Supplementary information for Synthetic Receptors for Biomolecules Design

Supplementary information for Synthetic Receptors for Biomolecules: Design Principles and Applications © The Royal Society of Chemistry 2015 Chapter 5. Synthetic Receptors for Carbohydrates Alexander Draganov, a Danzhu Wang, a Jalisa Holmes, a Kaili Ji, a Binghe Wang, a* Scott K. Shaw, b and Bradley D. Smithb* a. Department b. Department of Chemistry and Center for Diagnostics and Therapeutics, Georgia State University, Atlanta, Georgia 30303, USA of Chemistry and Biochemistry, 236 Nieuwland Science Hall, University of Notre Dame, 46556 IN, USA *Email: smith. 115@nd. edu, wang@gsu. edu
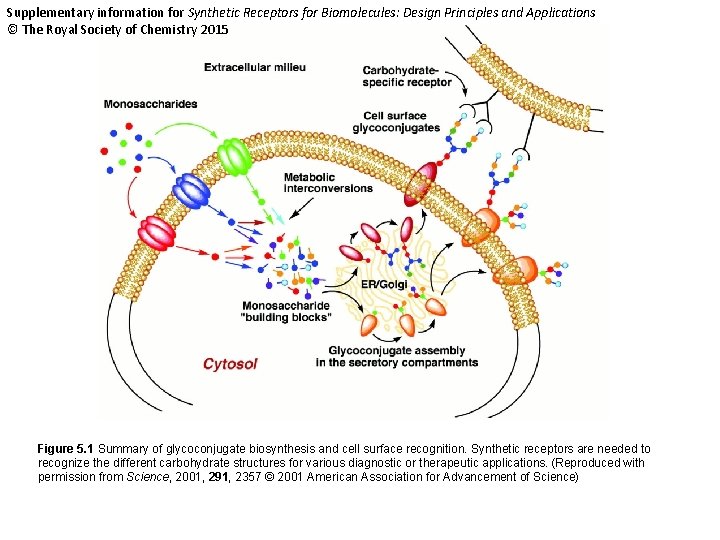
Supplementary information for Synthetic Receptors for Biomolecules: Design Principles and Applications © The Royal Society of Chemistry 2015 Figure 5. 1 Summary of glycoconjugate biosynthesis and cell surface recognition. Synthetic receptors are needed to recognize the different carbohydrate structures for various diagnostic or therapeutic applications. (Reproduced with permission from Science, 2001, 291, 2357 © 2001 American Association for Advancement of Science)
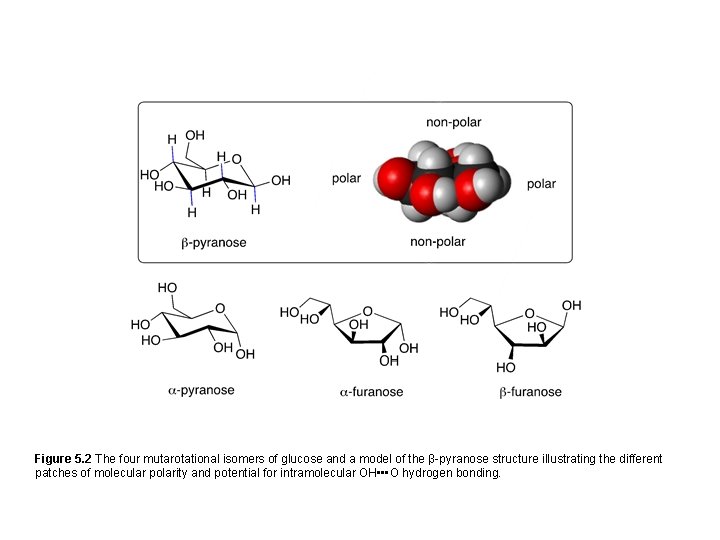
Figure 5. 2 The four mutarotational isomers of glucose and a model of the β-pyranose structure illustrating the different patches of molecular polarity and potential for intramolecular OH • • • O hydrogen bonding.
