Successive Ionization Energies One after another Subsequent ionisation

Successive Ionization Energies One after another

Subsequent ionisation energies and their relationship to electron shells • • 2 nd and 3 rd ionisation energy. X+(g) X 2+ (g) +e. X 2+ (g) X 3+ (g) +e. Successive ionization energy can give information about an element. • Such as the number of electrons in outer shell.
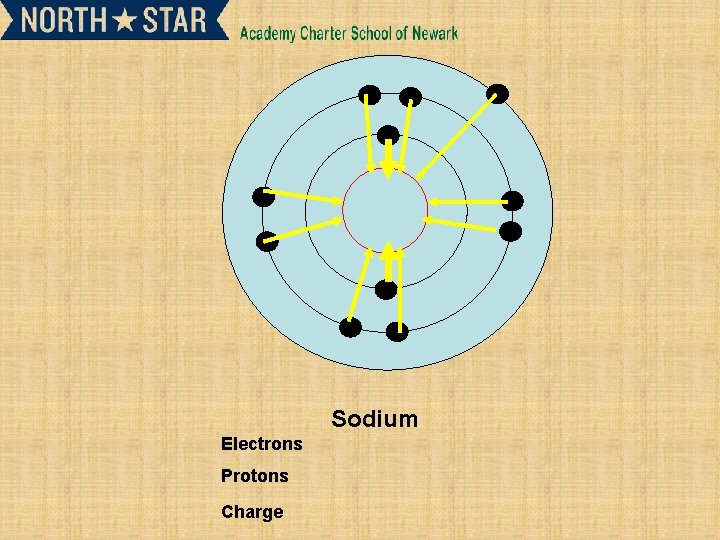
Sodium Electrons Protons Charge
