Superbug Presentation Definitions Genotypic the DNA makeup Phenotypic

Superbug Presentation


Definitions • Genotypic: the DNA makeup • Phenotypic: the expression of the DNA (i. e. say different proteins) • Antibiotic Resistance (wait for it)

How Many Cells in a Human? • How many cells do you think are in an average human? Take a quess. • 32 Trillion cells (how many zeros? ) • 32, 000, 000 human cells

How Many Bacterial Cells In a Human • 39. 7 trillion bacterial cells in a human • So, there are more bacterial cells in us than human cells • The good thing is most of the bacteria in and on us are good bacteria, they do most of the work to digest our food as well as protection most of the external bacteria on our skin.

PROBLEM ADDRESSING No simple solution allowing “unskilled” and marginally skilled individuals to measure yes/no test to determine if humans and domestic pet are carriers of antibiotic resistant bacteria Running out of new antibiotics Lack of good antibiotic stewardship


Initial Market and Development • Currently there is no simple solution allowing “unskilled” individuals to measure yes/no test to measure the presence or absence of specific resistant bacteria • Almost all of the testing is either sent off to an external lab for animals (as well as humans) to be tested. This typically takes 2 -3 days to turn around sample.
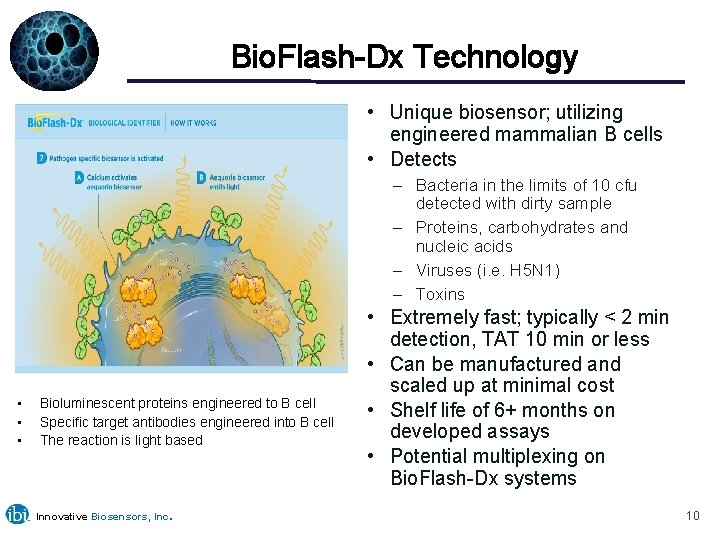
Bio. Flash-Dx Technology • Unique biosensor; utilizing engineered mammalian B cells • Detects – Bacteria in the limits of 10 cfu detected with dirty sample – Proteins, carbohydrates and nucleic acids – Viruses (i. e. H 5 N 1) – Toxins • • • Bioluminescent proteins engineered to B cell Specific target antibodies engineered into B cell The reaction is light based Innovative Biosensors, Inc. • Extremely fast; typically < 2 min detection, TAT 10 min or less • Can be manufactured and scaled up at minimal cost • Shelf life of 6+ months on developed assays • Potential multiplexing on Bio. Flash-Dx systems 10
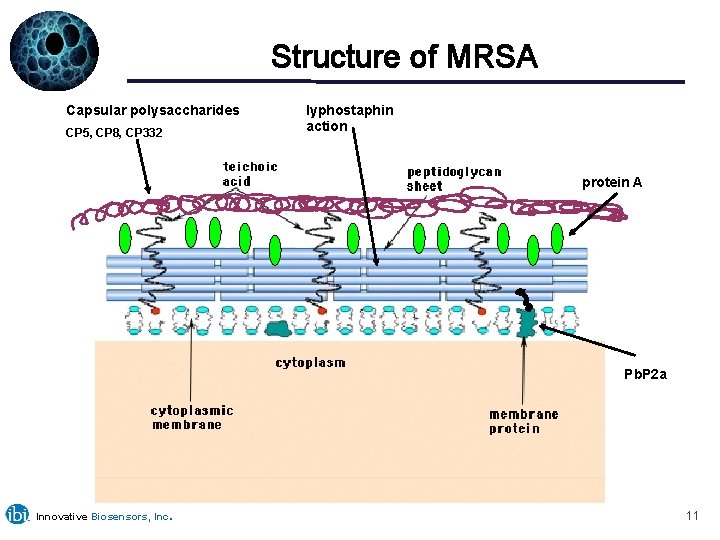
Structure of MRSA Capsular polysaccharides CP 5, CP 8, CP 332 lyphostaphin action protein A Pb. P 2 a Innovative Biosensors, Inc. 11

Typical Time-line for Development and Launch CSM done upfront work Why not start with classic IVD test and regulatory path? Vet Dx goes through FDA but at MUCH lower bar then human testing
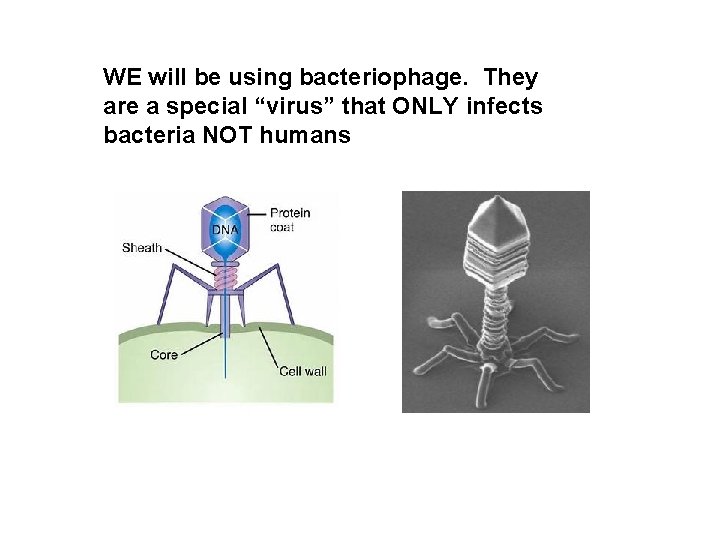
WE will be using bacteriophage. They are a special “virus” that ONLY infects bacteria NOT humans
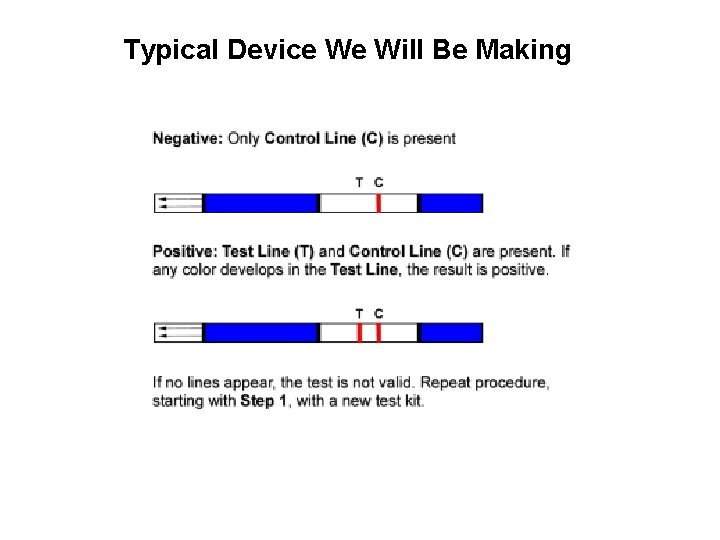
Typical Device We Will Be Making

QUESTIONS?
- Slides: 15