Summer 2008 Syllabus Biophysics II Cell Biophysics English

Summer 2008 Syllabus Biophysics II Cell Biophysics English: RM 224, 15: 15 -18: 30 Lecture notes with the according references will be published in the www. 1. Basic Cell Biology 2. Membrane Biophysics 3. 4. 5. 6. Intracellular Transport Active and Passive Physics of the Cytoskeleton Neurophysics Photosynthesis (parts are courtesy of Monika Möddel, rest collected by Carsten Selle )

Diffusion – Brownian motion Botanist Robert Brown noticed in 1828 that pollen grains (~ 1 µm) suspended in water do a peculiar „dance“ visible with his microscope. He assumed that he was observing some life process. But the pollen never stopped moving (and were not possible to be killed, even by boiling. Then he found that all other material of this size (which we call colloidal particles) performs this motion. • Spontaneous spreading of matter due to thermal motion • One type of transport phenomenon • E. g. typical molecule of air (N 2) with m=4. 7 x 10 -26 kg at room temperature (290 K): → Effective transport systems using this speed? • NO: Each molecule moves fast, but cannot move very far in a straight line without colliding with another particle → Random motion

Diffusion – Brownian motion • For a particle with step size ±δ , equal probability in both directions and one step every t seconds, one yields for the mean square distance (one dimension): Distance from origin Diffusion coefficient steps and gets • This kind of diffusion is called „normal diffusion“ (linear t dependence of MSD) D: diffusion coefficient

Diffusion – Brownian motion ick‘s first equation of diffusion Fick‘s first diffusion law With the conservation of mass This gives the diffusion equation (or Fick‘s second equation): The step size distribution is a solution to the diffusion equation, with the initial condition n(r, 0)=d(r), here in two dimensions Conveniently, the mean square displacement (MSD) characterizes diffusion.

Diffusion – Brownian motion Diffusion speed depends on friction z (Einstein relation) In three dimensions, the friction coefficient z depnds on a, the radius of a diffusing sphere, with the viscosity h: z=6 pha (Stokes relation). In 2 D, z is not well defined. If one considers, however, a 2 D membrane with viscosity h within a 3 D liquid, the diffusion cooefficient of a cylinderical protein with height h and radius a is given by the following expression: With h‘, the viscosity of the aqueous phase (h>>h‘). Note that D depends logarithmically on a within this approximation. P. G. Safman & M. Delbrück, PNAS 72 (1975), 3111
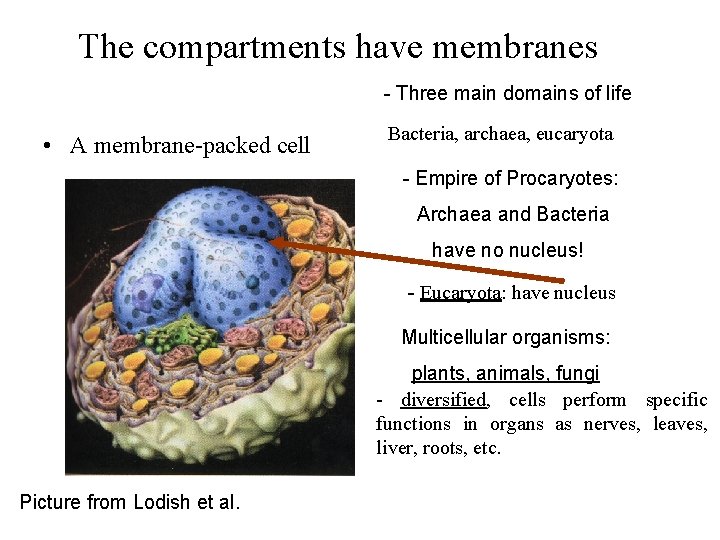
The compartments have membranes - Three main domains of life • A membrane-packed cell Bacteria, archaea, eucaryota - Empire of Procaryotes: Archaea and Bacteria have no nucleus! - Eucaryota: have nucleus Multicellular organisms: plants, animals, fungi - diversified, cells perform specific functions in organs as nerves, leaves, liver, roots, etc. Picture from Lodish et al.
