Supplement 155 Imaging Reports using HL 7 CDA
























- Slides: 34

Supplement 155 Imaging Reports using HL 7 CDA Status Report to WG-06 2014/04/01 Harry Solomon

Goals • Foundational architecture for an evolving family of imaging reports • Initial templates for radiology reports, aligned with RSNA Rad. Lex and Reporting Initiative • Primarily narrative, with some discrete data; evolving to more discrete data over time • Diagnostic and screening; evolving to interventional, cardiac, anatomic path, etc. 4/1/2014 Sup 155 review 2

HL 7 Clinical Document Architecture • The scope of the CDA is the standardization of clinical documents for exchange. • A clinical document is a record of observations and other services with the following characteristics: – Persistence – Stewardship – Potential for authentication – Wholeness – Human readability

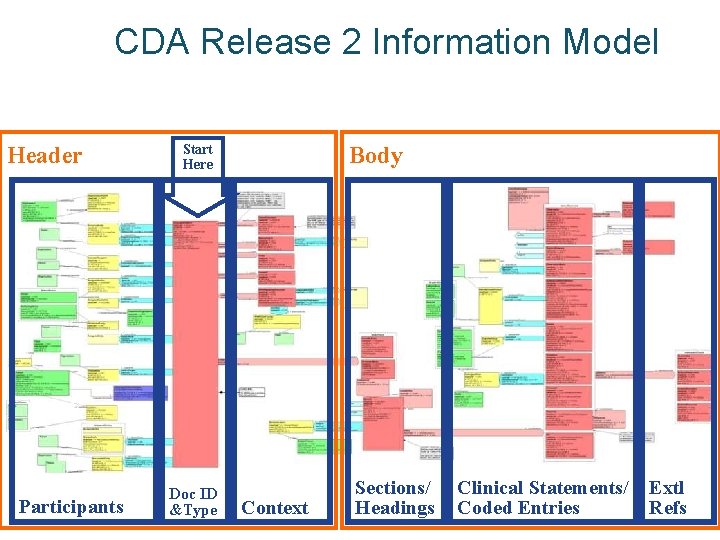
Key Aspects of the CDA documents are encoded in Extensible Markup Language (XML) CDA documents derive their meaning from the HL 7 v 3 Reference Information Model (RIM ) and use HL 7 v 3 Data Types A CDA document consists of a header and a body • Header is consistent across all clinical documents - identifies and classifies the document, provides information on patient, provider, encounter, and authentication; allows document management, compilation of an individual patient's clinical documents into an electronic patient record • Body contains narrative text / multimedia content (level 1), optionally structured into sections with coded titles and tagged narrative content (level 2), optionally augmented by coded equivalents to narrative (level 3) CDA documents for a particular use may be constrained by a Template 4/ Harry Solomon / Module 3 -1 - CDA/

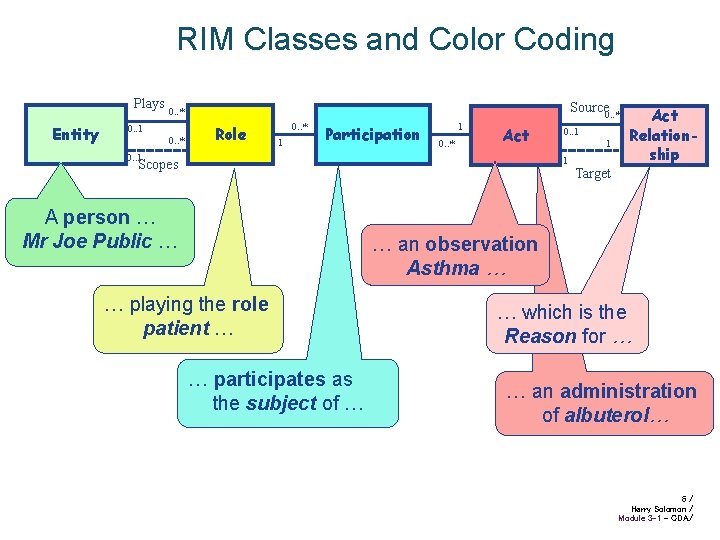
Core concepts of RIM Every happening is an Act • Procedure, observation, medication, supply, registration, etc. Acts are related through an Act. Relationship • composition, preconditions, revisions, support, etc. Participation defines involvement in an Act • author, performer, subject, location, etc. The participants are Roles • patient, agent, responsible party. Roles are played and scoped by Entities • persons, organizations, material, places, devices, etc. 5/ Harry Solomon / Module 3 -1 - CDA/