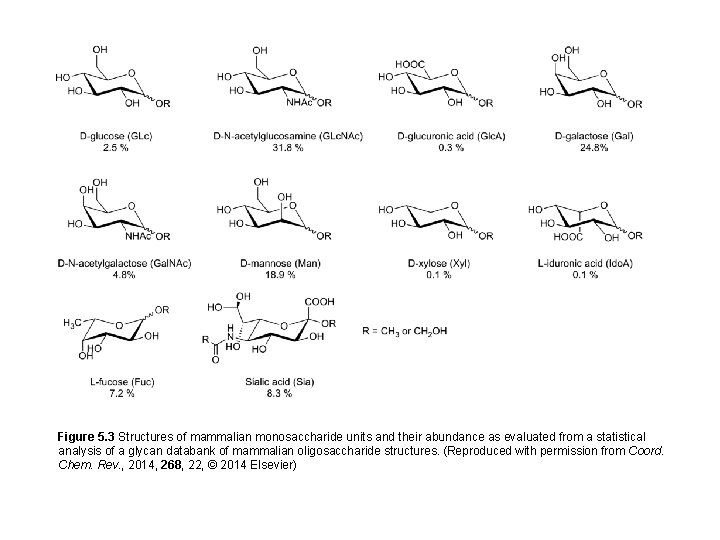
Figure 5. 3 Structures of mammalian monosaccharide units and their abundance as evaluated from a statistical analysis of a glycan databank of mammalian oligosaccharide structures. (Reproduced with permission from Coord. Chem. Rev. , 2014, 268, 22, © 2014 Elsevier)
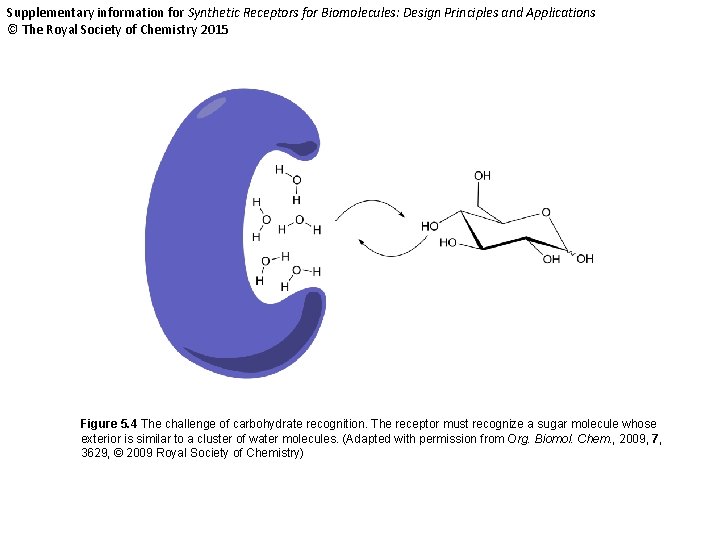
Supplementary information for Synthetic Receptors for Biomolecules: Design Principles and Applications © The Royal Society of Chemistry 2015 Figure 5. 4 The challenge of carbohydrate recognition. The receptor must recognize a sugar molecule whose exterior is similar to a cluster of water molecules. (Adapted with permission from Org. Biomol. Chem. , 2009, 7, 3629, © 2009 Royal Society of Chemistry)
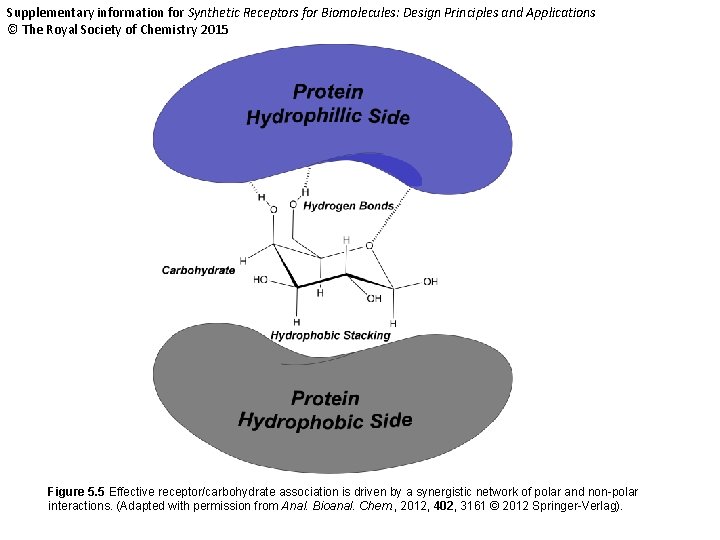
Supplementary information for Synthetic Receptors for Biomolecules: Design Principles and Applications © The Royal Society of Chemistry 2015 Figure 5. 5 Effective receptor/carbohydrate association is driven by a synergistic network of polar and non-polar interactions. (Adapted with permission from Anal. Bioanal. Chem. , 2012, 402, 3161 © 2012 Springer-Verlag).