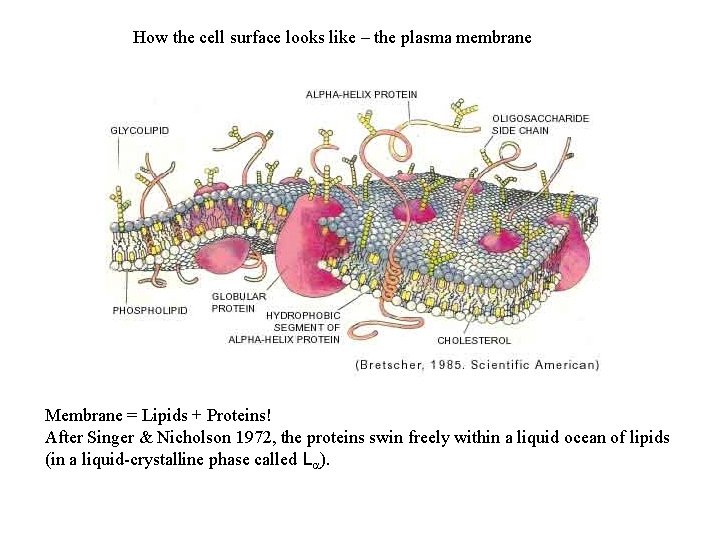
How the cell surface looks like – the plasma membrane Membrane = Lipids + Proteins! After Singer & Nicholson 1972, the proteins swin freely within a liquid ocean of lipids (in a liquid-crystalline phase called La).

History of membrane models Synopsis from: O. G. Mouritsen, Life – as a Matter of Fat Increasing complexity!!
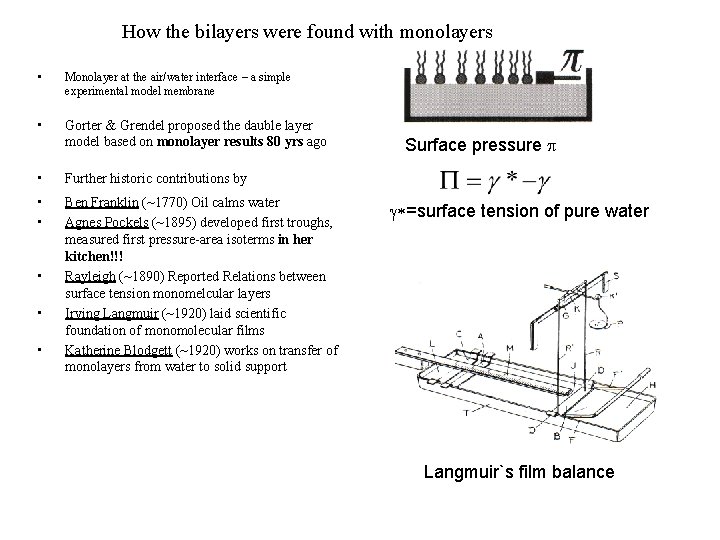
How the bilayers were found with monolayers • Monolayer at the air/water interface – a simple experimental model membrane • Gorter & Grendel proposed the dauble layer model based on monolayer results 80 yrs ago • Further historic contributions by • • Ben Franklin (~1770) Oil calms water Agnes Pockels (~1895) developed first troughs, measured first pressure-area isoterms in her kitchen!!! Rayleigh (~1890) Reported Relations between surface tension monomelcular layers Irving Langmuir (~1920) laid scientific foundation of monomolecular films Katherine Blodgett (~1920) works on transfer of monolayers from water to solid support • • • Surface pressure p g*=surface tension of pure water Langmuir`s film balance
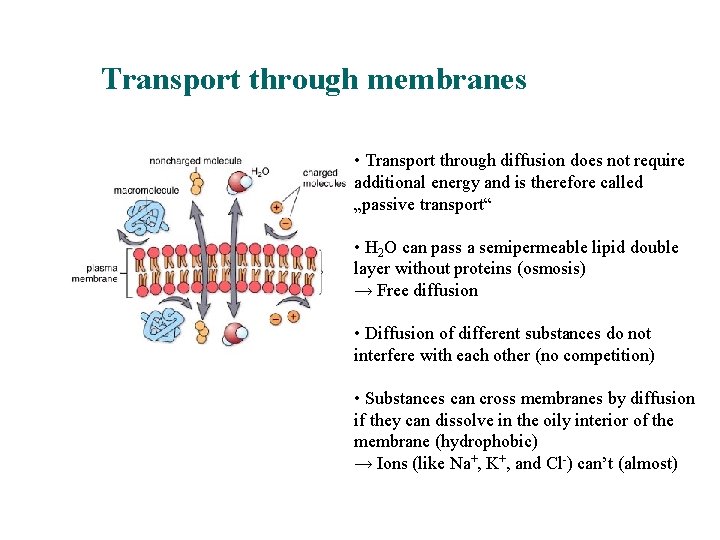
Transport through membranes • Transport through diffusion does not require additional energy and is therefore called „passive transport“ • H 2 O can pass a semipermeable lipid double layer without proteins (osmosis) → Free diffusion • Diffusion of different substances do not interfere with each other (no competition) • Substances can cross membranes by diffusion if they can dissolve in the oily interior of the membrane (hydrophobic) → Ions (like Na+, K+, and Cl-) can’t (almost)
