Supplemental Figure 1 B AM Probe M k
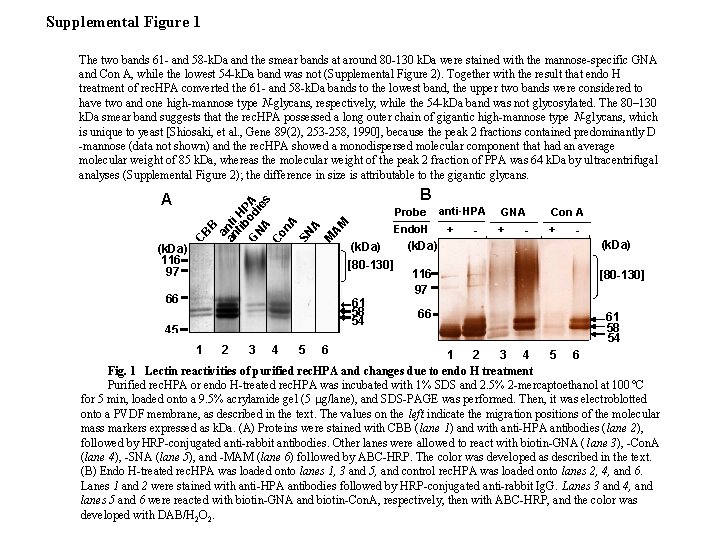
Supplemental Figure 1 B AM Probe M (k. Da) 116 97 CB A B an an ti tib HP G od A NA ie s Co n. A SN A The two bands 61 - and 58 -k. Da and the smear bands at around 80 -130 k. Da were stained with the mannose-specific GNA and Con A, while the lowest 54 -k. Da band was not (Supplemental Figure 2). Together with the result that endo H treatment of rec. HPA converted the 61 - and 58 -k. Da bands to the lowest band, the upper two bands were considered to have two and one high-mannose type N-glycans, respectively, while the 54 -k. Da band was not glycosylated. The 80– 130 k. Da smear band suggests that the rec. HPA possessed a long outer chain of gigantic high-mannose type N-glycans, which is unique to yeast [Shiosaki, et al. , Gene 89(2), 253 -258, 1990], because the peak 2 fractions contained predominantly D -mannose (data not shown) and the rec. HPA showed a monodispersed molecular component that had an average molecular weight of 85 k. Da, whereas the molecular weight of the peak 2 fraction of PPA was 64 k. Da by ultracentrifugal analyses (Supplemental Figure 2); the difference in size is attributable to the gigantic glycans. Endo. H (k. Da) [80 -130] 66 61 58 54 45 1 2 3 4 5 6 (k. Da) 116 97 66 anti- HPA + - GNA + - Con A + (k. Da) [80 -130] 61 58 54 1 2 3 4 5 6 Fig. 1 Lectin reactivities of purified rec. HPA and changes due to endo H treatment Purified rec. HPA or endo H-treated rec. HPA was incubated with 1% SDS and 2. 5% 2 -mercaptoethanol at 100 ºC for 5 min, loaded onto a 9. 5% acrylamide gel (5 mg/lane), and SDS-PAGE was performed. Then, it was electroblotted onto a PVDF membrane, as described in the text. The values on the left indicate the migration positions of the molecular mass markers expressed as k. Da. (A) Proteins were stained with CBB (lane 1) and with anti-HPA antibodies (lane 2), followed by HRP-conjugated anti-rabbit antibodies. Other lanes were allowed to react with biotin-GNA ( lane 3), -Con. A (lane 4), -SNA (lane 5), and -MAM (lane 6) followed by ABC-HRP. The color was developed as described in the text. (B) Endo H-treated rec. HPA was loaded onto lanes 1, 3 and 5, and control rec. HPA was loaded onto lanes 2, 4, and 6. Lanes 1 and 2 were stained with anti-HPA antibodies followed by HRP-conjugated anti-rabbit Ig. G. Lanes 3 and 4, and lanes 5 and 6 were reacted with biotin-GNA and biotin-Con. A, respectively, then with ABC-HRP, and the color was developed with DAB/H 2 O 2.
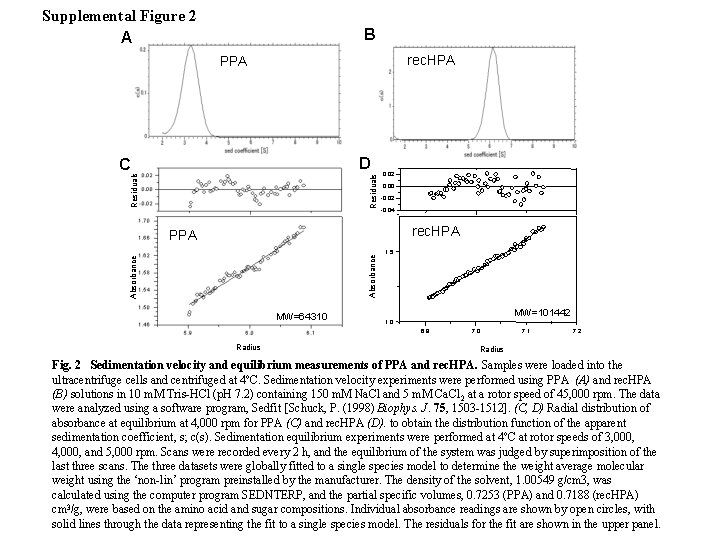
Supplemental Figure 2 A B rec. HPA PPA D Residuals C rec. HPA 0. 02 0. 00 -0. 02 -0. 04 rec. HPA Absorbance PPA MW=64310 1. 5 MW=101442 1. 0 6. 9 Radius 7. 0 7. 1 7. 2 Radius Fig. 2 Sedimentation velocity and equilibrium measurements of PPA and rec. HPA. Samples were loaded into the ultracentrifuge cells and centrifuged at 4ºC. Sedimentation velocity experiments were performed using PPA (A) and rec. HPA (B) solutions in 10 m. M Tris-HCl (p. H 7. 2) containing 150 m. M Na. Cl and 5 m. M Ca. Cl 2 at a rotor speed of 45, 000 rpm. The data were analyzed using a software program, Sedfit [Schuck, P. (1998) Biophys. J. 75, 1503 -1512]. (C, D) Radial distribution of absorbance at equilibrium at 4, 000 rpm for PPA (C) and rec. HPA (D). to obtain the distribution function of the apparent sedimentation coefficient, s; c(s). Sedimentation equilibrium experiments were performed at 4ºC at rotor speeds of 3, 000, 4, 000, and 5, 000 rpm. Scans were recorded every 2 h, and the equilibrium of the system was judged by superimposition of the last three scans. The three datasets were globally fitted to a single species model to determine the weight average molecular weight using the ‘non-lin’ program preinstalled by the manufacturer. The density of the solvent, 1. 00549 g/cm 3, was calculated using the computer program SEDNTERP, and the partial specific volumes, 0. 7253 (PPA) and 0. 7188 (rec. HPA) cm 3/g, were based on the amino acid and sugar compositions. Individual absorbance readings are shown by open circles, with solid lines through the data representing the fit to a single species model. The residuals for the fit are shown in the upper panel.
