Supplementary Table S 1 InclusionExclusion table Study design
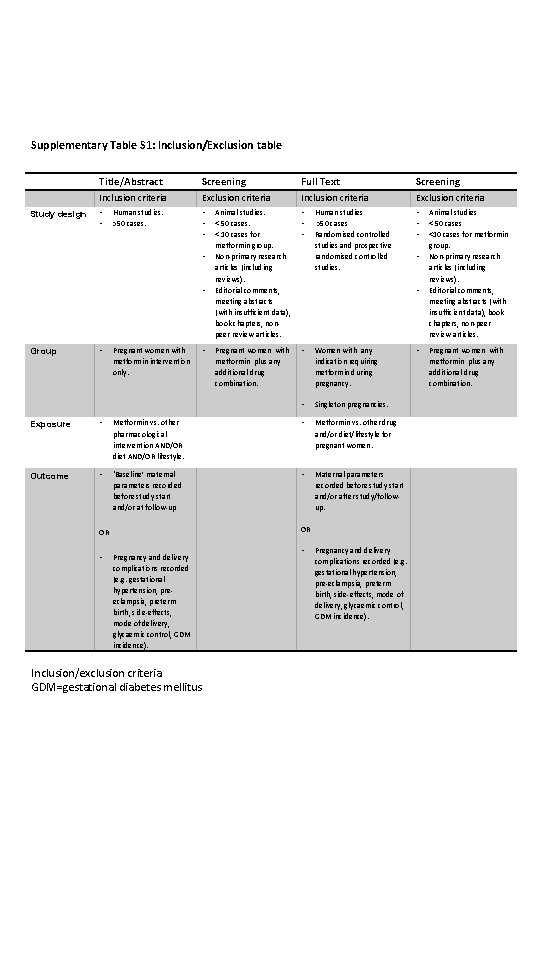
Supplementary Table S 1: Inclusion/Exclusion table Study design Title/Abstract Screening Full Text Screening Inclusion criteria Exclusion criteria • • • Animal studies. < 50 cases. < 10 cases for metformin group. Non-primary research articles (including reviews). Editorial comments, meeting abstracts (with insufficient data), book chapters, nonpeer review articles. • • • Pregnant women with metformin plus any additional drug combination. • Women with any indication requiring metformin during pregnancy. • Singleton pregnancies. Human studies. >50 cases. • • Group • Pregnant women with metformin intervention only. • Human studies >50 cases Randomised controlled studies and prospective randomised controlled studies. • Exposure • Metformin vs. other pharmacological intervention AND/OR diet AND/OR lifestyle. • Metformin vs. other drug and/or diet/lifestyle for pregnant women. Outcome • ‘Baseline’ maternal parameters recorded before study start and/or at follow-up • Maternal parameters recorded before study start and/or after study/followup. OR OR • Pregnancy and delivery complications recorded (e. g. gestational hypertension, preeclampsia, preterm birth, side-effects, mode of delivery, glycaemic control, GDM incidence). Inclusion/exclusion criteria GDM=gestational diabetes mellitus • • Pregnancy and delivery complications recorded (e. g. gestational hypertension, pre-eclampsia, preterm birth, side-effects, mode of delivery, glycaemic control, GDM incidence). • Animal studies < 50 cases <10 cases for metformin group. Non-primary research articles (including reviews). Editorial comments, meeting abstracts (with insufficient data), book chapters, non-peer review articles. Pregnant women with metformin plus any additional drug combination.
- Slides: 1