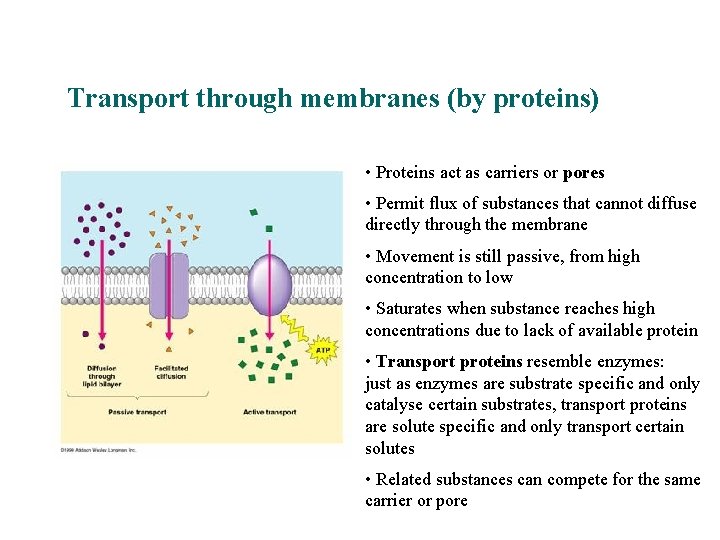
Transport through membranes (by proteins) Facilitated diffusion of ions • Proteins act as carriers or pores • Permit flux of substances that cannot diffuse directly through the membrane • Movement is still passive, from high concentration to low • Saturates when substance reaches high concentrations due to lack of available protein • Transport proteins resemble enzymes: just as enzymes are substrate specific and only catalyse certain substrates, transport proteins are solute specific and only transport certain solutes • Related substances can compete for the same carrier or pore
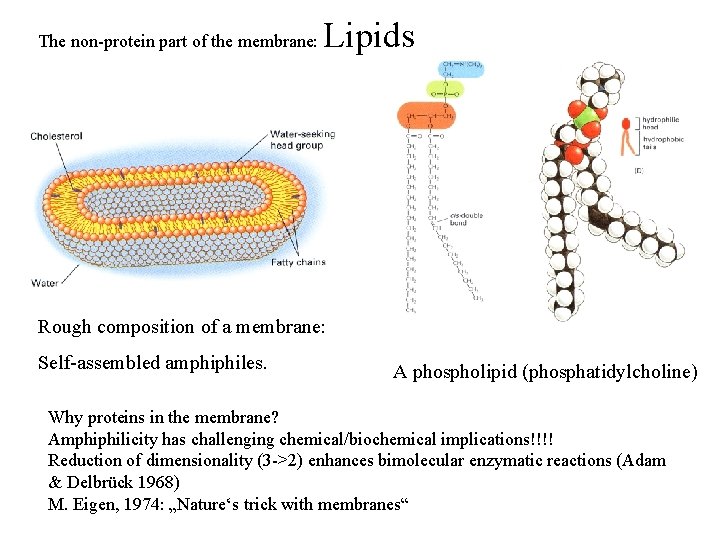
The non-protein part of the membrane: Lipids Rough composition of a membrane: Self-assembled amphiphiles. A phospholipid (phosphatidylcholine) Why proteins in the membrane? Amphiphilicity has challenging chemical/biochemical implications!!!! Reduction of dimensionality (3 ->2) enhances bimolecular enzymatic reactions (Adam & Delbrück 1968) M. Eigen, 1974: „Nature‘s trick with membranes“
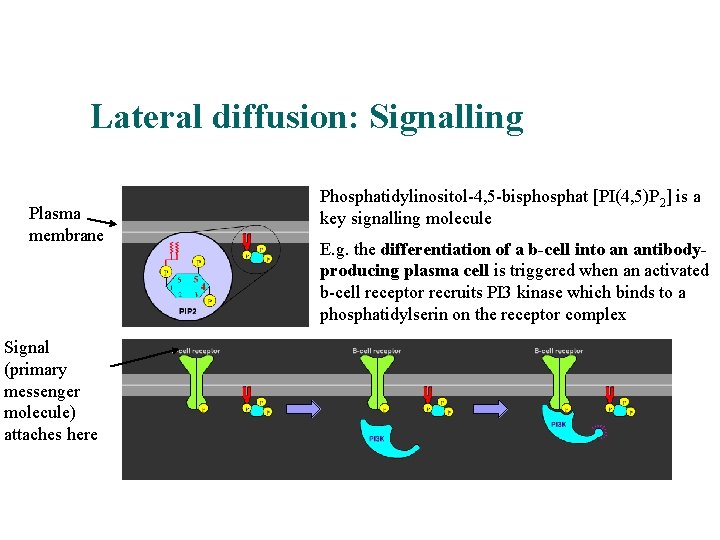