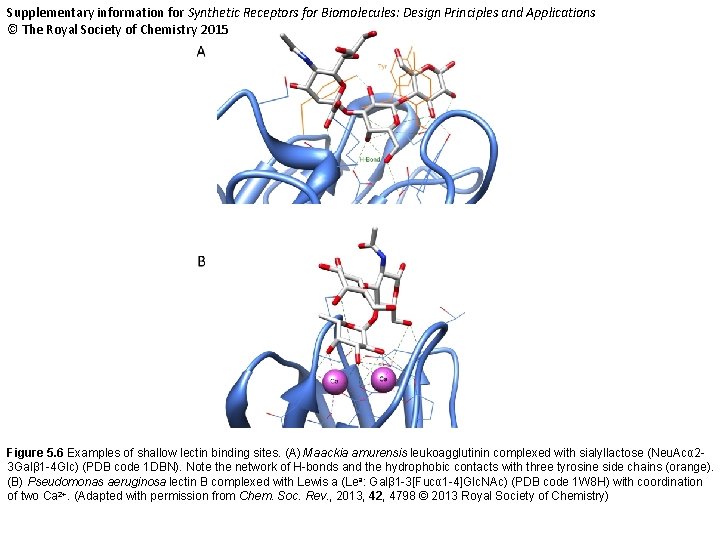
Supplementary information for Synthetic Receptors for Biomolecules: Design Principles and Applications © The Royal Society of Chemistry 2015 Figure 5. 6 Examples of shallow lectin binding sites. (A) Maackia amurensis leukoagglutinin complexed with sialyllactose (Neu. Acα 23 Galβ 1 -4 Glc) (PDB code 1 DBN). Note the network of H-bonds and the hydrophobic contacts with three tyrosine side chains (orange). (B) Pseudomonas aeruginosa lectin B complexed with Lewis a (Lea: Galβ 1 -3[Fucα 1 -4]Glc. NAc) (PDB code 1 W 8 H) with coordination of two Ca 2+. (Adapted with permission from Chem. Soc. Rev. , 2013, 42, 4798 © 2013 Royal Society of Chemistry)
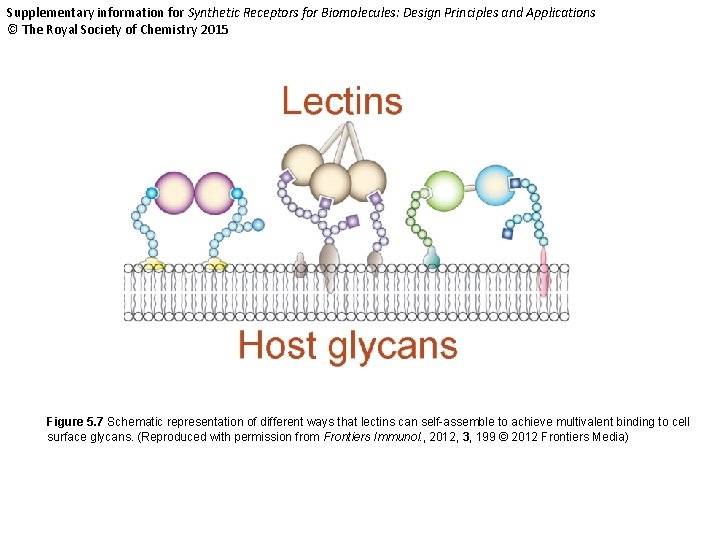
Supplementary information for Synthetic Receptors for Biomolecules: Design Principles and Applications © The Royal Society of Chemistry 2015 Figure 5. 7 Schematic representation of different ways that lectins can self-assemble to achieve multivalent binding to cell surface glycans. (Reproduced with permission from Frontiers Immunol. , 2012, 3, 199 © 2012 Frontiers Media)
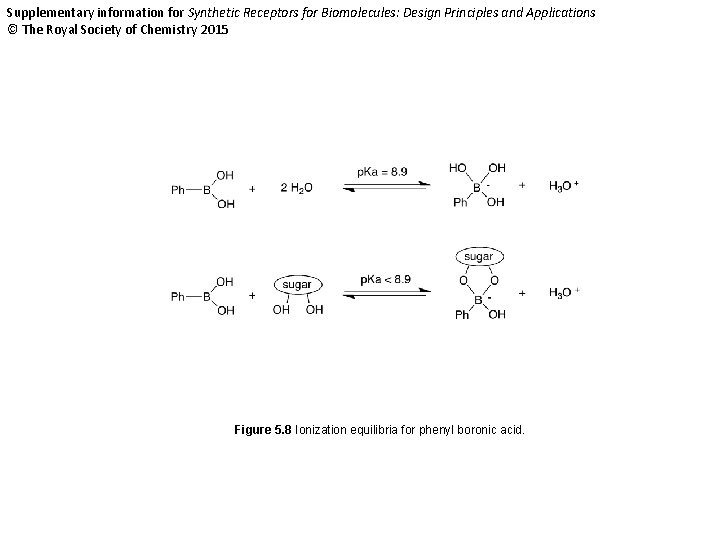
Supplementary information for Synthetic Receptors for Biomolecules: Design Principles and Applications © The Royal Society of Chemistry 2015 Figure 5. 8 Ionization equilibria for phenyl boronic acid.