RIM Classes and Color Coding Plays Entity Source 0. . * 0. . 1 0. . * Role 0. . * 1 Participation 1 0. . * Act 0. . 1 1 1 Scopes A person … Mr Joe Public … Act Relationship Target … an observation Asthma … … playing the role patient … … participates as the subject of … … which is the Reason for … … an administration of albuterol… 6/ Harry Solomon / Module 3 -1 - CDA/

CDA Release 2 Information Model Header Participants Start Here Doc ID &Type Body Context Sections/ Headings Clinical Statements/ Extl 7 / Harry. Refs Solomon / Coded Entries Module 3 -1 - CDA/

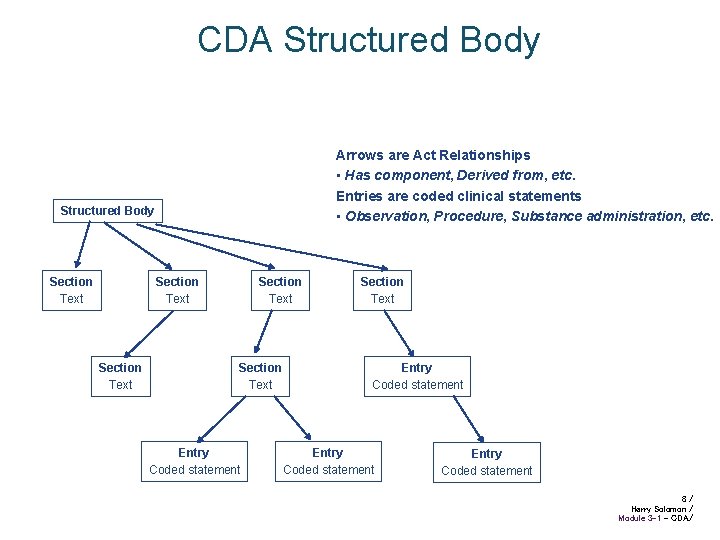
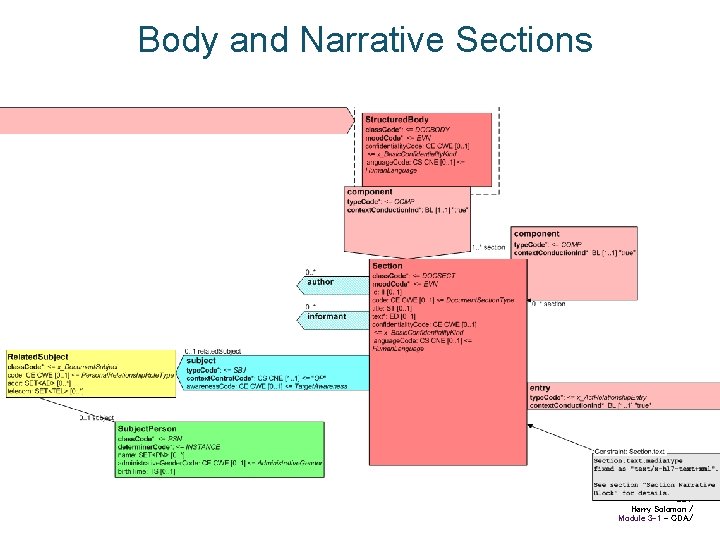
CDA Structured Body Arrows are Act Relationships • Has component, Derived from, etc. Entries are coded clinical statements • Observation, Procedure, Substance administration, etc. Structured Body Section Text Section Text Entry Coded statement 8/ Harry Solomon / Module 3 -1 - CDA/