Lateral diffusion: Signalling An example Plasma membrane Signal (primary messenger molecule) attaches here Phosphatidylinositol-4, 5 -bisphosphat [PI(4, 5)P 2] is a key signalling molecule E. g. the differentiation of a b-cell into an antibodyproducing plasma cell is triggered when an activated b-cell receptor recruits PI 3 kinase which binds to a phosphatidylserin on the receptor complex
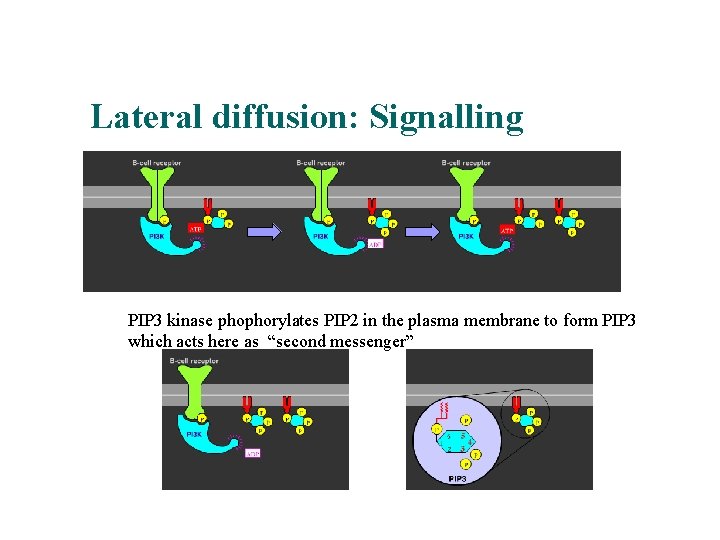
Lateral diffusion: Signalling PIP 3 kinase phophorylates PIP 2 in the plasma membrane to form PIP 3 which acts here as “second messenger”
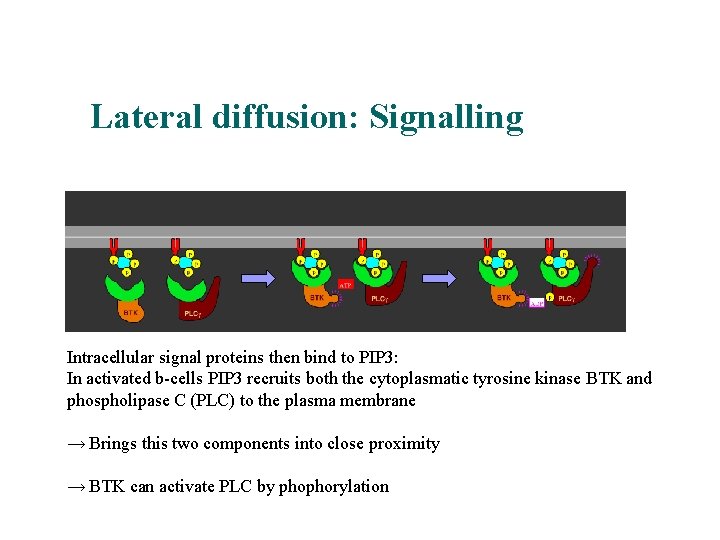
Lateral diffusion: Signalling Intracellular signal proteins then bind to PIP 3: In activated b-cells PIP 3 recruits both the cytoplasmatic tyrosine kinase BTK and phospholipase C (PLC) to the plasma membrane → Brings this two components into close proximity → BTK can activate PLC by phophorylation
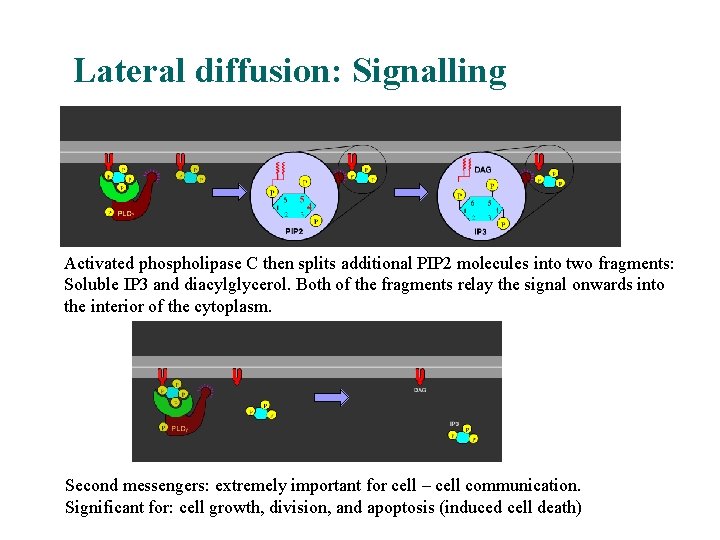
Lateral diffusion: Signalling Activated phospholipase C then splits additional PIP 2 molecules into two fragments: Soluble IP 3 and diacylglycerol. Both of the fragments relay the signal onwards into the interior of the cytoplasm. Second messengers: extremely important for cell – cell communication. Significant for: cell growth, division, and apoptosis (induced cell death)

Anomalous diffusion is a diffusion-like process characterised by a non-linear dependence of the mean squared deviation (MSD) of a particle on time Example: thermal motion of colloidal tracer particles in entangled actin filament (Factin) • Particle radius is comparable