet, and lactic acid Density of the base: Glycero-gelatin base is 1. 2 times denser than theobroma oil so the mould should be calibrated with it.

PREPARATION 1. Lubricate the mould with liquid paraffin or arachis oil; invert and cool. 2. Take glycerol in evaporating dish and keep it in water bath and heat it to 100°C 3. Dissolve thermostable water soluble medicament in 30 ml of water and heat it to boiling in evaporating dish for few minutes and avoid excessive evaporation 4. Add water to powder gelatin and stir gently to wet the material. If gelatin sheets are used then trim off the hard outer edges and cut into small pieces. 5. Add hot glycerol, restir and transfer to the boiling water bath for 15 min or until solution is complete.

Make sure that the dish is low in water bath for maximum heating. Stir gently to avoid formation of air bubbles. Heating may be done direct on bunsen burner instead on water bath provided following precautions are taken. 1. The preparation is watched constantly. 2. The bunsen is used as little as possible and only to restore the temp. to 100°C. Overheating may reduce the gelatin strength. 3. The gelatin is not allowed to settled at the bottom of the dish 4. If settled, gelatin may char or remain on the sides above the liquid level where it may denature and insoluble. In this method there is less risk of skin formation than the water bath method.

6. When the solution is complete weigh the dish and the contents 7. If necessary add or evaporate the purified water to obtain the correct weight and remove the air bubbles by passing it thorough muslin cloth 8. Pour the hot mass to the chilled moulds 9. Overfilling of the mould is not necessary because glycero-gelatin bases contract very little on cooling and also the excess cannot be removed off neatly. So fill it to slight convexity 10. Leave the mould in an ice box or refrigerator for about half an hour

12. Remove the mould and open it. 13. If there are signs of spliting of suppositories from the centre line then invert the mould and try to open it from the bottom 14. Peel away each suppository from the base to tip and stand it on its base on a tile very thinly lubricated with liquid paraffin or oil 15. If necessary trim off the base and then roll each suppository to apply a coat of lubricant to prevent its sticking to the container.

16. Soluble thermolabile medicaments are dissolved in water or specified solvent and this solution is incorporated in molten mass 17. Insoluble substances are rubbed down on a tile with part of the glycerol and melted base is not used for this purpose it is difficult to remelt the base

GLYCERO-GELATIN AS PESSARY BASE Glycero-gelatin pessaries heated to about 100°C for 1 hour with high concentration of glycerol (antibacterial agent) because gelatin is obtained from natural source so may be contaminated with pathogenic micro-organisms like E. Coli.

SUPPOSITORIES WITH A MACROGOL BASE Prepared in the same as that of synthetic base with following changes Mould must not be lubricated or chilled – rapid cooling causes brittleness Do not lubricate the mould – Lubrications spoils the surface Medicaments and base are melted together at same temperature; mixed and stirred to form a stable emulsion Water soluble ingredients are dissolved in water before addition Insoluble medicaments are added same to that of synthetic bases Mass is poured as cool as possible and moulds are over filled
![I] HAND ROLLING Hand molding is useful when we are preparing a small number I] HAND ROLLING Hand molding is useful when we are preparing a small number](http://slidetodoc.com/presentation_image_h2/5105b10de68476a1c89a17c00ce77fcf/image-103.jpg)
I] HAND ROLLING Hand molding is useful when we are preparing a small number of suppositories The drug is made into fine powder Incorporated into suppository base By trituration or kneading Desired shape is given by hand Rods are cut into pieces Mass is rolled into rod shape by fingers

II)Hot process or Fusion method is commonly used method for Preparation of suppositories. Method of preparation: - (3 marks) i) Thoroughly Clean & lubricate the mould with suitable lubricant. Keep it on ice in inverted position to cool & drain excess lubricant. ii) Heat the china dish over water bath. To this add required qty. of cocoa butter after taking into account the displacement value of medicament. & calculating for two extra suppositories for unavoidable wastage. iii) Remove the dish from water bath, when 2/3 rd of base melts & stir throughly until whole mass melts. To avoid overheating. iv) Place the weighed qty of medicament on an ointment tile. Pour about half of melted base over it. Mix it thoroughly with spatula.