Sample CDA 9/ Harry Solomon / Module 3 -1 - CDA/

Narrative and Coded Info CDA structured body requires human-readable “Narrative Block”, all that is needed to reproduce the legally attested clinical content CDA allows optional machine-readable coded “Entries”, which drive automated processes Narrative may be flagged as derived from Entries • Textual rendering of coded entries’ content, and contains no clinical content not derived from the entries General method for coding clinical statements is a hard, unsolved problem • CDA allows incremental improvement to amount of coded data without breaking the model 10 / Harry Solomon / Module 3 -1 - CDA/

Narrative and Coded Entry Example 11 / Harry Solomon / Module 3 -1 - CDA/

Purposes of templates • • Reduce variability, improve interoperability Normalize best practice Support automation of report production Support validation of report content 4/1/2014 Sup 155 review 12

Multiple layers of constraint Professional Content Specific content for specialized procedures DICOM (Sup 155) Additional requirements and templates for radiology reports CDA r 2 Refined Data Classes and structures for clinical documents HL 7 v 3 RIM Abstract Data Classes 4/1/2014 Sup 155 review 13

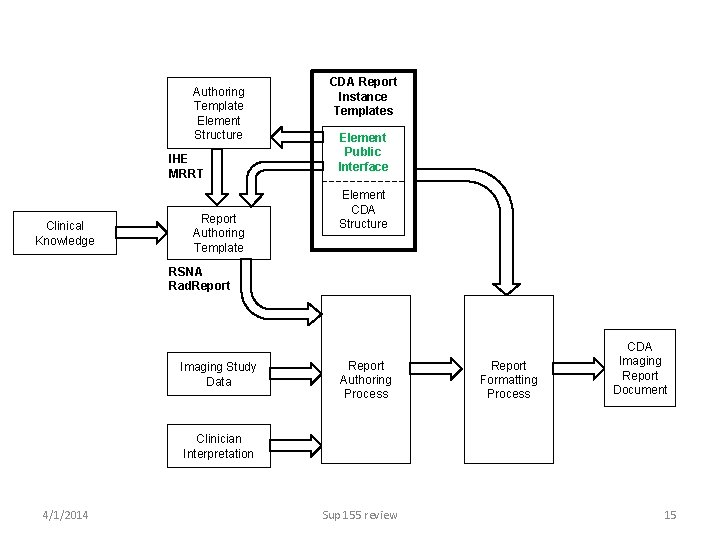
Sup 155 as green. CDA • green. CDA is the concept that CDA implementation can be simplified by abstractions bridging between use cases and CDA formal syntax requirements • Defines data elements, using “business names”, that can be invoked by professional content specifications, without knowledge of CDA structures • E. g. , professional content can specify “impression: text”, or “contrast: name” and “contrast: volume”, and Sup 155 specifies how/where those are instantiated in a CDA structure 4/1/2014 Sup 155 review 14

Authoring Template Element Structure IHE MRRT Clinical Knowledge Report Authoring Template CDA Report Instance Templates Element Public Interface Element CDA Structure RSNA Rad. Report Imaging Study Data Report Authoring Process Report Formatting Process CDA Imaging Report Document Clinician Interpretation 4/1/2014 Sup 155 review 15

Sup 155 editorial style • Following conventions of HL 7 CDA implementation guides, including Consolidated CDA, and emerging HL 7 Templates Standard (in ballot reconciliation) • Library of document, section, and entry templates • Templates specified as table aligned to XML, plus supplemental narrative conformance statements (conceptually similar to Part 3 module style) • XML examples provided • Not being generated from tooling (MDHT or Trifolia) 4/1/2014 Sup 155 review 16