to the mesh size of the F-actin network • In this regime, the ensemble-averaged MSD of the particles is proportional to where and depends only on the ratio of the probe radius to mesh size • By directly imaging hundreds of particles over 20 min it‘s found that this anomalous subdiffusion is due to the dynamics of infrequent and large jumps particles make between distinct pores in the network
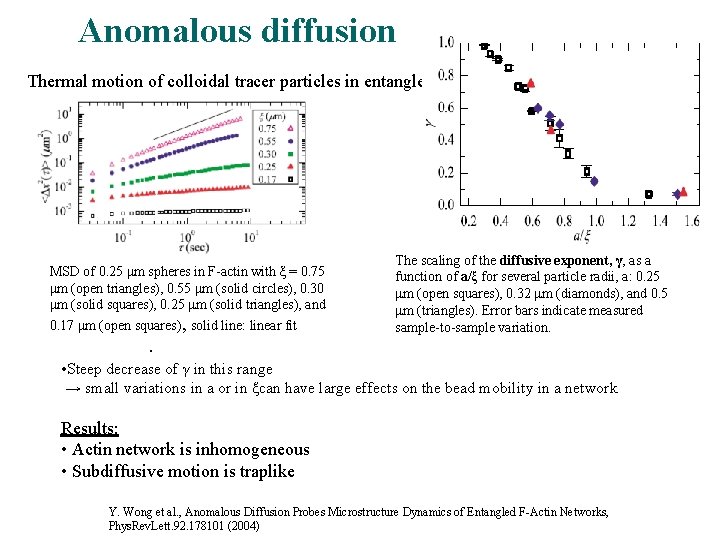
Anomalous diffusion Thermal motion of colloidal tracer particles in entangled F-actin MSD of 0. 25 μm spheres in F-actin with ξ = 0. 75 μm (open triangles), 0. 55 μm (solid circles), 0. 30 μm (solid squares), 0. 25 μm (solid triangles), and 0. 17 μm (open squares), solid line: linear fit The scaling of the diffusive exponent, γ, as a function of a/ξ for several particle radii, a: 0. 25 μm (open squares), 0. 32 μm (diamonds), and 0. 5 μm (triangles). Error bars indicate measured sample-to-sample variation. . • Steep decrease of γ in this range → small variations in a or in ξcan have large effects on the bead mobility in a network Results: • Actin network is inhomogeneous • Subdiffusive motion is traplike Y. Wong et al. , Anomalous Diffusion Probes Microstructure Dynamics of Entangled F-Actin Networks, Phys. Rev. Lett. 92. 178101 (2004)
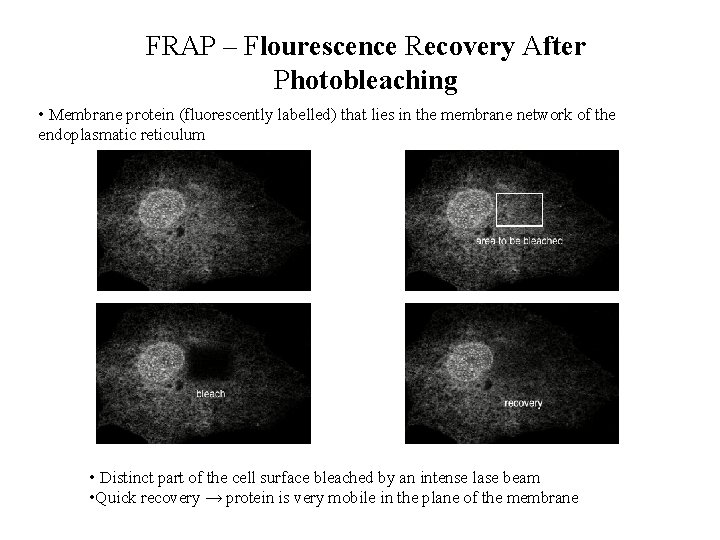