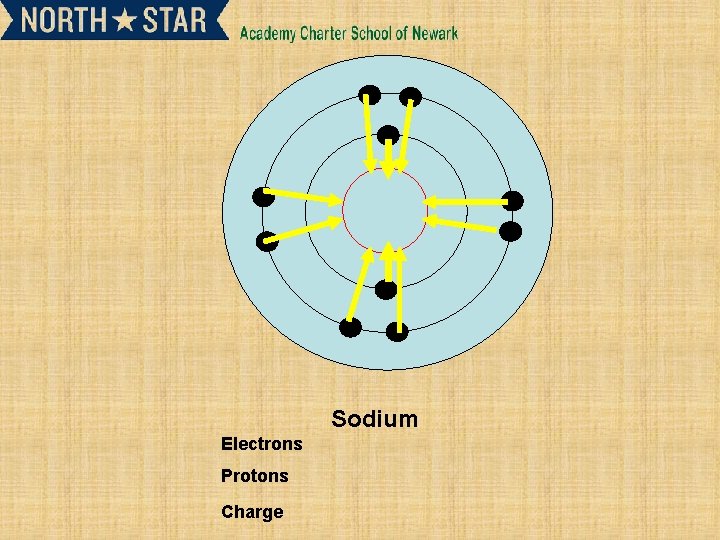
Sodium Electrons Protons Charge
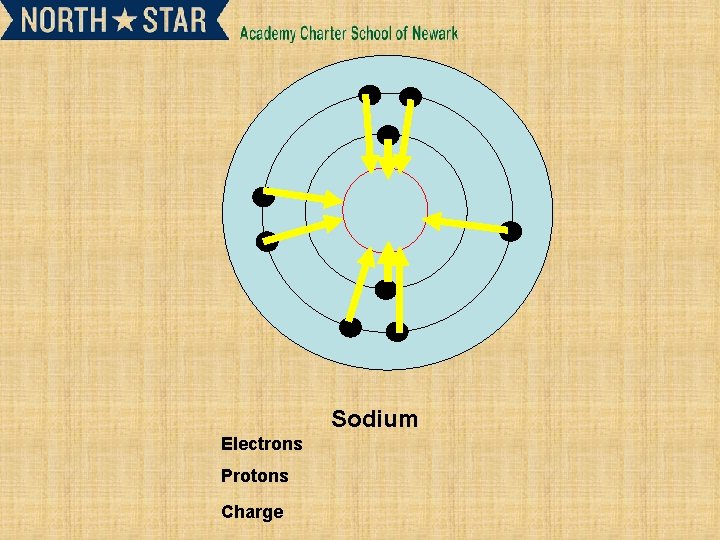
Sodium Electrons Protons Charge
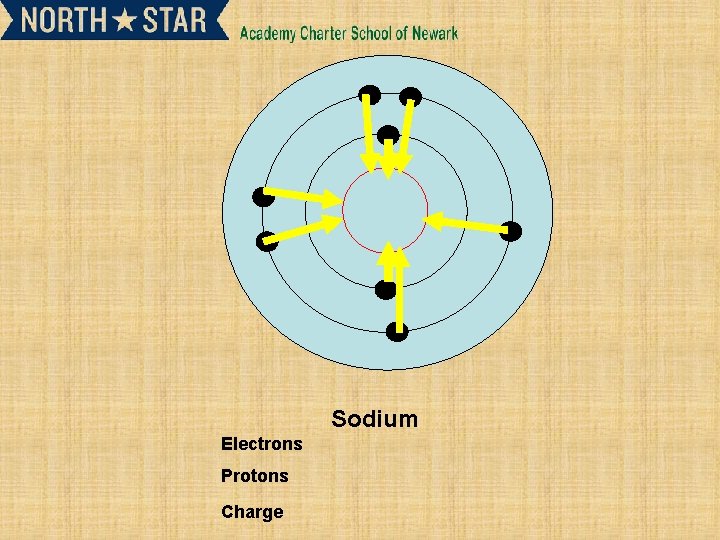
Sodium Electrons Protons Charge
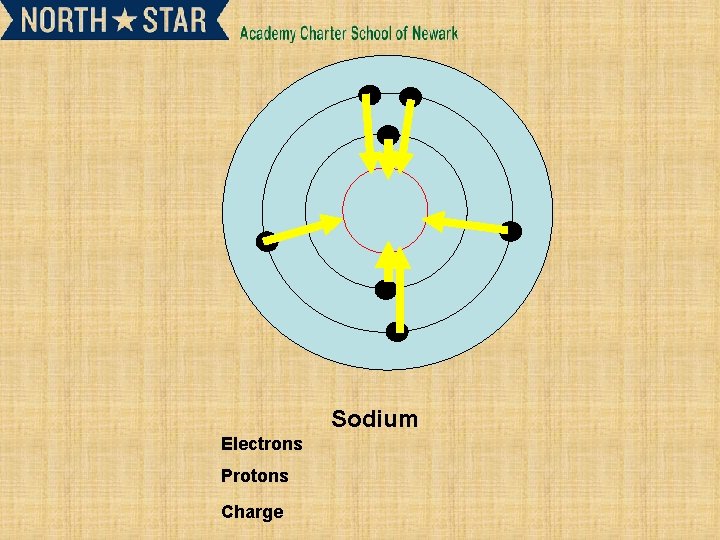
Sodium Electrons Protons Charge
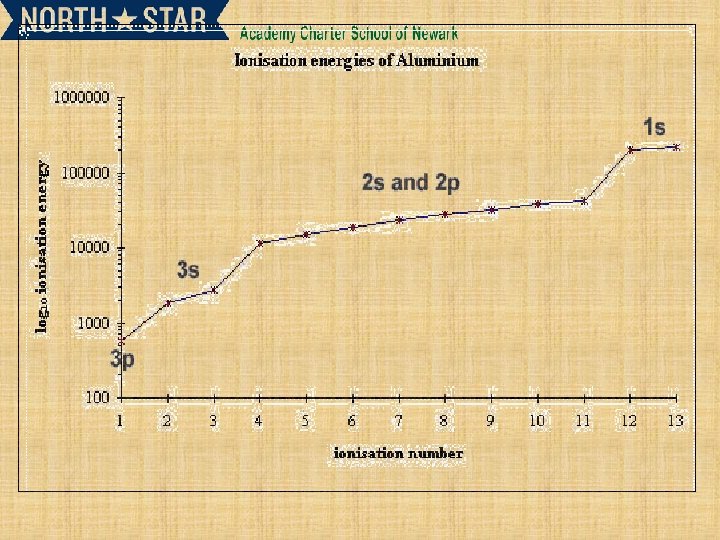

SUCCESSIVE IONISATION ENERGIES Defining second ionization energy Second ionization energy is defined by the equation: It is the energy needed to remove an electron from a +1 ion, to make a +2 ion Follows the first ionization energy