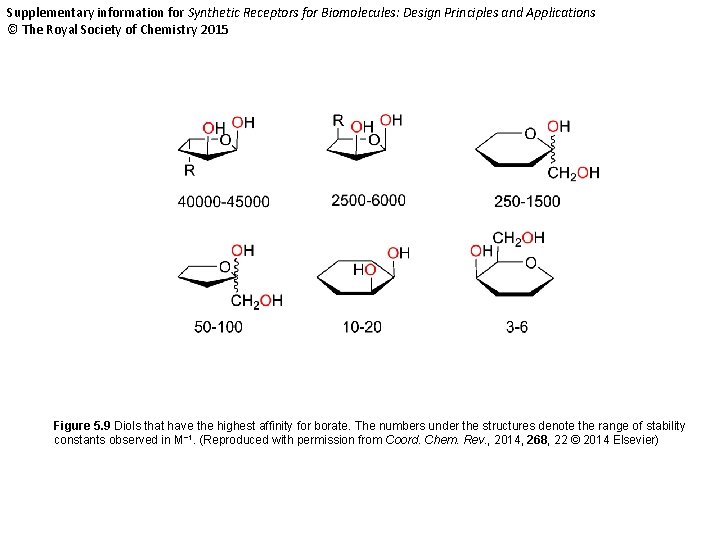
Supplementary information for Synthetic Receptors for Biomolecules: Design Principles and Applications © The Royal Society of Chemistry 2015 Figure 5. 9 Diols that have the highest affinity for borate. The numbers under the structures denote the range of stability constants observed in M− 1. (Reproduced with permission from Coord. Chem. Rev. , 2014, 268, 22 © 2014 Elsevier)
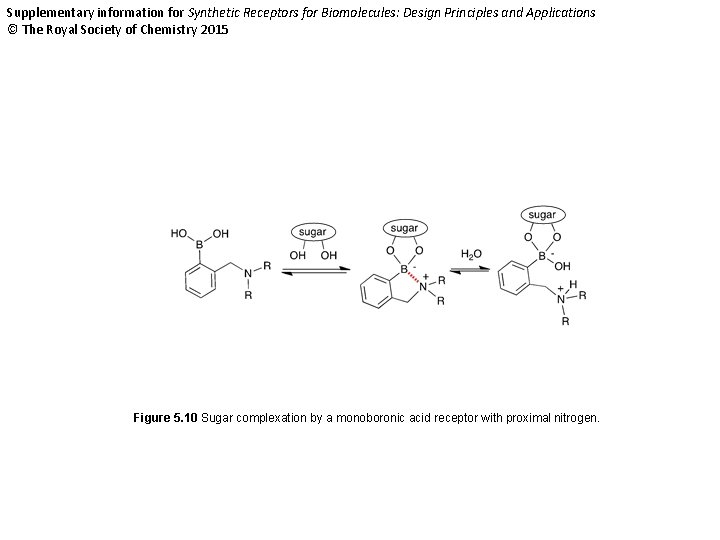
Supplementary information for Synthetic Receptors for Biomolecules: Design Principles and Applications © The Royal Society of Chemistry 2015 Figure 5. 10 Sugar complexation by a monoboronic acid receptor with proximal nitrogen.