FRAP – Flourescence Recovery After Photobleaching • Membrane protein (fluorescently labelled) that lies in the membrane network of the endoplasmatic reticulum • Distinct part of the cell surface bleached by an intense lase beam • Quick recovery → protein is very mobile in the plane of the membrane

Graphical presentation of data collected during a FRAP experiment D = w 2/4 t 1/2 With w, the spot width and t 1/2, time required to recover half of its original intensity (Y/ X) x 100 = % recovery (almost never reaches 100% in practice) X: Percentage of fluorescence lost due to photobleaching Y: Amount of fluorescence that returned to the bleached area lateral mobility is determined by the slope of the curve
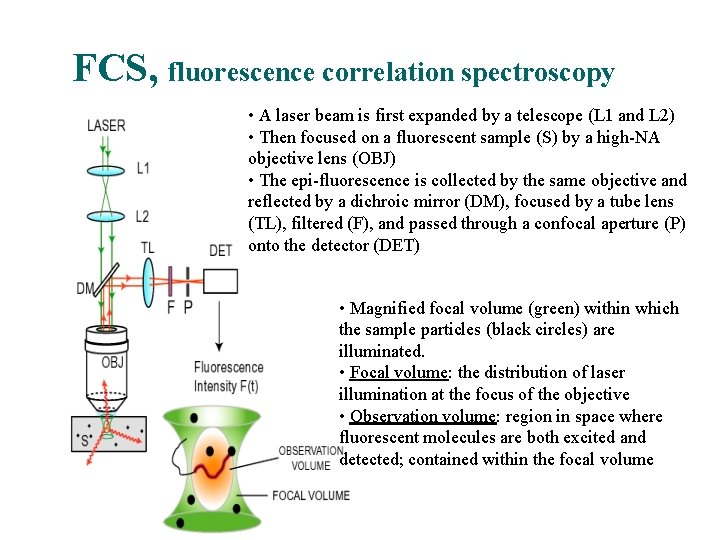
FCS, fluorescence correlation spectroscopy • A laser beam is first expanded by a telescope (L 1 and L 2) • Then focused on a fluorescent sample (S) by a high-NA objective lens (OBJ) • The epi-fluorescence is collected by the same objective and reflected by a dichroic mirror (DM), focused by a tube lens (TL), filtered (F), and passed through a confocal aperture (P) onto the detector (DET) • Magnified focal volume (green) within which the sample particles (black circles) are illuminated. • Focal volume: the distribution of laser illumination at the focus of the objective • Observation volume: region in space where fluorescent molecules are both excited and detected; contained within the focal volume
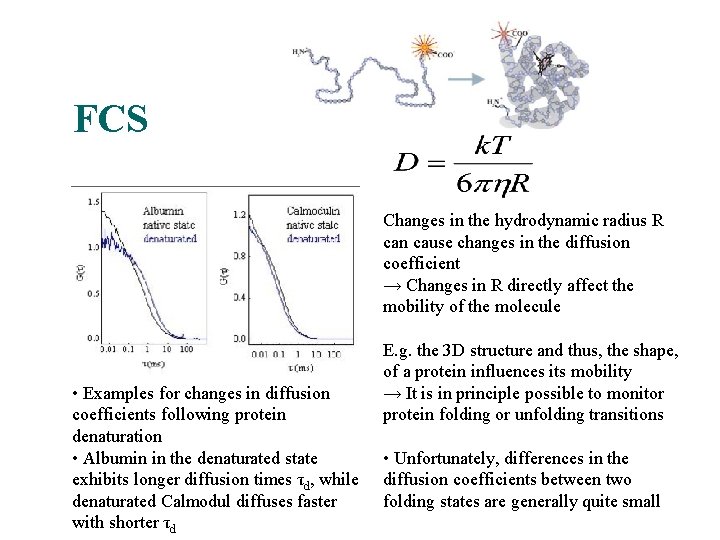
