Supply of OPV by Ms RITES Twenty Fourth

Supply of OPV by M/s RITES Twenty Fourth Meeting Of the India Expert Advisory Group (IEAG) for Polio Eradication on 15 th March 2012 By S N Panda GM/Rites

Outline of Presentation • • • Introduction Procurement Objective Supply overview Our Experience Recommendation

Introduction • M/s RITES Limited(A PSU under Ministry of Rlys) assisting the Department of Health & Family Welfare as Procurement Agent for procurement of OPVs funded by External Agencies like World Bank since May’ 2010 previously handled by UNICEF • In FY 2010 -11 five indents were processed for four types of vaccines like b. OPV, t. OPV, m. OPV 1 and m. OPV 3 • In FY 2011 -12 two indents were processed for two types of vaccines like b. OPV and t. OPV only • In FY 2012 -13 one indent is already processed for b. OPV (Supply in April – June 2012)

Vaccine Supply Objectives To ensure a sustained and uninterrupted supply of quality vaccines Through • WHO Prequalification • Appropriate contracting
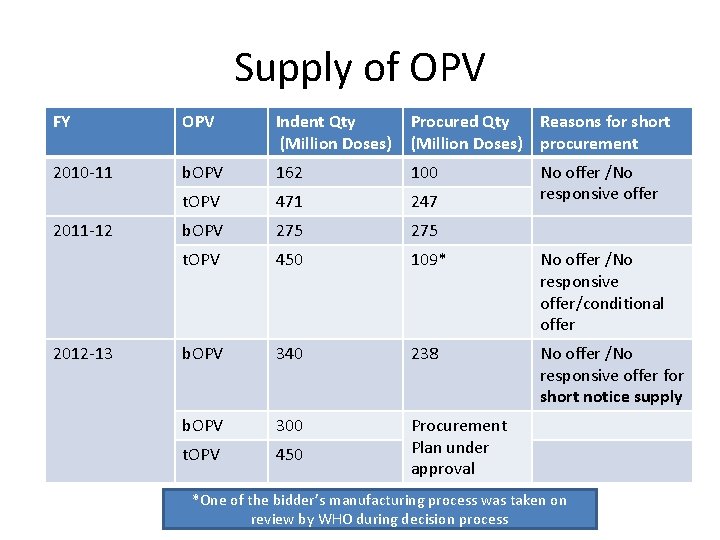
Supply of OPV FY OPV Indent Qty (Million Doses) Procured Qty Reasons for short (Million Doses) procurement 2010 -11 b. OPV 162 100 t. OPV 471 247 b. OPV 275 t. OPV 450 109* No offer /No responsive offer/conditional offer b. OPV 340 238 No offer /No responsive offer for short notice supply b. OPV 300 t. OPV 450 Procurement Plan under approval 2011 -12 2012 -13 No offer /No responsive offer *One of the bidder’s manufacturing process was taken on review by WHO during decision process
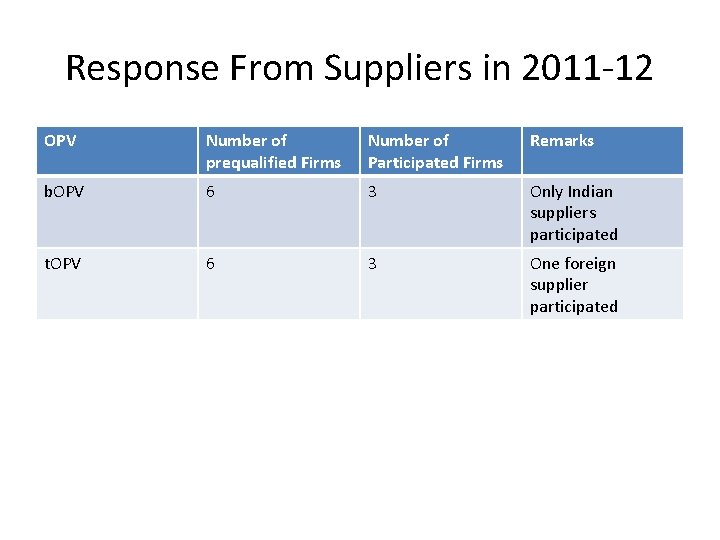
Response From Suppliers in 2011 -12 OPV Number of prequalified Firms Number of Participated Firms Remarks b. OPV 6 3 Only Indian suppliers participated t. OPV 6 3 One foreign supplier participated
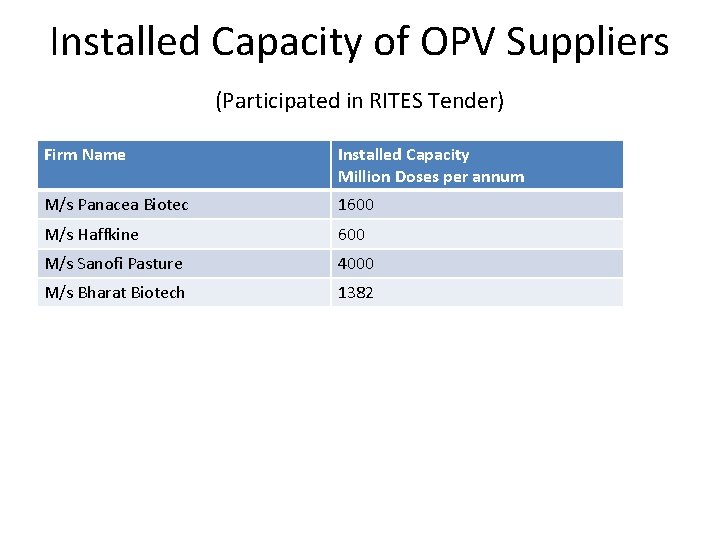
Installed Capacity of OPV Suppliers (Participated in RITES Tender) Firm Name Installed Capacity Million Doses per annum M/s Panacea Biotec 1600 M/s Haffkine 600 M/s Sanofi Pasture 4000 M/s Bharat Biotech 1382

