Supply Chain Management of Mono Oral Polio Vaccine















- Slides: 17

Supply Chain Management of Mono Oral Polio Vaccine Type 2 (m. OPV 2) Polio Knowledge Team March 9, 2017

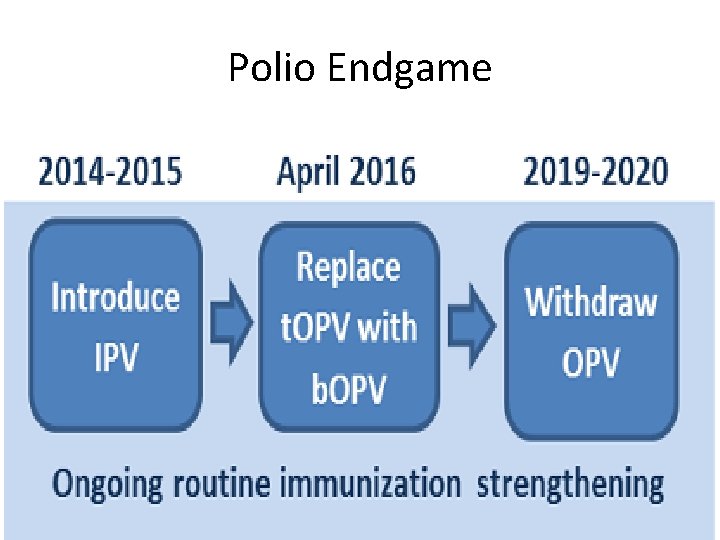
Polio Eradication Endgame Strengthening Routine Immunization • IPV introduction, 2014 -2015 • Worldwide t. OPV-b. OPV switch and withdrawal of m. OPV 2 stocks, April-May 2016 • All Polio vaccines type withdrawal, 2019 -2020

Polio Endgame

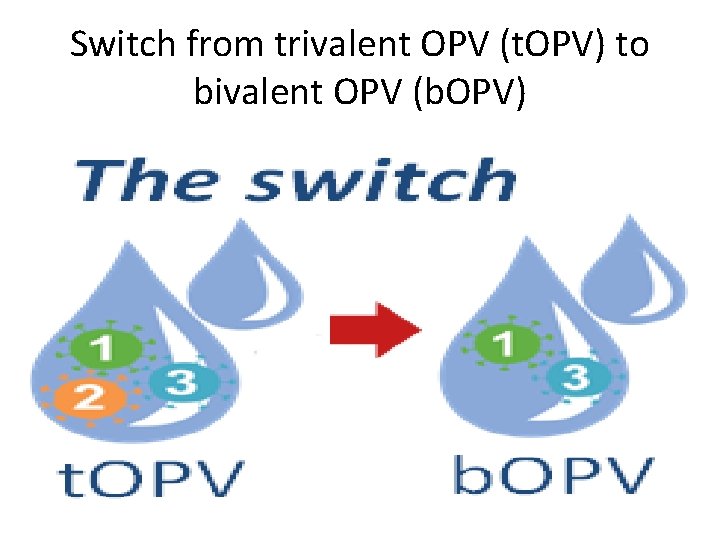
Switch from trivalent OPV (t. OPV) to bivalent OPV (b. OPV)

Rationale for m. OPV 2 withdrawal q. Last case of WILD Polio type 2 was in 1999 qthe eradication of Wild Polio Virus type 2 declared by Global Commission of the Certification of Polio (GCC) in 2015 qm. OPV 2/t. OPV stock were removed and destroyed worldwide, Switch, in April 2016 q. After switch, countries use b. OPV 1&3

t. OPV Withdrawal and Destruction

m. OPV 2 q. WHO prequalified qspecific for type 2 polio virus qadministrated orally q. Presented in 20 dose package vial q. Keep at -20 c or 2 -8 c q. Heat sensitive

m. OPV 2 However m. OPV is a unique in several ways: qhas the risk of Circulating Vaccine Derived Poliovirus type 2 (c. VDPV) qnot available at countries or markets after switch qavailable only in a global stockpile qm. OPV 2 importation required country’s acceptance and WHO DG release authorization based on recommendation of the Advisory Group

The Need to use m. OPV 2 qin response to c. VDPV 2 outbreak/event qthough m. OPV 2 can re-introduce live attenuated polio virus type 2 into population, it is the only vaccine that can mitigate and stop c. VDPV 2 outbreak/event qa strict vaccine supply management protocol must be seriously taken to prevent polio 2 virus reintroduction

m. OPV 2 Supply Management Protocol Strict supply chain managment at all stages: I. Release II. Handling III. Withdrawal IV. Validation

m. OPV 2 Supply Management Protocol I. Release ü Confirmed outbreak/event ü Country fill a special request form and signed by high level MOH ü Advisory Group approval ü WHO DG authorization ü Supplied to countries by UNICEF

m. OPV 2 Supply Management Protocol II. Handling ü Country complete Vaccine Arrival Report (VAR), submit the VAR to UNICEF within 24 hours ü Label the vaccine clearly visible ü Store and transport the vaccine separately from other vaccine in the cold rooms ü Supply to outbreak zones in separate, clearly identified cold chain with frozen icepacks ü During campaign vaccines must be put in zipped plastic bags inside the vaccine carriers

m. OPV 2 Supply Management Protocol III. Withdrawal/Removal At the end of each vaccination round ü All open and unopened vaccine vials must be returned to health facility or higher level ü All open vials must be destroyed completely at facility or higher level ü All unopened vials must be stored safely in reliable cold chain (prefer at central warehouse) follow the same protocol mentioned in transport

m. OPV 2 Supply Management Protocol IV. Removal Validation ü Wait for OBRA decision to destroy unopened vials and validation ü Provide full documentation on the number of vials used ü Conduct final validation to ensure that m. OPV 2 removed from the country endorsed by entrusted body üReturn to ZERO stock

Resources Available Online Access resources on the GPEI website: http: //polioeradication. org/tools-and-library/ 15

CCL/VM During Polio SIAs E-Learning Modules Open-access FREE interactive e-learning course complements the GPEI/UNICEF Guidance note on CCL & VM during polio SIAs, available in both English and French. Course objectives include: • planning and estimating vaccine needs for SIAs • cold chain management • monitoring vaccine quality issues 16

THANK YOU