FCS A nice application, in principle… Changes in the hydrodynamic radius R can cause changes in the diffusion coefficient → Changes in R directly affect the mobility of the molecule • Examples for changes in diffusion coefficients following protein denaturation • Albumin in the denaturated state exhibits longer diffusion times τd, while denaturated Calmodul diffuses faster with shorter τd E. g. the 3 D structure and thus, the shape, of a protein influences its mobility → It is in principle possible to monitor protein folding or unfolding transitions • Unfortunately, differences in the diffusion coefficients between two folding states are generally quite small

SPT – Single Particle Tracking • The MSD can be calculated from the evaluation of recorded tracer trajectories position of particle at time step between two successive pictures of the labeled molecule n # of steps such that N total # of steps in a track Qian et al. , Biophysical Journal 1994
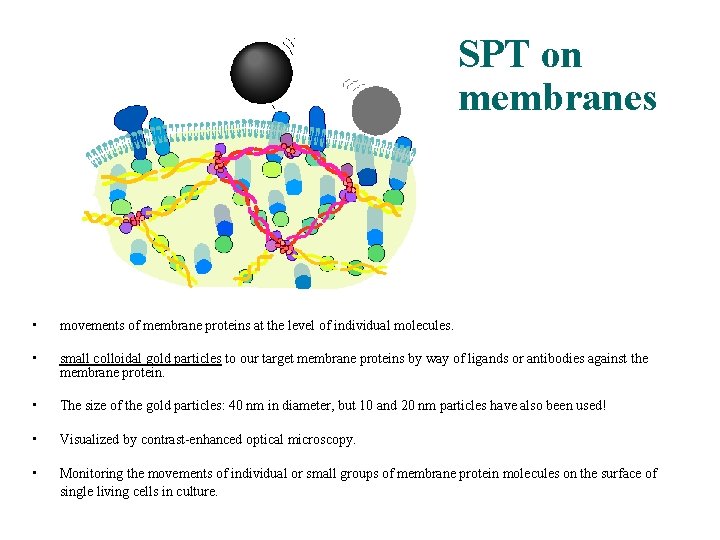
SPT on membranes • movements of membrane proteins at the level of individual molecules. • small colloidal gold particles to our target membrane proteins by way of ligands or antibodies against the membrane protein. • The size of the gold particles: 40 nm in diameter, but 10 and 20 nm particles have also been used! • Visualized by contrast-enhanced optical microscopy. • Monitoring the movements of individual or small groups of membrane protein molecules on the surface of single living cells in culture.