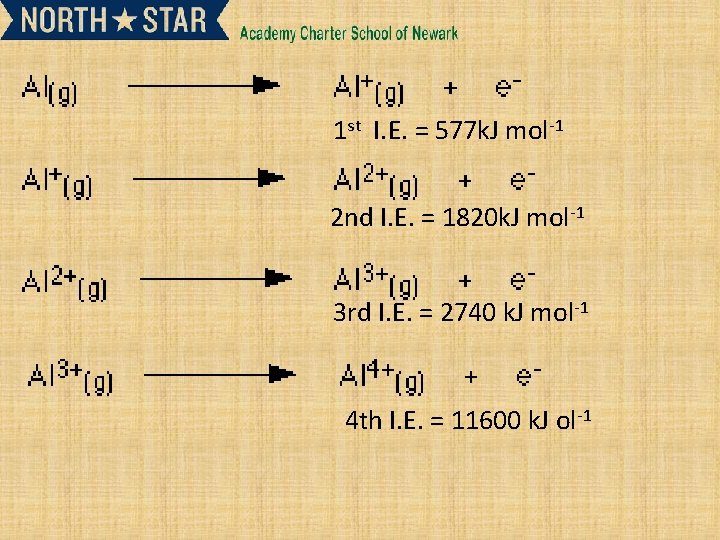
1 st I. E. = 577 k. J mol-1 2 nd I. E. = 1820 k. J mol-1 3 rd I. E. = 2740 k. J mol-1 4 th I. E. = 11600 k. J ol-1

Why do successive ionization energies get larger? Why is the fourth ionization energy of aluminum so large? 1 s 22 p 63 s 23 px 1.

Using ionization energies to work out which group an element is in Magnesium (1 s 22 p 63 s 2) is in group 2 of the Periodic Table and has successive ionization energies: ).