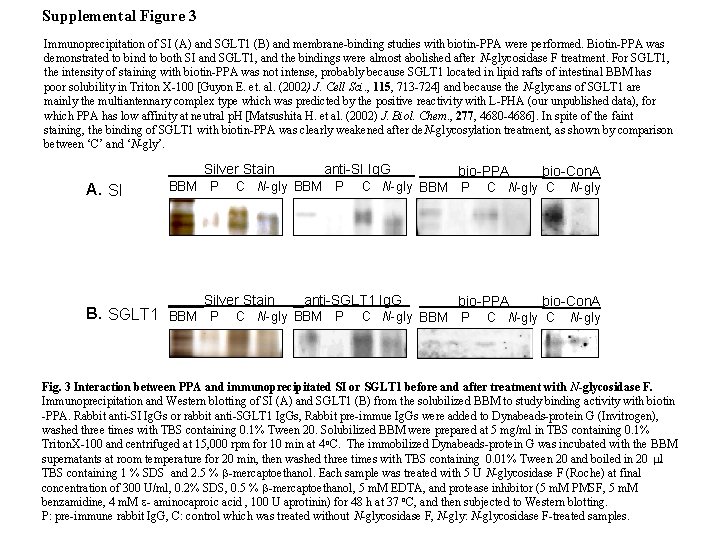
Supplemental Figure 3 Immunoprecipitation of SI (A) and SGLT 1 (B) and membrane-binding studies with biotin-PPA were performed. Biotin-PPA was demonstrated to bind to both SI and SGLT 1, and the bindings were almost abolished after N-glycosidase F treatment. For SGLT 1, the intensity of staining with biotin-PPA was not intense, probably because SGLT 1 located in lipid rafts of intestinal BBM has poor solubility in Triton X-100 [Guyon E. et. al. (2002) J. Cell Sci. , 115, 713 -724] and because the N-glycans of SGLT 1 are mainly the multiantennary complex type which was predicted by the positive reactivity with L-PHA (our unpublished data), for which PPA has low affinity at neutral p. H [Matsushita H. et al. (2002) J. Biol. Chem. , 277, 4680 -4686]. In spite of the faint staining, the binding of SGLT 1 with biotin-PPA was clearly weakened after de. N-glycosylation treatment, as shown by comparison between ‘C’ and ‘N-gly’. Silver Stain A. SI BBM P C N-gly BBM P Silver Stain B. SGLT 1 BBM P anti-SI Ig. G C N-gly BBM P anti-SGLT 1 Ig. G C N-gly BBM P bio-PPA C N-gly BBM P bio-Con. A C N-gly Fig. 3 Interaction between PPA and immunoprecipitated SI or SGLT 1 before and after treatment with N-glycosidase F. Immunoprecipitation and Western blotting of SI (A) and SGLT 1 (B) from the solubilized BBM to study binding activity with biotin -PPA. Rabbit anti-SI Ig. Gs or rabbit anti-SGLT 1 Ig. Gs, Rabbit pre-immue Ig. Gs were added to Dynabeads-protein G (Invitrogen), washed three times with TBS containing 0. 1% Tween 20. Solubilized BBM were prepared at 5 mg/ml in TBS containing 0. 1% Triton. X-100 and centrifuged at 15, 000 rpm for 10 min at 4 o. C. The immobilized Dynabeads-protein G was incubated with the BBM supernatants at room temperature for 20 min, then washed three times with TBS containing 0. 01% Tween 20 and boiled in 20 ml TBS containing 1 % SDS and 2. 5 % b-mercaptoethanol. Each sample was treated with 5 U N-glycosidase F (Roche) at final concentration of 300 U/ml, 0. 2% SDS, 0. 5 % b-mercaptoethanol, 5 m. M EDTA, and protease inhibitor (5 m. M PMSF, 5 m. M benzamidine, 4 m. M e- aminocaproic acid , 100 U aprotinin) for 48 h at 37 o. C, and then subjected to Western blotting. P: pre-immune rabbit Ig. G, C: control which was treated without N-glycosidase F, N-gly: N-glycosidase F-treated samples.
- Slides: 3