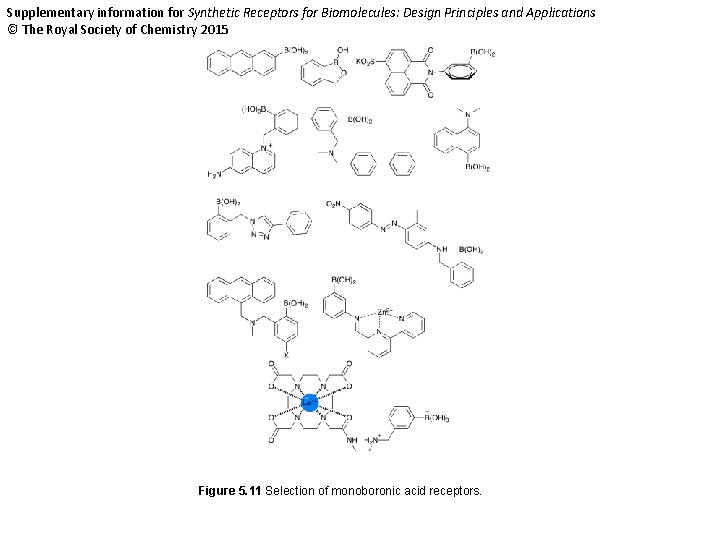
Supplementary information for Synthetic Receptors for Biomolecules: Design Principles and Applications © The Royal Society of Chemistry 2015 Figure 5. 11 Selection of monoboronic acid receptors.
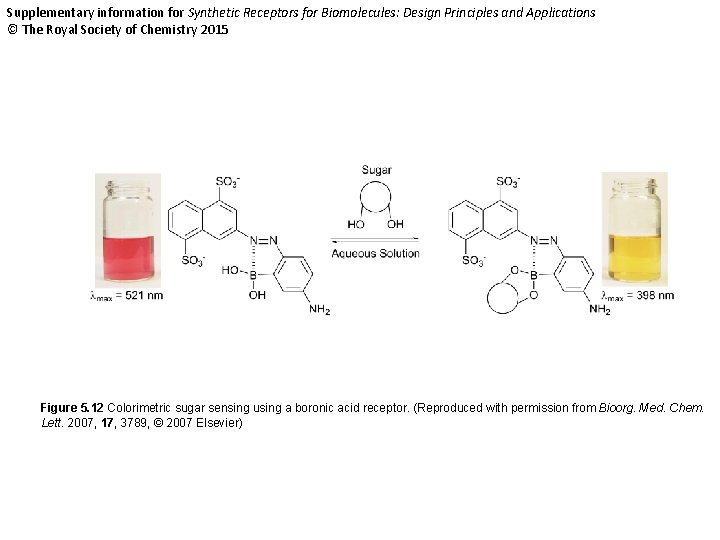
Supplementary information for Synthetic Receptors for Biomolecules: Design Principles and Applications © The Royal Society of Chemistry 2015 Figure 5. 12 Colorimetric sugar sensing using a boronic acid receptor. (Reproduced with permission from Bioorg. Med. Chem. Lett. 2007, 17, 3789, © 2007 Elsevier)
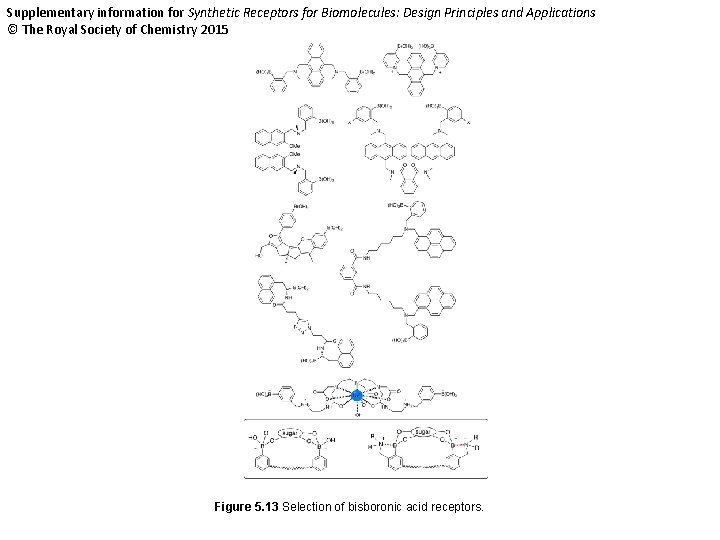
Supplementary information for Synthetic Receptors for Biomolecules: Design Principles and Applications © The Royal Society of Chemistry 2015 Figure 5. 13 Selection of bisboronic acid receptors.