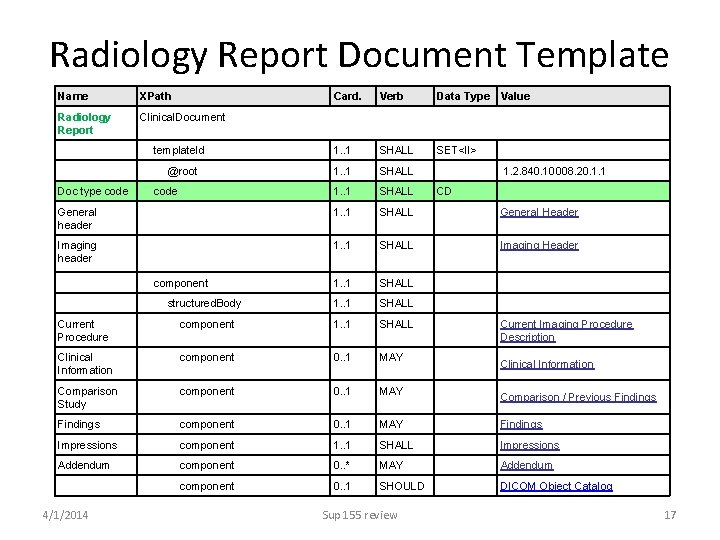
Radiology Report Document Template Name XPath Radiology Report Clinical. Document Card. Verb Data Type Value template. Id 1. . 1 SHALL SET<II> @root 1. . 1 SHALL 1. 2. 840. 10008. 20. 1. 1 1. . 1 SHALL CD Doc type code General header 1. . 1 SHALL General Header Imaging header 1. . 1 SHALL Imaging Header 1. . 1 SHALL structured. Body 1. . 1 SHALL Current Procedure component 1. . 1 SHALL Current Imaging Procedure Description Clinical Information component 0. . 1 MAY Comparison Study component 0. . 1 MAY Findings Impressions component 1. . 1 SHALL Impressions Addendum component 0. . * MAY Addendum component 0. . 1 SHOULD DICOM Object Catalog 4/1/2014 component Sup 155 review Clinical Information Comparison / Previous Findings 17

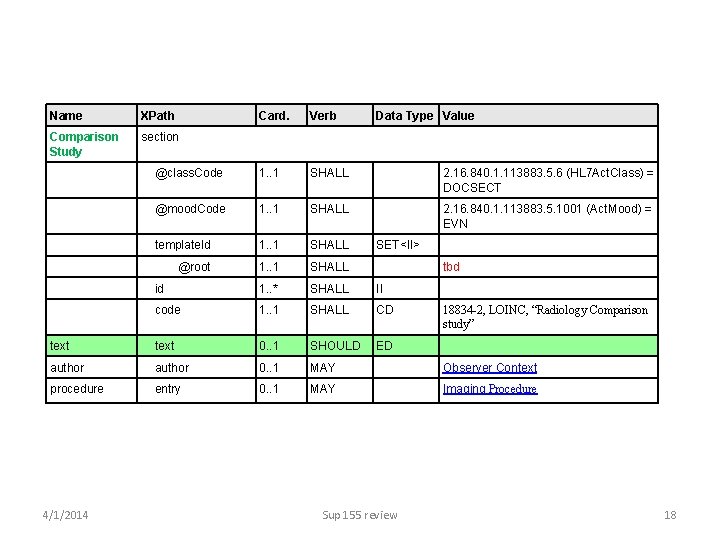
Name XPath Comparison Study section Card. Verb Data Type Value @class. Code 1. . 1 SHALL 2. 16. 840. 1. 113883. 5. 6 (HL 7 Act. Class) = DOCSECT @mood. Code 1. . 1 SHALL 2. 16. 840. 1. 113883. 5. 1001 (Act. Mood) = EVN template. Id 1. . 1 SHALL SET<II> @root 1. . 1 SHALL tbd id 1. . * SHALL II code 1. . 1 SHALL CD 18834 -2, LOINC, “Radiology Comparison study” text 0. . 1 SHOULD ED author 0. . 1 MAY Observer Context 0. . 1 MAY Imaging Procedure procedure 4/1/2014 entry Sup 155 review 18

