Summary The 5 different chemical reactions 1 SynthesisCombination

Summary • The 5 different chemical reactions 1. Synthesis/Combination 2. Decomposition 3. Single Displacement 4. Double Displacement 5. Combustion/Oxidation

Chemical Reactions

Vocabulary: Ion: An atom with a charge Hydrocarbon: A compound that contains Hydrogen and Carbon

Synthesis (combination) aka composition reaction A + B AB Two or more substances combine to form a new compound. 2 H 2(g) + O 2(g) 2 H 2 O(l)

Synthesis (combination) • Medicine • Flavorings • Plastics • High energy fuels

Decomposition A single compound undergoes a reaction that produces two or more simpler substances. AB A + B (NH 4)2 Cr 2 O 7 N 2 + 4 H 2 O + Cr 2 O 3 electricity 2 H 2 O(l) 2 H 2(g) + O 2(g) Electrolysis– the decomposition of a substance by an electric current.

Decomposition • Speed up decomposition • Toxic waste • Waste • Fats • Explosives • Slow down decomposition • Anti-rust • Anti-aging

Single replacement aka Displacement reaction One element replaces a similar element in a compound. A + BC B + AC • Design of batteries 3 Zn + 2 Cu 3 PO 4 6 Cu + Zn 3(PO 4)2

Each potato generates about 0. 5 volts and 0. 2 milliamps. The entire 500 lb battery generated around 5 volts and 4 milliamps.

Double replacement The ions of two compounds exchange places to form two new compounds. AB + CD AD + CB • Purification (barium is poisonous) • Ba. Cl 2 + Mg. SO 4 • Ba 2+(aq)+2 Cl-(aq)+Mg 2+(aq)+SO 42 -(aq) Ba. SO 4 + Mg. Cl 2 Ba. SO 4(s)+Mg 2+(aq)+2 Cl-(aq)
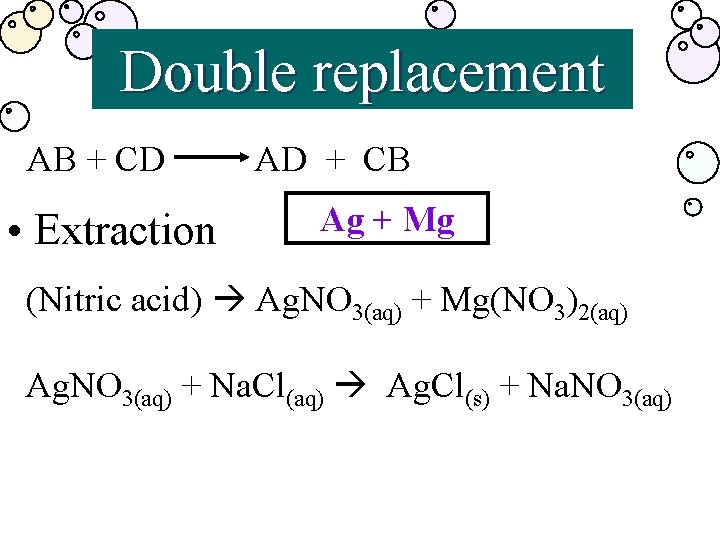
Double replacement AB + CD • Extraction AD + CB Ag + Mg (Nitric acid) Ag. NO 3(aq) + Mg(NO 3)2(aq) Ag. NO 3(aq) + Na. Cl(aq) Ag. Cl(s) + Na. NO 3(aq)

Double replacement AB + CD AD + CB • Neutralizing corrosives • Na. OH + CH 3 COOH Na. C 2 H 3 O 2 +HOH
![Combustion [loss of electrons] Hydrocarbon is burned to react with oxygen to produce CO Combustion [loss of electrons] Hydrocarbon is burned to react with oxygen to produce CO](http://slidetodoc.com/presentation_image_h2/4352d8d8d218fb02903d964038d3a201/image-13.jpg)
Combustion [loss of electrons] Hydrocarbon is burned to react with oxygen to produce CO 2 and H 2 O. • C 4 H 10 + O 2 CO 2 + H 2 O
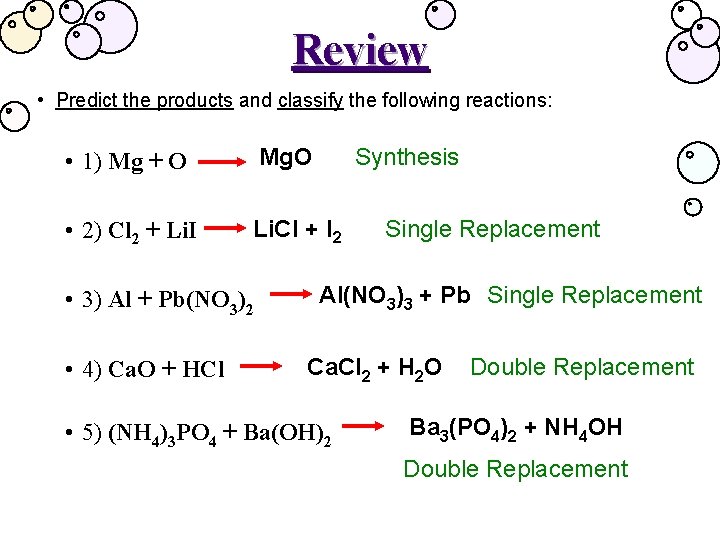
Review • Predict the products and classify the following reactions: • 1) Mg + O Mg. O • 2) Cl 2 + Li. I Li. Cl + I 2 • 3) Al + Pb(NO 3)2 • 4) Ca. O + HCl Synthesis Single Replacement Al(NO 3)3 + Pb Single Replacement Ca. Cl 2 + H 2 O • 5) (NH 4)3 PO 4 + Ba(OH)2 Double Replacement Ba 3(PO 4)2 + NH 4 OH Double Replacement

Ø Practice: • Predict the products and classify the following reactions: • 6) C 4 H 10 + O 2 • 7) H 2 SO 4 + KOH • 8) Cu + Ag. NO 3 • 9) Ag. NO 3 + KCl • 10) Cl 2 + KI CO 2 + H 2 O Combustion K 2 SO 4 + H 2 O Double Replacement Cu(NO 3)2 + Ag Single Replacement KNO 3 + Ag. Cl Double Replacement KCl + I 2 Single Replacement
- Slides: 15