Summary Chapter 2 1 Solution for Hlike atomion











- Slides: 13

Summary Chapter 2 1. Solution for H-like atom/ion (one-electron system) radial & angular functions of Atomic orbitals, electron cloud, quantum numbers (n, l, m, and ms) electronic orbital/spin angular momentum & space quantization. Atomic-orbital wavefunctions are eigenfunctions of such operators as H, L 2, & Lz!

Key points: For H-like atom/ions, the wavefunctions to describe their atomic orbitals (AO s) that are derived from their Schrodinger equations can be characterized using three quantum numbers, i. e. , n, l, m, and symbolized as nlm. ^ ^ They are eigenfunctions of those hermitian operators Ĥ, L 2 (M 2) ^ ), by the following eigenequations, ^ (M and L z z Magnitude of orbital angular momentum Note: Angular momentum L is a vector!

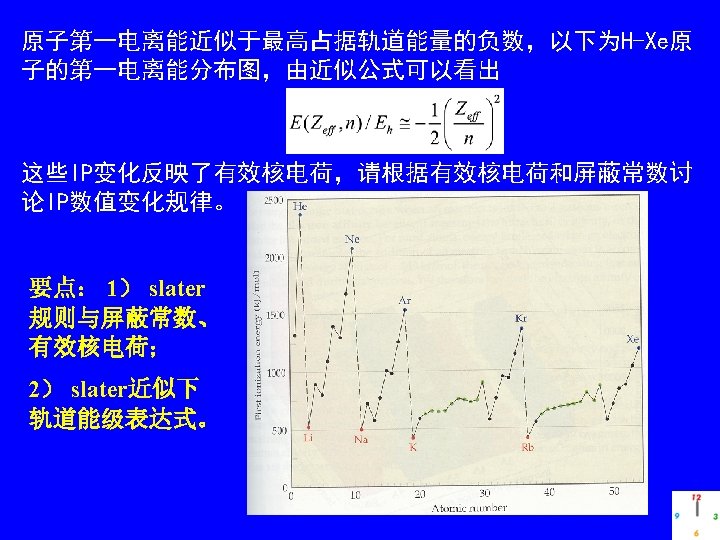
Summary Chapter 2 2. Solution for many-electron atom a) Separation of variables ---- Independent particle approximation (mean field model) i) one-particle eigenequations ii) wavefunction of all electrons = product of one-particle eigenfunctions. iii) one-particle eigenfunctions atomic orbitals of H-like atoms. attainable by HF-SCF process. b) Slater’s approximation: screen constant & effective nuclear charge.

c) Addition of Angular momenta and its physical meaning/applications in understanding electronic states of many-e atom. i) Details of the e-e interactions can be symbolized by L (ML) and S(MS)! ii) Atomic spectral terms: • physical meaning (energy state of an electronic configurations that contain one to several microstates). • derivation of terms for a given electronic configuration (normally simple) containing equivalent electrons and/or nonequivalent electrons. • Pauli exclusion and Hund’s rules, and derivation of the groundstate term for a given electronic configuration.






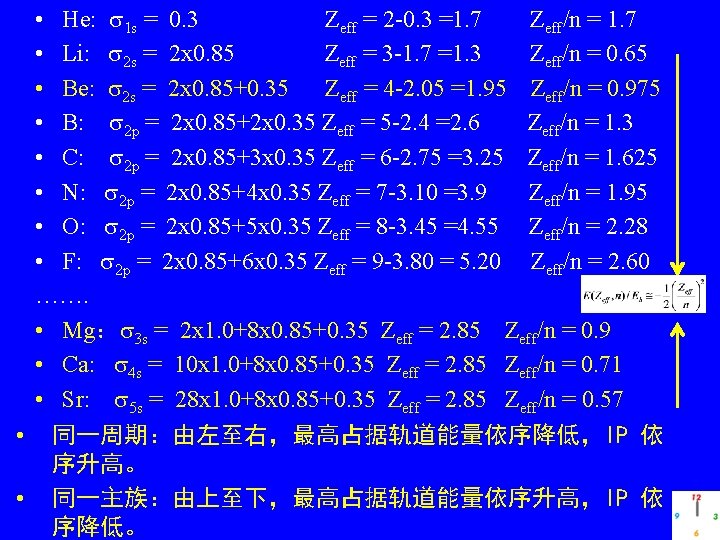
• He: 1 s = 0. 3 Zeff = 2 -0. 3 =1. 7 Zeff/n = 1. 7 • Li: 2 s = 2 x 0. 85 Zeff = 3 -1. 7 =1. 3 Zeff/n = 0. 65 • Be: 2 s = 2 x 0. 85+0. 35 Zeff = 4 -2. 05 =1. 95 Zeff/n = 0. 975 • B: 2 p = 2 x 0. 85+2 x 0. 35 Zeff = 5 -2. 4 =2. 6 Zeff/n = 1. 3 • C: 2 p = 2 x 0. 85+3 x 0. 35 Zeff = 6 -2. 75 =3. 25 Zeff/n = 1. 625 • N: 2 p = 2 x 0. 85+4 x 0. 35 Zeff = 7 -3. 10 =3. 9 Zeff/n = 1. 95 • O: 2 p = 2 x 0. 85+5 x 0. 35 Zeff = 8 -3. 45 =4. 55 Zeff/n = 2. 28 • F: 2 p = 2 x 0. 85+6 x 0. 35 Zeff = 9 -3. 80 = 5. 20 Zeff/n = 2. 60 ……. • Mg: 3 s = 2 x 1. 0+8 x 0. 85+0. 35 Zeff = 2. 85 Zeff/n = 0. 9 • Ca: 4 s = 10 x 1. 0+8 x 0. 85+0. 35 Zeff = 2. 85 Zeff/n = 0. 71 • Sr: 5 s = 28 x 1. 0+8 x 0. 85+0. 35 Zeff = 2. 85 Zeff/n = 0. 57 • 同一周期:由左至右,最高占据轨道能量依序降低,IP 依 序升高。 • 同一主族:由上至下,最高占据轨道能量依序升高,IP 依 序降低。