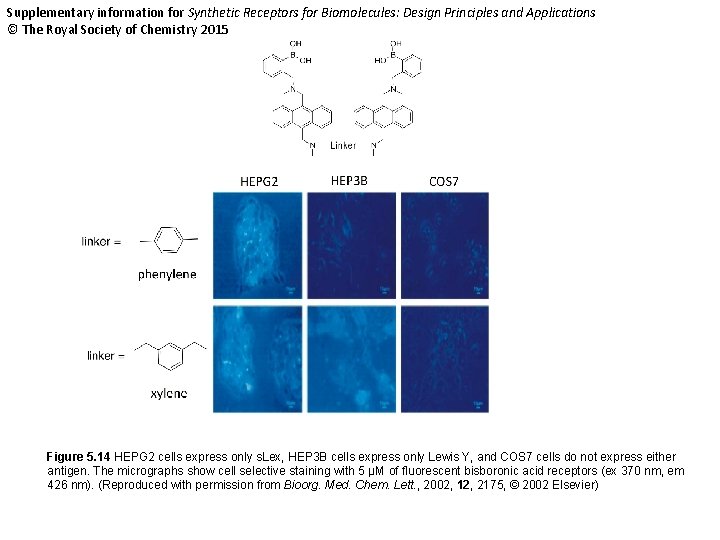
Supplementary information for Synthetic Receptors for Biomolecules: Design Principles and Applications © The Royal Society of Chemistry 2015 Figure 5. 14 HEPG 2 cells express only s. Lex, HEP 3 B cells express only Lewis Y, and COS 7 cells do not express either antigen. The micrographs show cell selective staining with 5 μM of fluorescent bisboronic acid receptors (ex 370 nm, em 426 nm). (Reproduced with permission from Bioorg. Med. Chem. Lett. , 2002, 12, 2175, © 2002 Elsevier)
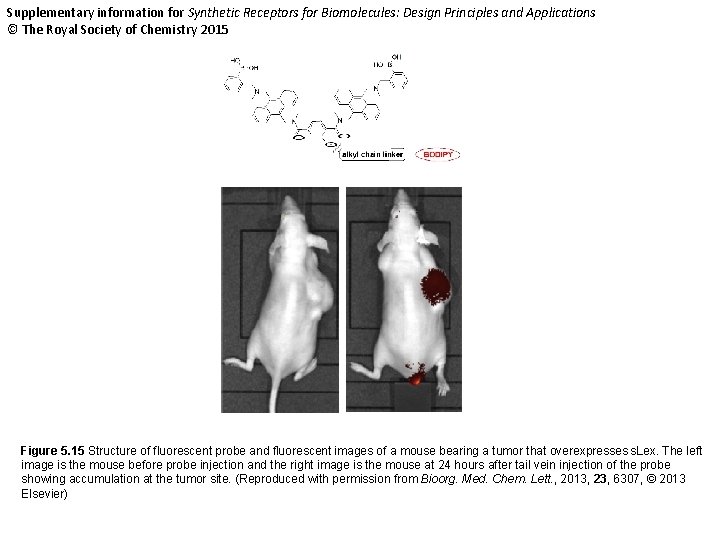
Supplementary information for Synthetic Receptors for Biomolecules: Design Principles and Applications © The Royal Society of Chemistry 2015 Figure 5. 15 Structure of fluorescent probe and fluorescent images of a mouse bearing a tumor that overexpresses s. Lex. The left image is the mouse before probe injection and the right image is the mouse at 24 hours after tail vein injection of the probe showing accumulation at the tumor site. (Reproduced with permission from Bioorg. Med. Chem. Lett. , 2013, 23, 6307, © 2013 Elsevier)
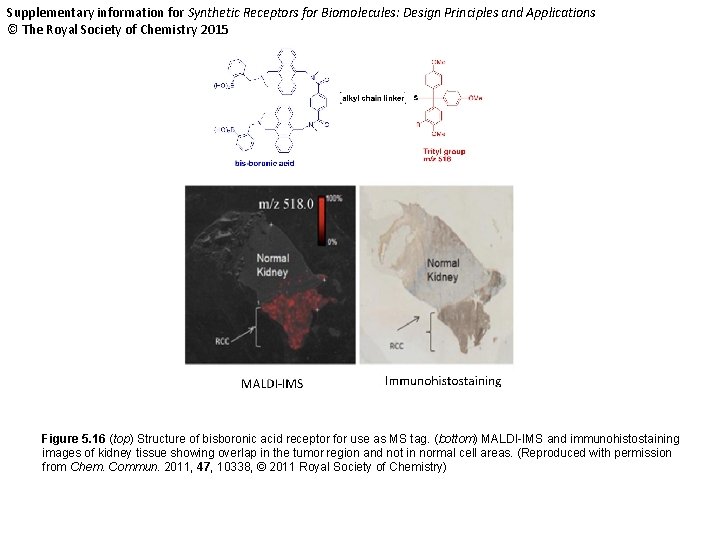