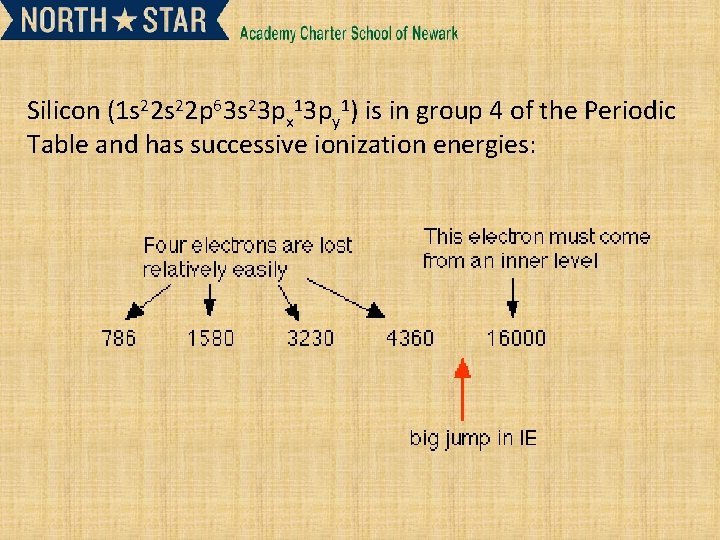
Silicon (1 s 22 p 63 s 23 px 13 py 1) is in group 4 of the Periodic Table and has successive ionization energies:
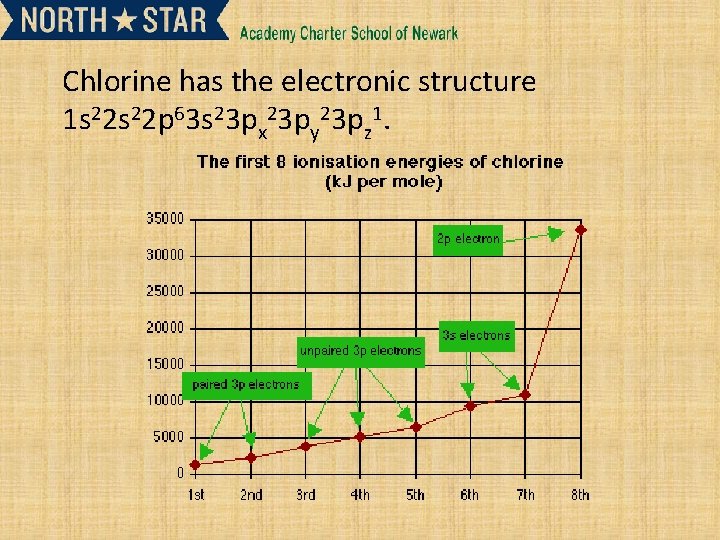
Chlorine has the electronic structure 1 s 22 p 63 s 23 px 23 py 23 pz 1.
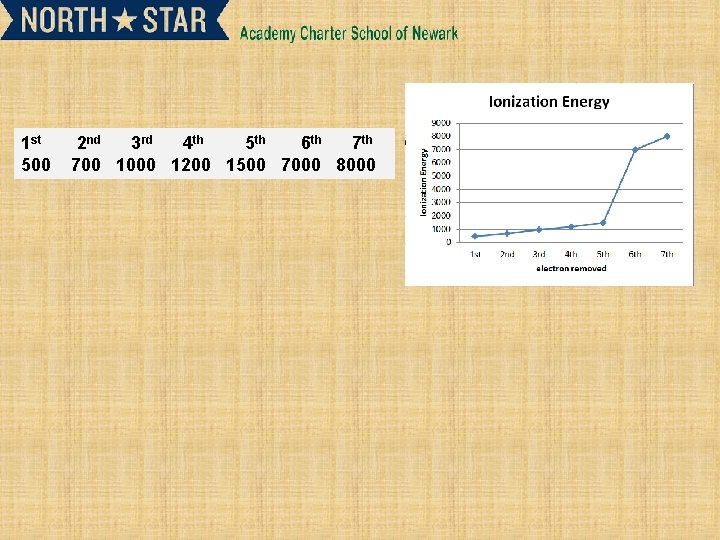
1 st 500 2 nd 3 rd 4 th 5 th 6 th 700 1000 1200 1500 7000 8000
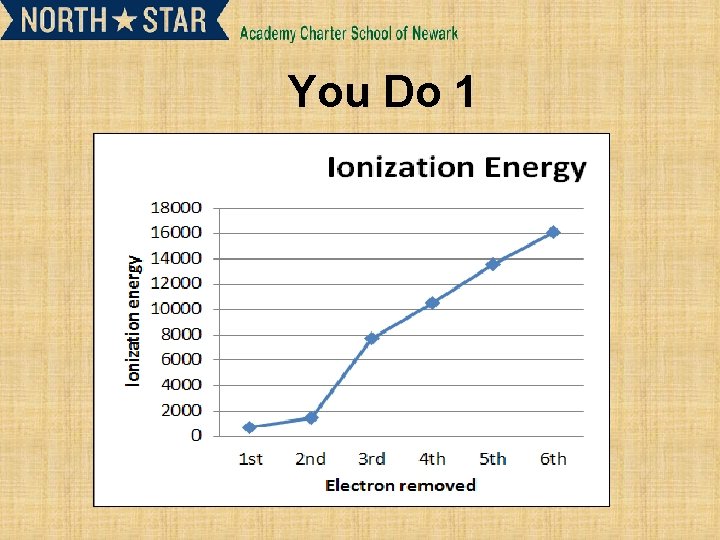
You Do 1
- Slides: 16