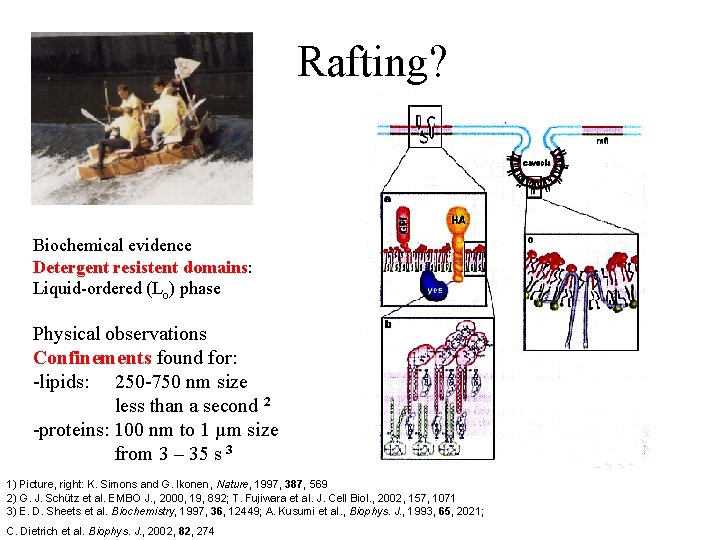
Rafting? Biochemical evidence Detergent resistent domains: Liquid-ordered (Lo) phase Physical observations Confinements found for: -lipids: 250 -750 nm size less than a second 2 -proteins: 100 nm to 1 µm size from 3 – 35 s 3 1) Picture, right: K. Simons and G. Ikonen, Nature, 1997, 387, 569 2) G. J. Schütz et al. EMBO J. , 2000, 19, 892; T. Fujiwara et al. J. Cell Biol. , 2002, 157, 1071 3) E. D. Sheets et al. Biochemistry, 1997, 36, 12449; A. Kusumi et al. , Biophys. J. , 1993, 65, 2021; C. Dietrich et al. Biophys. J. , 2002, 82, 274
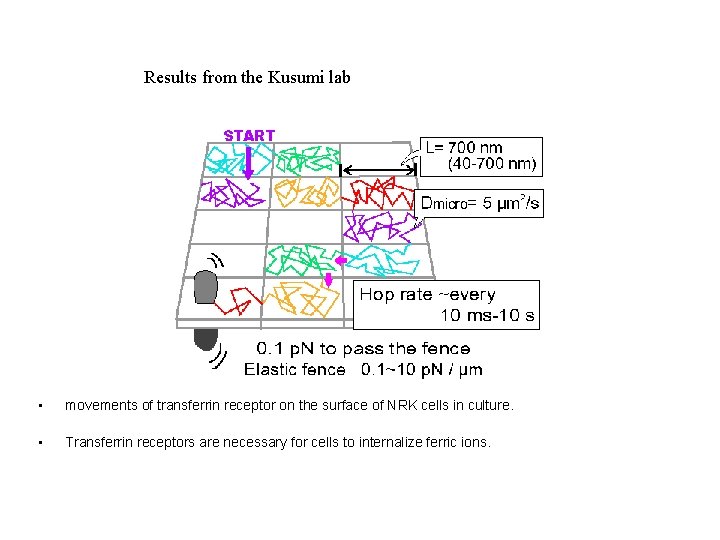
Results from the Kusumi lab • movements of transferrin receptor on the surface of NRK cells in culture. • Transferrin receptors are necessary for cells to internalize ferric ions.

Model on how the “membrane skeleton” can interact with protein motion (A. Kusumi, cf. Web site) A) membrane proteins bound to the cytoskeleton B) Free motion, confined within a mesh from the cytosekeleton C) partitioning into raft-like regions

Homework SPT measurements of a colloidal model protein within a membrane with domains (rafts), Trajectories of the protein are depicted in red. Black area: raft-like ordered lipid phase, white area: liquid-like lipid phase. Bright spot: fluorescent model protein. Consider the differences measured for D and a in these observations: Can you explain them? Draw conclusions about a possible biological significance.
- Slides: 28