Super chlorination Chlorination beyond the stage of break

Super chlorination • Chlorination beyond the stage of break point. Residual chlorine content may raised up to 0. 5 -2 ppm (normally up to 0. 1 ppm). It is usually applied in case of epidemics. Superchlorinated water may have objectionable taste and require de-chlorination to remove it. • Dose may as high as 10 -15 ppm for 10 -30 min.


Break-point chlorination • Breakpoint chlorination consists of a continual addition of chlorine to the water up to the point where the chlorine enquiry is met and all present ammonia is oxidized, so that only free chlorine remains. This is usually applied for disinfection, but it also has other benefits, such as smell and taste control. In order to reach the breakpoint, a super-chlorination is applied. To achieve this, one uses chlorine concentrations which largely exceed the 1 mg/L concentration required for disinfection ".


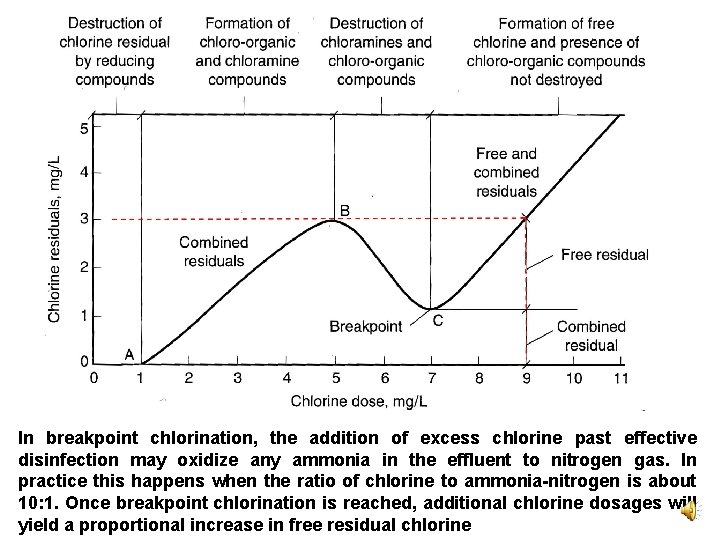
In breakpoint chlorination, the addition of excess chlorine past effective disinfection may oxidize any ammonia in the effluent to nitrogen gas. In practice this happens when the ratio of chlorine to ammonia-nitrogen is about 10: 1. Once breakpoint chlorination is reached, additional chlorine dosages will yield a proportional increase in free residual chlorine

Break-point chlorination • In breakpoint chlorination, the addition of excess chlorine past effective disinfection may oxidize any ammonia in the effluent to nitrogen gas. In practice this happens when the ratio of chlorine to ammonianitrogen is about 10: 1. • Once breakpoint chlorination is reached, additional chlorine dosages will yield a proportional increase in free residual chlorine.



De-chlorination • Partial or complete removal of residual chlorine. • Methods: Chemically by using of sod. Thiosulphate, sod. Bisulphite, sod. Sulphate, activated carbon, ammonia, sulpher dioxide or aeration.

De-chlorination 1. • • • 2. 3. 4. 5. Sulpher dioxide: Used in large plants. SO 2+ Cl 2+H 2 O-----H 2 SO 4+HCl The acid produced is neutralized by water alkalinity. Dose: 1 ppm of SO 2/1 ppm Cl 2. Sod. Thiosulphate (Lab. ) Na. S O +Cl ---Na. S O + Nacl Activated carbon: C+Cl 2+H 2 O---CO 2+HCl. Aeration: remove small amount of Cl 2 and/or HOCl. Ammonia: Economic because it removes Cl 2 and form chloramine. 2 3 2 4 6

Water-Borne Diseases

Diseases Related to Water-borne Diseases Water-based Diseases Water-washed Diseases Water-related Diseases

What is a Water-Borne Disease? “Pathogenic microbes that can be directly spread through contaminated water. ” -CDC � Humans contract waterborne infections by contact with contaminated water or food. � May result from human actions, such as improper disposal of sewage wastes, or extreme weather events like storms and hurricanes. �

Climate Change Promotes Water-borne Disease � Rainfall: transport and dissemination of infectious agents � Flooding: sewage treatment plants overflow; water sources contaminated � Sea level rise: enhances risk of severe flooding � Higher temperatures: Increases growth and prolongs survival rates of infectious agents � Drought: increases concentrations of pathogens, impedes hygiene
- Slides: 16