Supplementary information for Synthetic Receptors for Biomolecules: Design Principles and Applications © The Royal Society of Chemistry 2015 Figure 5. 16 (top) Structure of bisboronic acid receptor for use as MS tag. (bottom) MALDI-IMS and immunohistostaining images of kidney tissue showing overlap in the tumor region and not in normal cell areas. (Reproduced with permission from Chem. Commun. 2011, 47, 10338, © 2011 Royal Society of Chemistry)
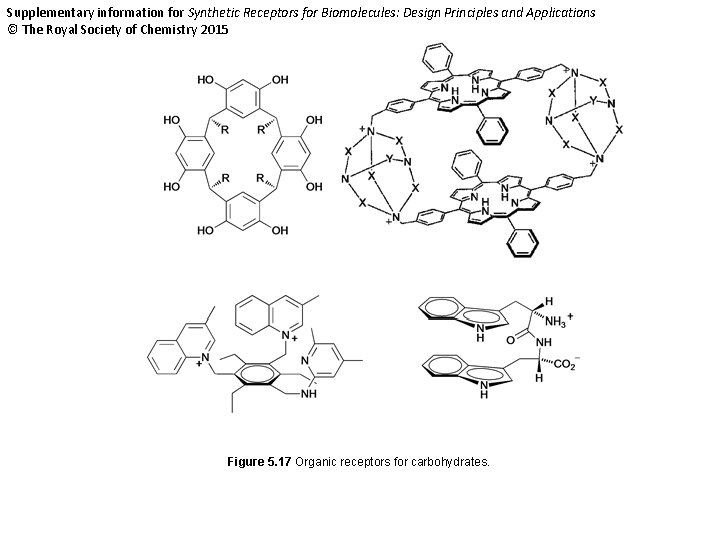
Supplementary information for Synthetic Receptors for Biomolecules: Design Principles and Applications © The Royal Society of Chemistry 2015 Figure 5. 17 Organic receptors for carbohydrates.
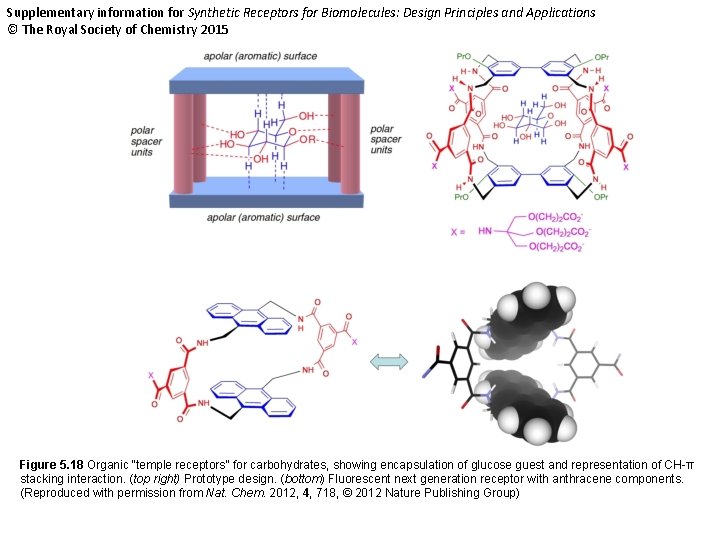
Supplementary information for Synthetic Receptors for Biomolecules: Design Principles and Applications © The Royal Society of Chemistry 2015 Figure 5. 18 Organic “temple receptors” for carbohydrates, showing encapsulation of glucose guest and representation of CH-π stacking interaction. (top right) Prototype design. (bottom) Fluorescent next generation receptor with anthracene components. (Reproduced with permission from Nat. Chem. 2012, 4, 718, © 2012 Nature Publishing Group)
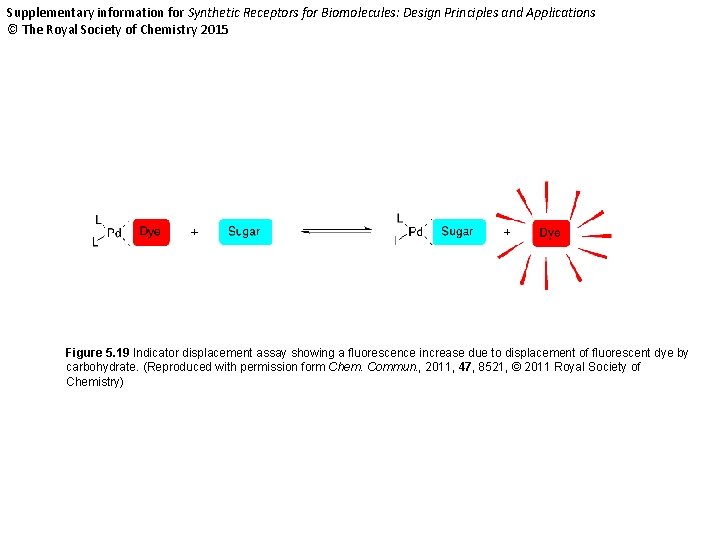