Our OPV Procurement Experience • 50% of qualified suppliers participated in Bid • Mostly suppliers from India participated though it was a LIB except M/s Sanofi from France • No offer for full Bid quantity • Appreciable gap in offered price of different suppliers

Reasons for low response • Demand- Supply gap • Shortage of bulk in International market • Non participation of foreign suppliers due to (i) supply of OPV in short notice (ii) shelf life of minimum 18 months on the date of arrival at port of entry in India (iii) Marking of “CGS Not for Sale” on primary packaging
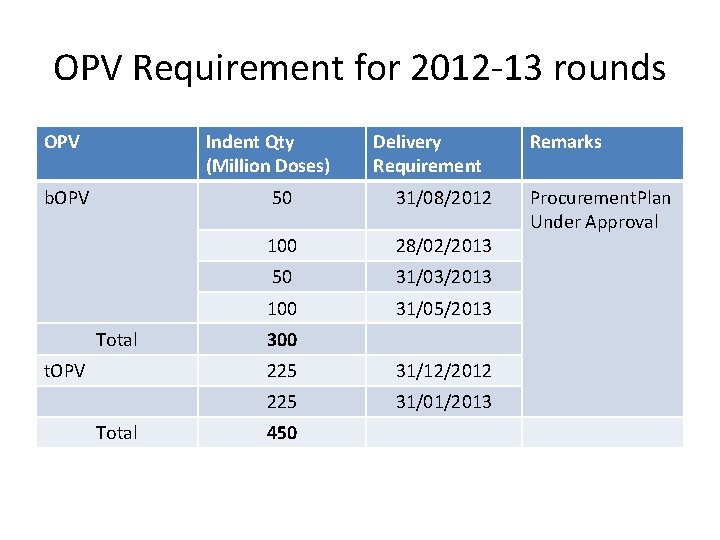
OPV Requirement for 2012 -13 rounds OPV Indent Qty (Million Doses) b. OPV Total t. OPV Total Delivery Requirement 50 31/08/2012 100 28/02/2013 50 31/03/2013 100 31/05/2013 300 225 31/12/2012 225 31/01/2013 450 Remarks Procurement. Plan Under Approval

Recommendation of RITES • Indent should be sent at least 12 months in advance with respect to supply requirement so that tender can be finalised six months prior to requirements. This implies advanced planning and long term IEAG recommendations. • Residual Shelf Life of vaccine at the time of supply as per Indian Drug & Cosmetic Act (i. e. 60% of total life of 24 months) instead of 18 months • Waiver of “CGS Not for Sale” on primary packaging

Thank You
- Slides: 12