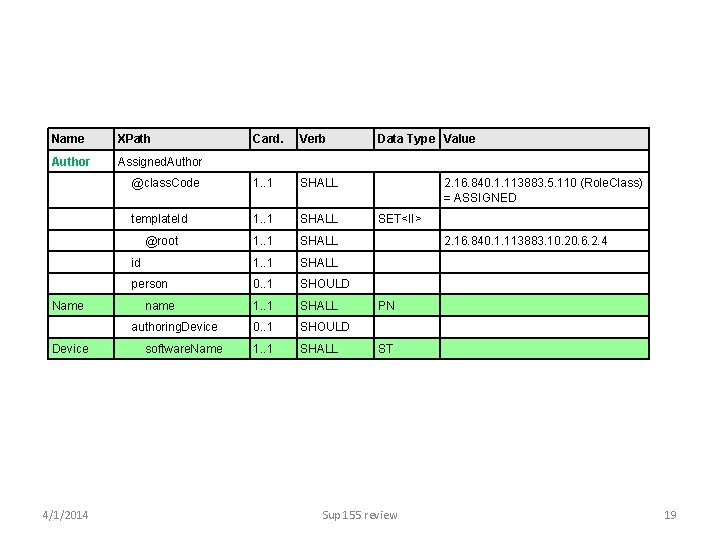
Name XPath Author Assigned. Author Card. Verb Data Type Value @class. Code 1. . 1 SHALL 2. 16. 840. 1. 113883. 5. 110 (Role. Class) = ASSIGNED template. Id 1. . 1 SHALL SET<II> @root 1. . 1 SHALL 2. 16. 840. 1. 113883. 10. 20. 6. 2. 4 id 1. . 1 SHALL person 0. . 1 SHOULD 1. . 1 SHALL PN 0. . 1 SHOULD 1. . 1 SHALL ST Name Device 4/1/2014 name authoring. Device software. Name Sup 155 review 19

Subsections • Some defined in templates for specific uses – Contrast, Radiation Dose, Fetus Finding • Requirements for user-labeled subsections – Content for an arbitrary topic (a particular organ or anatomic feature, a lesion, a tumor, etc. ) – Required section. title with topic name – No section. code, hence no semantic postcoordination to section entries 4/1/2014 Sup 155 review 20

Why no section. code in user-labeled subsection? • section. code uses LOINC codes with scale=NAR – i. e. , its value in section. text is a narrative block – A concept that scopes a value (observable entity) • The concepts used as user-labels (typically anatomy) are the wrong “part of speech” – Do not have formal semantics of scoping a value – Need something like a post-coordinated phrase: Findings. Section+Finding. Site=<anatomy> (cf. DICOM) – CDA does not have section target. Site. Code or Participation for anatomic site 4/1/2014 Sup 155 review 21

Body and Narrative Sections 22 / Harry Solomon / Module 3 -1 - CDA/

External Relations • HL 7 • RSNA – Radiology Reporting – Rad. Lex • IHE – MRRT

Relationship to HL 7 • Builds on HL 7 Diagnostic Imaging Report (DIR) CDA IG informative standard • Leverages harmonization done for Consolidated CDA IG r 2 (C-CDA) • Public comment to be solicited in HL 7 ballot cycle parallel to DICOM PC and LB periods • May be balloted as HL 7 standard (DIR r 2), recognizing updates will be handled in DICOM continuous maintenance process 4/1/2014 Sup 155 review 24

Reuse of C-CDA Templates • Intent is to be harmonized with C-CDA • But C-CDA is US Realm only – DICOM needs to be Universal Realm • Challenges to re-use based on shortcuts taken – Vocabulary bindings hard coded into templates – Vocabulary bindings US Realm only 4/1/2014 Sup 155 review 25

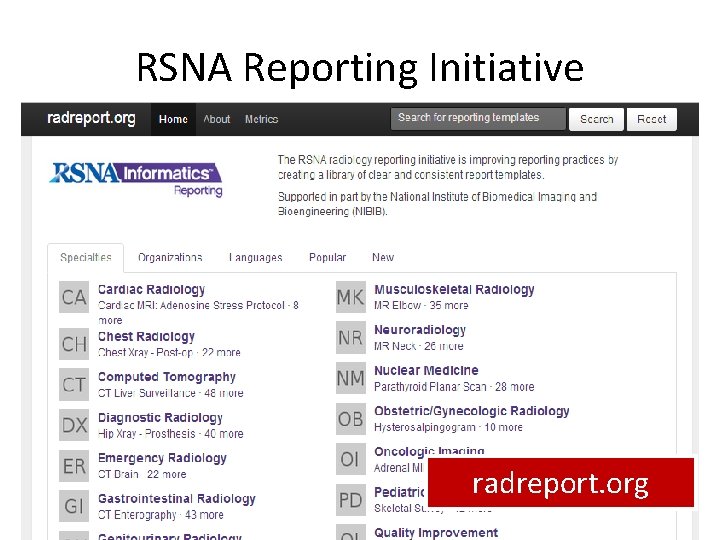
RSNA Reporting Initiative radreport. org 4/1/2014 Sup 155 review 26