v) Transfer the mixed mass again to china dish, mix it throughly & warm china dish over water bath for few seconds with constant stirring. vi) Pour the melted mass into the cavities of suppository mould. Kept over ice. Fill each cavity to over flowing, to prevent the formation of holes in suppositories. vii) Remove excess of mass with the help of sharp knife. viii) Open the mould & remove suppositories. ix) Wipe off the suppository lightly with a clean cloth or filter paper. x) Wrap the individual suppository in a wax paper.
![III] COMPRESSION MOLDING - LARGE SCALE OPERATION Prepared mass C is placed in a III] COMPRESSION MOLDING - LARGE SCALE OPERATION Prepared mass C is placed in a](http://slidetodoc.com/presentation_image_h2/5105b10de68476a1c89a17c00ce77fcf/image-106.jpg)
III] COMPRESSION MOLDING - LARGE SCALE OPERATION Prepared mass C is placed in a cylinder A It is forced through narrow opening D by means of piston B into a mould. Threads of mass pass in the mould G and are compressed until a homogenous fused mass is formed in E. On removal of retaining plate F the suppositories are ejected by further pressure. The mass and compression cylinder of the machine may be chilled to prevent heat of compression from making the mass too fluid. Useful for moulding suppositories containing insoluble solids (no risk of sedimentation) or thermolabile medicaments. Unsuitable for glycero-gelatin products
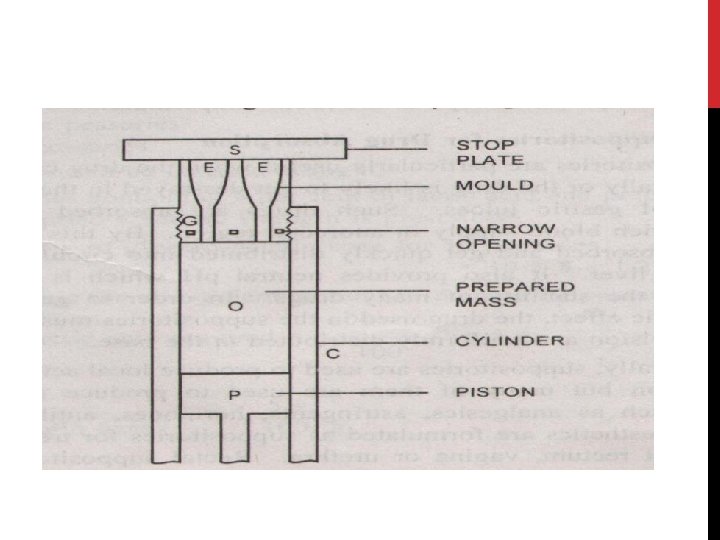

Advantages: 1. It is a simple method 2. It gives suppositories that are more elegant than hand moulded suppositories 3. In this method sedimentation of solids in the base is prevented 4. Suitable for heat labile medicaments Disadvantages: 1. Air entrapment may take place 2. This air may cause weight variation 3. The drug and/or the base may be oxidized by this air

PACKAGING AND LABELLING FOR SUPPOSITORIES Rigid paperboard boxes Flat box with a lid Labelling : 1. Store in a cool place 2. For rectal use only or not to be taken orally 3. Moisten before use (for glycero-gelatin and macrogol bases)

PACKAGING AND LABELLING FOR PESSARIES Glass bottles Plastic bottles Labelling : 1. Moisten the pessaries with water before insertion to reduce stinging caused by osmotic withdrawl of water to reduce the tissues during solution in vagina. 2. For vaginal use only or not to be taken orally. 3. Store in a cool place.

QUALITY CONTROL OF SUPPOSITORIES Appearance Uniformity of weight Disintegration
- Slides: 111