Supplementary information for Synthetic Receptors for Biomolecules: Design Principles and Applications © The Royal Society of Chemistry 2015 Figure 5. 19 Indicator displacement assay showing a fluorescence increase due to displacement of fluorescent dye by carbohydrate. (Reproduced with permission form Chem. Commun. , 2011, 47, 8521, © 2011 Royal Society of Chemistry)
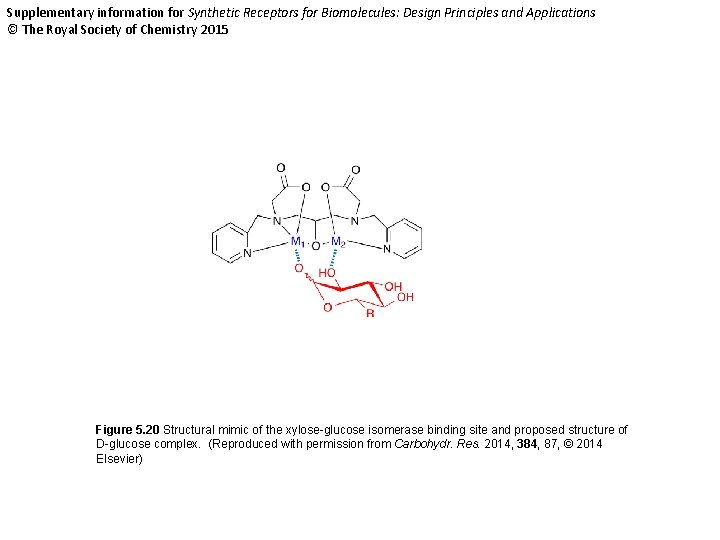
Supplementary information for Synthetic Receptors for Biomolecules: Design Principles and Applications © The Royal Society of Chemistry 2015 Figure 5. 20 Structural mimic of the xylose-glucose isomerase binding site and proposed structure of D-glucose complex. (Reproduced with permission from Carbohydr. Res. 2014, 384, 87, © 2014 Elsevier)
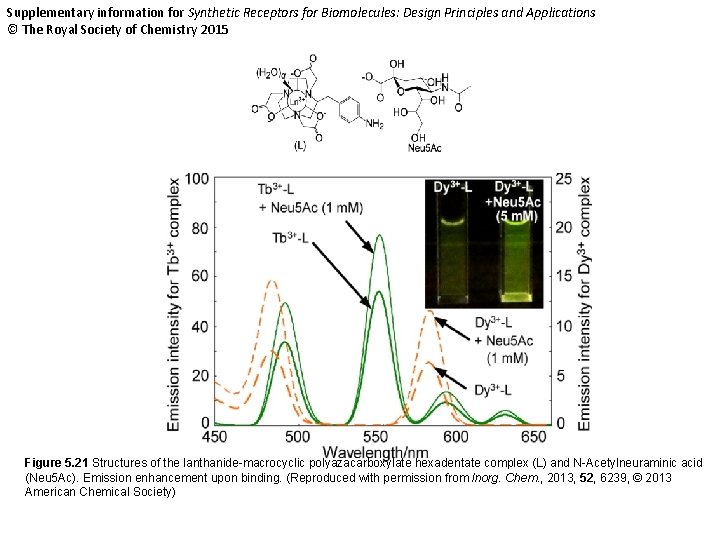
Supplementary information for Synthetic Receptors for Biomolecules: Design Principles and Applications © The Royal Society of Chemistry 2015 Figure 5. 21 Structures of the lanthanide-macrocyclic polyazacarboxylate hexadentate complex (L) and N-Acetylneuraminic acid (Neu 5 Ac). Emission enhancement upon binding. (Reproduced with permission from Inorg. Chem. , 2013, 52, 6239, © 2013 American Chemical Society)
- Slides: 22