CT Brain (example) Clinical History * Medical history * Risk factors * Allergies, if relevant * Reason for exam, including medical necessity: [headache | stroke | dizziness | trauma] Imaging Technique * Time of image acquisition * Imaging device [MR, CT] * Image acquisition parameters, such as device settings, patient positioning, interventions (e. g. , Valsalva) * Contrast materials and other medications administered (including name, dose, route, and time) * Radiation dose Comparison * Date and type of previous exams reviewed, if applicable Observations * Extra axial spaces: [normal in size and morphology for the patient's age* | widened] * Hemorrhage: [none* ; subdural ; subarachnoid; epidural, intraventricular, parenchymal] * Ventricular system: [normal in size and morphology for the patient's age* | enlarged | small] * Basal cisterns: [normal* | enlarged | small] * Cerebral parenchyma: [normal*; microvascular changes; infarction; encephalomalacia; gliosis; hemorrhage] * Midline shift: [none* | leftward shift | rightward shift] … Impressions

Rad. Lex tagged content • Rad. Lex is an RSNA lexicon designed to tag concepts for subsequent indexing/retrieval • Atomic concepts only – no compositional grammar • Use CDA <link. Html> non-attested link in narrative block <section>. . . <text>. . . <content>There is focal opacity at the right lung base most likely representing right lower lobe atelectasis. <link. Html href=http: //www. radlex. org/RID 1302 /> <link. Html href=http: //www. radlex. org/RID 28493 /> </content> </text>. . . </section> 4/1/2014 Sup 155 review 28

Relationship to IHE MRRT • Management of Radiology Report Templates is an IHE Profile that (inter alia) defines an HTML scheme for encoding report production templates • Revisions needed for MRRT to use DICOM defined business names, supporting encoding of reports as CDA using Sup 155 • Informative annex in DICOM about use of templates in report production, including MRRT • Should we invite IHE Radiology to submit revised HTML scheme for standardization in Sup 155? 4/1/2014 Sup 155 review 29

DICOM Stuff 4/1/2014 Sup 155 review

What about Part 20? • Designed for mapping SR to CDA • Includes CDA structures also used in Sup 155 templates (e. g. , DICOM Object Catalog, header structures, image references, etc. ) • Suggest general reworking combining current Part 20 with new material in Sup 155, to produce a replacement Part 20 • How to handle new version of current Part 20 material? 4/1/2014 Sup 155 review 31

Conformance • What needs to specified in Part 2 with respect to conformance? • Creator claim of document level templates – That would just be one, for now – How about specific uses and/or RSNA templates? • No expected receiver claims beyond general HL 7 CDA receiver conformance – Not intended to be in the DICOM conformance world 4/1/2014 Sup 155 review 32

Schedule • April 1: WG-06 – review overall strategy • May-Jun: WG-06 webinars for line by line review • Jun 23 -27: WG-06 –Release for PC • Mid July-mid Sept: Public Comment • Sept 8 -12: WG-06 – review comments and necessary revisions • Nov 10 -14: WG-06 – Release for LB • Nov-Jan: Letter Ballot • Jan 12 -16: WG-06 – Final Text Draft • ? ? Publish FT 4/1/2014 • May 5 -9: HL 7 – review with Structured Documents WG, and “clean draft” Supplement • Aug 8 -Sep 8: HL 7 Comment Only ballot • Sep 15 -19: HL 7 – review comments • Dec 12 -Jan 12: HL 7 Comment Only ballot • Jan 19 -23: HL 7 – review comments and Final Text Draft Sup 155 review 33

Should Sup 155 be DSTU? • DICOM generally has not found DSTU to be useful • HL 7 uses it regularly, especially for CDA IG’s 4/1/2014 Sup 155 review 34